Summary Corona Virus Update by H. Robert Silverstein, MD, FACC for the Preventive Medicine Center
Updated version — available as pdf and original docx.
Clyde W. Yancy, MD, Vice Dean for Diversity and Inclusion Chief of Cardiology in the Department of Medicine, Feinberg School of Medicine at Northwestern University in Chicago
Clyde W. Yancy, MD: “I’m exhausted by the stress; disheartened by the toll on human life; concerned deeply about the exposure to healthcare workers- BUT, I am emboldened by the display of courage, selflessness, compassion, and sacrifice that I see in physicians, nurses and health care workers across the country.”
“It is not a case of ‘don’t confuse me with the facts’, but the best clinical insights exceed so called knowledge by at least one step.”
“Early 2020 saw the world break into what has been described as a “war-like situation”: A pandemic, caused by the severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2), the likes of which majority of the living generations across most of the planet have not ever seen. This pandemic has downed economies and resulted in hundreds of thousands of deaths.” Tokyo University of Science 7/1/21
History, precedents, similarities, virus structure and invasion, pathology, physiology, lethality vs safety in perspective, China, geography, EU vs USA comparison, time-line, media, politics, pandemic modelling, symptoms, lockdown, economics, joblessness, vascular-platelet-glycocalyx clotting, testing, ventilators, medications, vaccines, supplements, diet:
This 2019 corona virus CoV2-19 is an entirely new RNA virus with 30 proteins. Corona viruses have the largest known viral genome. The RNA of a corona virus is single-stranded. The word “VIRUS” means “poison.” A human cell has 20,000 different proteins. Being an RNA virus, it is similar to hepatitis C; it is not a DNA virus like hepatitis B. There are 200 viruses that can cause the common cold and several of these are corona viruses. “Corona” is Latin for “crown” which is how the virus looks in the microscope as if it has an encircling crown. The specific CoV2-19 genetic RNA fact and its “SPIKE” projections will affect anti-viral treatment design and decisions. That virus spike binds to and fuses with host cells. “The SARS-CoV-2 spike protein trimer is only ~10nm in size (1/100,000 of a millimeter) and there are approximately 100 of these on the surface of a single viral particle, which itself is about 100 nm in diameter.” CoV2-19 was detected by it having a new genetic sequence as recognized by GenBank—it may have been around for a thousand years, but it is just now discovered. The Chinese symbol for it is pronounced “wayGee” and means both “crisis” and “opportunity”: two sides of the same coin. The first known novel and important coronavirus was called SARS = Severe Acute Respiratory syndrome. There are only 2 known previous serious corona virus outbreaks: SARS and Middle East respiratory syndrome = MERS, the latter epidemic was smaller, but with a 1/3 (33%!) death rate!
Why some people are less naturally resistant to COVID-19
by Bob Yirka , Medical Xpress 9/29/21
Transmission electron micrograph of SARS-CoV-2 virus particles, isolated from a patient. Image captured and color-enhanced at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID
A large team of researchers affiliated with a host of institutions in the U.K. and Brazil has partially solved the mystery of why some people are less naturally resistant to COVID-19 than others. In their paper published in the journal Science, the group describes their study of the interferon system and the role it plays in combating the SARS-CoV-2 virus.
As the global pandemic has unfolded, it has become clear that some people have much more serious symptoms when contracting COVID-19 than others. Indeed, some people have been found to exhibit no symptoms at all, while others become so sick that they die. In this new effort, the researchers conducted extensive interferon-stimulated gene expression screening to isolate possible enzymes involved in alerting the immune system to an infection. Interferons are signaling proteins that alert the body when invasive entities such as bacteria and viruses are detected.
The work by the researchers led them to OAS1, an enzyme that reacts to interferon signaling by calling for an immune response when the SARS-CoV-2 virus is detected. Prior research has shown that OAS1 attaches to membranes using a prenyl group as part of the signaling process. Prior research has also shown that this signaling can inhibit replication of the SARS-CoV-2 virus. Noting its value in protecting people against COVID-19, the researchers looked at the transcriptomes of 500 COVID-19 patients who had experienced a wide range of symptoms and found that those who did not have prenylated OAS1 experienced much more severe symptoms. Why some people are born without the enzyme is still a mystery, but the work by the team could help lead to new types of vaccines against COVID-19 and other types of infections.
Intrigued by their findings, the researchers turned their attention to another mammal possibly involved in the pandemic—the horseshoe bat. They found it did not possess the form of prenylated OAS1 that protects humans from the virus, helping to explain why the virus is so deadly to that species. The finding could also help explain why the bats are such prolific hosts to a variety of viruses.
More information: Arthur Wickenhagen et al, A prenylated dsRNA sensor protects against severe COVID-19, Science (2021). DOI: 10.1126/science.abj3624
A REMARABLY lucid and up-to-date VIDEO explanation of of corona virus variants, their genetic make-up, infectivity, and epidemiology. BASIC RESEARCH by Jeremy Kamil of Louisiana State University as of 3/34/21. In case the link does not work, go to VuMedi to view this:
https://www.vumedi.com/video/convergent-evolution-in-sars-cov-2-what-do-7-of-the-emerging-variants-have-in-common-is-the-virus-ru/?token=64cc855f-7b31-4533-bfeb-bdf40e2f7c7f&utm_source=COVID%20Interests%20Criteria_79170&utm_medium=Video&utm_campaign=%2803/29%20covid%29%20Convergent%20Evolution%20in%20SARS-CoV-2%3A%20What%20Do%207%20of%20the%20Emerging%20Variants%20Have%20in%20Common%3F%20Is%20the%20Virus%20Running%20Out%20of%20New%20Major%20Adaptions%3F&utm_content=Convergent%20Evolution%20in%20SARS-CoV-2%3A%20What%20Do%207%20of%20the%20Emerging%20Variants%20Have%20in%20Common%3F%20Is%20the%20Virus%20Running%20Out%20of%20New%20Major%20Adaptations%3F&utm_term=COVID-19%20Prevention&link_data=eyJidWxrX21haWxfYWN0aW9uIjoiYyIsInJlY2lwaWVudF9pZCI6MTE4MzMwNTA3OSwibWFpbF9pZCI6NzkxNzB9%3A1lQte7%3AUmkEMNQUCzMTwB3o3t9K1bhcuQo&mail_id=79170
From Quora internet chat box 8/6/21: “ … The first human coronavirus sample was identified 56 years ago if I recall correctly. mRNA vaccine research dates from 1990. They figured out how to stabilize synthetic mRNA around 2001–2002, founded Moderna in 2003. SARS research kicked into high gear in 2001, and then the SARS near pandemic of 2011 …”
Here is a perspective on plagues by G Pascal in the Lancet Infectious Disease 7/2021 21: 928 reviewing the book by John Froude titled ‘Plagued.’ “The devastation from plagues is far worse. More soldiers have lost their lives from disease than the wars they thought in. At least 300 million people have died from bubonic plague alone. When yellow fever hit Memphis, Tennessee, in 1878, it became a “city of corpses,” and malaria has killed between 200-300 million people in the 20th century alone. Smallpox spread around the world within 200 years and last year it took CoV2-19 just 4 months.”
Here is SUPERB reporting from ‘Vanity Fair’ 5/27/21 by Kartherine Eban regarding the possibility of a lab-leak of the Corona Virus resulting in the CoV2-19 pandemic. A small amount of politics is therein.
A profound and entire Wall Street Journal 6/6/21 editorial was written by the heavily credentialled Quay and Muller regarding the origin of the Wuhan Corona Virus CoV2-19
An article on the origin of the Wuhan Corona Virus:
https://img-prod.tgcom24.mediaset.it/images/2020/02/16/114720192-5eb8307f-017c-4075-a697-348628da0204.pdf
A 9/25/21 ‘Nature News’ article on Laos bats BANAL-52 SARS virus having 98.6% SIMILARITY to the original CoV2-19 virus + the also having (the new) ability to bind to the Receptor Binding Domain (RBD) ACE II:
“Viruses found in Laos bats are closest known relatives to SARS-CoV-2
By Rachael Rettner
Researchers have discovered coronaviruses lurking in Laotian bats that appear to be the closest known relatives to SARS-CoV-2, the virus that causes COVID-19, found to date, according to news reports.
In a new study, researchers from the Pasteur Institute in France and the University of Laos captured 645 bats from limestone caves in northern Laos and screened them for viruses related to SARS-CoV-2. They found three viruses — which they dubbed BANAL-52, BANAL-103 and BANAL-236 — that infected horseshoe bats and shared more than 95% of their overall genome with SARS-CoV-2.
One of the viruses, BANAL-52, was 96.8% identical to SARS-CoV-2, according to Nature News. That makes BANAL-52 more genetically similar to SARS-CoV-2 than any other known virus. Previously, the closest known relative to SARS-CoV-2 was RaTG13, which was found in horseshoe bats in 2013 and shares 96.1% of its genome with SARS-CoV-2, Nature News reported.
What’s more, all three of the newly discovered viruses are more similar to SARS-CoV-2 in a key part of their genome — called the receptor binding domain (RBD) — than other known viruses. The RBD is the part of the virus that allows it to bind to host cells. With SARS-CoV-2, the RBD binds to a receptor known as ACE2 on human cells, and the virus uses this receptor as a gateway into cells.
Critically, the new study found that BANAL-52, BANAL-103 and BANAL-236 can bind to ACE2 and use it to enter human cells. So far, other candidates proposed as ancestors of SARS-CoV-2 found in bats, including RaTG13, haven’t been able to do this, the researchers said. The three viruses could bind to ACE2 about as well as early strains of SARS-CoV-2 found in Wuhan, they said.
The findings, which were posted to the preprint server Research Square on Sept. 17, add to the evidence that SARS-CoV-2 had a natural origin, rather than escaping from a lab.
The results show “that sequences very close to those of the early strains of SARS-CoV-2 … exist in nature,” the researchers wrote in their paper, which has yet to be peer-reviewed.
“The receptor binding domain of SARS-CoV-2 looked unusual when it was first discovered because there were so few viruses to compare it to,” Edward Holmes, an evolutionary biologist at the University of Sydney, who wasn’t involved in the research, told Bloomberg. “Now that we are sampling more from nature, we are starting to find these closely related bits of gene sequence,” Holmes said.
The authors say their findings support the hypothesis that SARS-CoV-2 resulted from a recombination of viral sequences existing in horseshoe bats.
Still, even though the newly discovered viruses are closely related to SARS-CoV-2, all three viruses lack a sequence for what is known as the “furin cleavage site,” which is seen in SARS-CoV-2 and aids the virus’s entry into cells, according to Nature News. This means that in order to better understand the origins of SARS-CoV-2 further research is needed to show how and when the furin site was introduced.
The findings are currently being considered for publication in a Nature journal, Bloomberg reported.
Originally published on Live Science.
REDOX AND BIOCHEMISTRY of CoV2-19 8/24/2021 Proceedings of the National Academy of Sciences
Perspective
Redox imbalance links COVID-19 and myalgic encephalomyelitis/chronic fatigue syndrome: Proceeding of the National Academy of Sciences
Bindu D. Paul, Marian D. Lemle, Anthony L. Komaroff, and Solomon H. Snyder
PNAS August 24, 2021 118 (34) e2024358118; https://doi.org/10.1073/pnas.2024358118
1. Edited by Maureen R. Hanson, Cornell University, Ithaca, NY, and accepted by Editorial Board Member Philippa Marrack June 25, 2021 (received for review February 28, 2021)
Abstract
Although most patients recover from acute COVID-19, some experience postacute sequelae of severe acute respiratory syndrome coronavirus 2 infection (PASC). One subgroup of PASC is a syndrome called “long COVID-19,” reminiscent of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). ME/CFS is a debilitating condition, often triggered by viral and bacterial infections, leading to years-long debilitating symptoms including profound fatigue, postexertional malaise, unrefreshing sleep, cognitive deficits, and orthostatic intolerance. Some are skeptical that either ME/CFS or long COVID-19 involves underlying biological abnormalities. However, in this review, we summarize the evidence that people with acute COVID-19 and with ME/CFS have biological abnormalities including redox imbalance, systemic inflammation and neuroinflammation, an impaired ability to generate adenosine triphosphate, and a general hypometabolic state. These phenomena have not yet been well studied in people with long COVID-19, and each of them has been reported in other diseases as well, particularly neurological diseases. We also examine the bidirectional relationship between redox imbalance, inflammation, energy metabolic deficits, and a hypometabolic state. We speculate as to what may be causing these abnormalities. Thus, understanding the molecular underpinnings of both PASC and ME/CFS may lead to the development of novel therapeutics.
Acute COVID-19, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), can be a severe and even fatal disease. Beyond the acute illness, some survivors of COVID-19, even those who are only moderately ill during the acute infection, experience postacute sequelae of severe acute respiratory syndrome coronavirus 2 infection (PASC). They report persisting, debilitating symptoms that last for months (1). In some people, these symptoms may be secondary to COVID-19–induced damage to the lung (hypoxia) and heart (reduced cardiac output) (2), skeletal muscle (3), kidneys (abnormal acid-base or fluid balance), or brain (small infarcts or hemorrhages) (4). However, some of these PASC patients, without apparent organ damage, also have persisting, debilitating symptoms (an illness called “long COVID-19”) that are similar to myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) (5).
ME/CFS is a complex, multisystem disorder leading to debilitating symptoms including profound fatigue, postexertional malaise, unrefreshing sleep, cognitive deficits, and orthostatic intolerance. The US Centers for Disease Control and Prevention and the US National Academy of Medicine estimate that 836,000 to 2.5 million people have ME/CFS in the United States alone (6). Many cases occur following what appears to be a common, infectious-like illness. However, diagnostic tests are rarely performed to document the responsible infectious agents. Postinfectious fatigue syndromes also follow in the wake of well-documented acute infections with multiple viruses, bacteria, and even parasites (5).
In this review, we speculate that the symptoms of both long COVID-19 and ME/CFS may stem from redox imbalance—which in turn, is linked to inflammation and energy metabolic defects.
Redox Imbalance Occurs in Both COVID-19 and ME/CFS
Oxidative and Nitrosative Stress.
Oxidative and nitrosative stress have been reported in both acute COVID-19 and ME/CFS. Oxidative stress involves imbalance between reactive oxygen species (ROS) and antioxidant defense mechanisms. Nitrosative stress is characterized by excess reactive nitrogen species (RNS), such as peroxynitrite (ONOO−), generated by reaction of nitric oxide (NO) with superoxide anions (O2·−). NO has important physiological functions (including vasodilation and neurotransmission). However, increased RNS with excessive NO production can be at least as damaging as ROS and also can directly attack several antioxidant enzymes, including catalase (7). Thus, oxidative stress and nitrosative stress are linked bidirectionally.
Like NO, ROS mediates physiologic cellular signaling and defense against pathogens. However, excessive ROS, when not adequately countered by antioxidants, damage cellular components including proteins, lipids, and DNA (8, 9).
Redox Imbalance in Acute COVID-19.
Multiple examples of redox dysregulation have been reported in acute COVID-19, as is typical of many viral infections (10, 11). Overall levels of serum thiols are decreased in the serum of COVID-19 patients (12). As depicted in Fig. 1A, COVID-19 induces redox imbalance, in part because SARS-CoV-2 uses the angiotensin converting enzyme 2 (ACE2) receptor to enter cells (13, 14). This leads to accumulation of O2·− as well as ROS and RNS by inducing mitochondrial dysfunction and production of proinflammatory cytokines (15).
Fig. 1.
Oxidative stress in COVID-19. (A) The ACE2 pathway. SARS-CoV-2 infects cells harboring ACE2 and the protein transmembrane protease, serine 2 (TMPRSS2); together, these molecules prime the viral spike protein S, facilitating its entry by endocytosis. ACE2 converts angiotensin II (Ang II) to angiotensin 1 to 7 (Ang 1–7). This decreases ACE2 and elevates Ang II, which acts through the angiotensin 1 receptors (AT1-Rs), causing accumulation of superoxide radicals (O2•−) leading to hypertension and inhibition of vasodilation. Ang 1–7 binds the G-protein–coupled Mas receptor MasR, mediates vasorelaxation, and decreases O2•− production. SARS-CoV-2 induces formation of reactive oxygen radicals (ROS) and RNS by eliciting mitochondrial dysfunction and production of proinflammatory cytokines. (B) The NETs. NETs are web-like structures extruded from activated neutrophils, comprising proteins assembled on a scaffold of decondensed chromatin, which target invading pathogens. The component proteins include oxidative and proinflammatory enzymes such as NADPH oxidase (Nox), neutrophil elastase, myeloperoxidase (MPO), NOS, and peptidyl arginine deiminase 4 (PAD4), which deaminates arginine to citrulline, resulting in the formation of citrullinated proteins (such as histone H3, causing its dissociation from DNA). Excessive accumulation of NETs causes inflammation and damage in COVID-19.
In a computational study, binding affinity was significantly impaired when the disulfide bonds of both ACE2 and SARS-CoV-2 spike protein, which binds ACE2, were reduced to thiol groups (16). The spike protein has 40 cysteine residues, some of which contribute to the stability of interaction with the ACE2 receptor on the host (17). Analysis of the crystal structure of the spike protein with the ACE2 receptor revealed that the Cys480–Cys488 pair of the spike protein participates directly in binding to the ACE2 (18, 19). Similarly, certain cysteine residues on the ACE2 protein of the host play key roles in the interaction between the two proteins. A disulfide bond between Cys133 and Cys141 is present at the dimer interface, which has been linked to susceptibility to COVID-19 (17). Cattle and swine have a leucine residue at position 133 and are resistant to SARS-CoV-2.
Elderly subjects are more vulnerable to severe COVID-19. A linear oxidation of the plasma cysteine/cystine redox state over the entire age span and that of reduced glutathione/oxidized glutathione (GSH/GSSG) occur after ∼45 y (20). The age-dependent decrease in thiol/disulfide ratio of extracellular fluids could modulate interaction of CoV-2 with the host cell in the airways. This redox-modulated binding is expected to affect the risk of severe infection in an age-dependent manner (21). Similarly, low levels of the antioxidant enzyme, alveolar Type II cell superoxide dismutase 3 (SOD3), in the lungs of the elderly correlate with severity of COVID-19 (12).
Redox Imbalance in ME/CFS.
In people with ME/CFS, there are multiple biomarkers of oxidative stress: reduced levels of antioxidants (22); decreased levels of α-tocopherol (23); increased levels of peroxides and superoxide that correlate with severity of symptoms (24); increased levels of isoprostanes, both at rest and after exercise (25); and reduced levels of thiobarbituric acid reactive substances and malondialdehyde levels, as well as reduced ascorbic acid and glutathione levels (26⇓⇓⇓–30). These markers of redox imbalance also correlate with severity of symptoms (24, 31, 32). Brain magnetic resonance spectroscopy (MRS) reveals elevated levels of ventricular lactic acid consistent with oxidative stress (33⇓–35).
When compared with healthy control subjects, people with ME/CFS also have multiple biomarkers of nitrosative stress: increased inducible nitric oxide synthase (NOS) with consequent increased NO, peroxynitrite, and nitrate, particularly following exercise (36, 37).
Immune responses to oxidized fatty acids (oleic, palmitic, and myristic acids) and markers of lipid peroxidation (such as malondialdehyde, acetylcholine, S-farnesyl-l-cysteine, and several NO-modified amino acids) also are significantly greater in ME/CFS patients as compared with controls (38, 39).
Metabolomic studies in plasma from ME/CFS subjects are consistent with these findings, revealing altered plasma levels of choline, carnitine, and complex lipid metabolites—consistent with oxidative stress and mitochondrial dysfunction (40⇓⇓–43).
Elevated Levels of Prooxidants in COVID-19 and ME/CFS
As summarized in Table 1, increased levels of prooxidants have been reported in both acute COVID-19 and ME/CFS.
Table 1.
Redox-related alterations shared by both COVID-19 and ME/CFS
Dysregulated Heme and Iron Homeostasis.
Increased levels of free iron in cells (44) and increased ferritin levels (45) both can cause oxidative damage (44). Iron stored in heme molecules is degraded via the bilirubin pathway, an important component of antioxidant defense (46, 47). Evidence of dysregulated heme and iron homeostasis has been reported in acute COVID-19 and ME/CFS (Table 1).
Homocysteine.
Elevated levels of the amino acid homocysteine, seen in both acute COVID-19 and ME/CFS (Table 1), cause increased oxidative stress and are a risk factor for various cardiovascular diseases and dementia. Elevated homocysteine levels may indicate suboptimal activities of reverse transsulfuration enzymes or dysregulation of their cofactors and/or regulators (44, 48, 49).
Neutrophil Extracellular Traps.
Neutrophils attack invading pathogens by forming neutrophil extracellular traps (NETs) and generating both ROS and RNS (50⇓⇓–53) (Fig. 1B). Abnormalities of neutrophil biology have been reported in both acute COVID-19 and ME/CFS (Table 1).
Abnormal Metabolism Involving the Gaseous Signaling Molecules NO and Hydrogen Sulfide.
The major gaseous signaling molecules include NO, carbon monoxide, and hydrogen sulfide (H2S). They play key roles in the regulation of blood pressure, inflammation, and neurotransmission (54, 55). Under physiological conditions, both NO and H2S have anti-inflammatory effects (48, 56). However, both deficient and excess production of these gaseous signaling molecules can create brain pathology, immune dysfunction, and redox imbalance (56, 57).
NO.
NOS2 is significantly up-regulated in patients with severe and critical COVID-19 (58). There is evidence of nitrosative stress and disordered NO metabolism in people with ME/CFS (38). Levels of NO are higher in ME/CFS patients, which can accelerate nitrosative stress (27). Citrulline, a product of arginine metabolism by NOS, also is increased in ME/CFS (59).
H2S.
Normal H2S metabolism protects against inflammation and redox imbalance (48, 60, 61). One of the modes by which H2S functions is by a posttranslational modification termed persulfidation or sulfhydration (48, 62), which prevents irreversible oxidation of proteins (63, 64). H2S metabolism is disrupted in Alzheimer’s disease, Parkinson’s disease, and Huntington’s disease and also, during aging (63, 65⇓–67). H2S inhibits tau-phosphorylation, which may explain, in part, its role in protecting against Alzheimer’s disease (68, 69).
Dysregulated H2S metabolism has been reported in acute COVID-19. Survivors reportedly exhibit higher serum levels of H2S and higher numbers of circulating lymphocytes (70); H2S stimulates T cell proliferation (71). Indeed, exogenous H2S therapy may be beneficial in mild to moderate COVID-19 disease (72).
Dysregulation of H2S may play a role in ME/CFS since the gas can affect adenosine triphosphate (ATP) production from oxidative phosphorylation (73). H2S can induce a torpor-like state in mice (74). However, studies of H2S regulation in people with ME/CFS have not yet been reported.
Altered Levels of Tryptophan Metabolites.
Tryptophan serves as a precursor for nicotinamide adenine dinucleotide (NAD+) biosynthesis, making it important in redox balance. Abnormalities in the pathways by which tryptophan is transformed into serotonin or kynurenine pathway metabolites (75) have been reported in both acute COVID-19 and ME/CFS (Table 1), and can cause both oxidative stress and excitotoxicity (27).
Reduced Levels of Small Molecule Antioxidants in Acute COVID-19 and ME/CFS
As summarized in Table 1, decreased levels of antioxidants have been reported in both acute COVID-19 and ME/CFS. Foremost among these small molecules is glutathione, which helps modulate immune activation (76, 77). Glutathione also enhances vitamin D metabolism (78) and vitamin D, in turn, reciprocally increases glutathione and decreases oxidative stress and levels of inflammatory cytokines and chemokines (79). The antioxidant small molecules also include cysteine, a semiessential amino acid synthesized endogenously via the reverse transsulfuration pathway (80) (SI Appendix, Fig. S1); selenium, an essential micronutrient incorporated into various selenoproteins which have antioxidant roles; vitamin C/ascorbate, which exerts antiviral and immunomodulatory effects (81) and vitamin E compounds, which have free radical scavenging and antiinflammatory activities (82).
Discovery of re-purposed drugs that slow SARS-CoV-2 replication in human cells
• Adam Pickard , et al 9/9/21
• https://doi.org/10.1371/journal.ppat.1009840
COVID-19 vaccines based on the Spike protein of SARS-CoV-2 have been developed that appear to be largely successful in stopping infection. However, therapeutics that can help manage the disease are still required until immunity has been achieved globally. The identification of repurposed drugs that stop SARS-CoV-2 replication could have enormous utility in stemming the disease. Here, using a nano-luciferase tagged version of the virus (SARS-CoV-2-ΔOrf7a-NLuc) to quantitate viral load, we evaluated a range of human cell types for their ability to be infected and support replication of the virus, and performed a screen of 1971 FDA-approved drugs. Hepatocytes, kidney glomerulus, and proximal tubule cells were particularly effective in supporting SARS-CoV-2 replication, which is in-line with reported proteinuria and liver damage in patients with COVID-19. Using the nano-luciferase as a measure of virus replication we identified 35 drugs that reduced replication in Vero cells and human hepatocytes when treated prior to SARS-CoV-2 infection and found amodiaquine, atovaquone, bedaquiline, ebastine, LY2835219, manidipine, panobinostat, and vitamin D3 to be effective in slowing SARS-CoV-2 replication in human cells when used to treat infected cells. In conclusion, our study has identified strong candidates for drug repurposing, which could prove powerful additions to the treatment of COVID.
Impaired Energy Metabolism in COVID-19 and ME/CFS
Impaired Energy Metabolism in Acute COVID-19.
Mitochondrial dysfunction has long been associated with fatigue (83), causes elevated oxidative stress, and could contribute to the symptoms of fatigue found in both acute COVID-19 and ME/CFS (84).
Mitochondrial dysfunction also has been linked to the pathogenesis of COVID-19. SARS-CoV-2 hijacks mitochondrial function and alters host metabolic pathways and immune response to facilitate pathogenesis. For instance, mitochondrial dysfunction triggered by SARS-CoV-2 causes accumulation of mitochondrial DNA (mtDNA) in the cytosol, leading to mtDNA-induced inflammasome activation and suppression of innate and adaptive immunity (85). The virus interferes with the RIG1-MAVS pathway to decrease type I interferon (IFN) production (86). SARS-CoV-2 infection of white blood cells leads to elevated glycolysis, increased mitochondrial reactive oxygen species (mtROS) production, and dysregulated mitochondrial bioenergetics (87, 88). In this study, the role of redox imbalance secondary to mitochondrial dysfunction in SARS-CoV-2 pathology was apparent; two potent antioxidants reduced viral load and proinflammatory cytokines. Elevated levels of lactate dehydrogenase (LDH) were also reported in COVID-19 (89, 90), which could reflect mitochondrial dysfunction.
Impaired Energy Metabolism in ME/CFS.
Metabolomic studies have reported evidence of impaired ATP production from oxygen, glucose, fatty acids, and amino acids in multiple cell types (41, 42, 91⇓⇓–94). Not just oxidative phosphorylation but also glycolysis—and possibly, the citric acid and urea cycles—are incriminated. In people with ME/CFS, there also is a more general hypometabolic state as previously proposed (73), characterized by depressed levels of most metabolites, as occurs in hibernating animals (41). Later, we speculate as to the cause of this hypometabolic state.
Both structural and functional mitochondrial abnormalities have been found in ME/CFS. Branching and fusion of mitochondrial cristae are observed in muscle biopsies of some patients (84). Although some studies have reported deletions of mtDNA genes, the most extensive controlled study using contemporary technology did not find mtDNA variants that correlated with susceptibility to ME/CFS—although it did find a correlation between specific haplogroups and mtDNA single-nucleotide polymorphisms and specific symptoms (95). Other reports have identified a deficit in Complex V (ATP synthase) activity of the electron transport chain (ETC) in lymphocytes, with a compensatory up-regulation of respiratory capacity (96, 97), and a decrease in mitochondrial membrane potential in CD8+ T cells (98). Serum from ME/CFS patients reportedly contains a factor that induces mitochondrial fragmentation (99).
Clinical studies also indicate mitochondrial dysfunction. People with ME/CFS have significantly higher blood lactate levels after exercise as compared with controls, indicative of reduced oxidative phosphorylation and a switch to anaerobic glycolysis (100⇓–102). Elevated lactate levels also have been reported in the ventricles of ME/CFS patient brains (33⇓–35, 103). As in COVID-19, elevated serum LDH levels also are seen (104). LDH is a critically important enzyme in energy metabolism, catalyzing the bidirectional conversion of lactate to pyruvate and NAD+ to reduced NAD+ (NADH). Thus, an elevated level in the blood of subjects in a resting state could indicate a system struggling to generate energy. Alternatively, the elevated blood levels of LDH could indicate tissue destruction, such as occurs in malignancies or red blood cell hemolysis.
Finally, people with ME/CFS reportedly have significantly decreased levels of plasma coenzyme Q10 (CoQ10), whose levels correlate inversely with the degree of fatigue, impaired concentration and memory, and symptoms of autonomic dysfunction (105, 106). CoQ10/ubiquinone, a component of the ETC, can have both prooxidant and antioxidant effects, as well as anti-inflammatory effects (107).
The causes of mitochondrial dysfunction in people with ME/CFS remain speculative. Viral infection surely can cause impairment of mitochondrial structure (99) and function (97, 108), and impairment of mitochondrial function, in turn, encourages viral replication and T cell exhaustion (108). Immune activation, with the generation of proinflammatory cytokines, also can cause mitochondrial fragmentation, hyperpolarization of the mitochondrial membrane, and the generation of ROS (109).
Connecting Redox Imbalance to Inflammation in COVID-19 and ME/CFS
Connections between Inflammation and Redox Imbalance.
Systemic inflammation and neuroinflammation are seen in both acute COVID-19 and ME/CFS. Inflammation, in turn, is bidirectionally linked to redox imbalance (110); inflammation generates ROS and RNS, and redox imbalance causes cellular damage that evokes an inflammatory response, leading to vicious cycles (111, 112).
Glutathione plays a particularly important role in enabling and modulating the immune response (76). It is vital for proliferation of T lymphocytes; T cell activation, in turn, generates glutathione, which counters ROS levels and mediates a metabolic shift toward aerobic glycolysis and glutaminolysis (113)
Other connections between inflammation and redox imbalance exist as well. Higher interleukin-2 (IL-2) levels stimulate NO production (114, 115), and IL-6 and tumor necrosis factor-α (TNF-α) stimulate cells to produce O2•− (116, 117). Mitochondrial dysfunction also leads to increased proinflammatory responses and increased ROS levels.
Inflammation in COVID-19 and Redox Imbalance.
The cytokine storm seen in severe cases of COVID-19 has been well characterized (118, 119). The nucleotide-binding oligomerization domain-like receptor containing pyrin domain 3 (NLRP3) inflammasome plays a key role in the effects of the cytokine storm; NLRP3, in turn, is activated by oxidative stress (118). Increased levels of the proinflammatory markers, C-reactive protein and IL-6, were associated with the disease (120).
Immunologic factors that correlated with more severe disease and higher mortality include neutrophilia, lymphocytopenia, low CD4+ T cells, decreased C3, very low human leukocyte antigen D–related expression, and low numbers of CD19 lymphocytes and natural killer (NK) cells (121). In addition to these changes, COVID-19 is associated with inadequate Type I and Type III IFN responses and elevated chemokine expression (122⇓–124). Finally, T cell exhaustion is commonly seen in COVID-19 and could be explained by low levels of glutathione (125).
Inflammation in ME/CFS and Redox Imbalance.
The fatigue and cognitive deficits in people with ME/CFS are associated with neuroinflammation; positron emission tomography imaging reveals increased activation of microglia, astrocytes, and elevated levels of cytokines in the brain and spinal cord (126, 127). Increased levels of various cytokines can trigger many of the symptoms of ME/CFS (and post–COVID-19 syndrome), as became clear in the 1980s when various cytokines were synthesized and used as treatments. For example, when IFN I-α is given as antiviral therapy, it induces persistent fatigue in patients with chronic hepatitis C virus infection (128).
In ME/CFS, the systemic circulation often contains elevated levels of inflammatory biomarkers—proinflammatory cytokines like IL-1 and TNF-α (106). Also, circulating lymphocytes often produce increased transcripts of these cytokines (129). Both COVID-19 and ME/CFS are associated with activation of the protein kinase R and 2- to 5A synthetase antiviral IFN response pathways (130⇓–132).
People with ME/CFS often have increased numbers of CD8+ T cells bearing activation antigens (133), although persistent T cell activation then may lead to exhaustion (134). Another characteristic finding in people with ME/CFS is impaired NK cell function (135). Redox imbalance leading to increased levels of l-kynurenine and lactate can impair NK cell function (136).
Connecting Redox Imbalance, Inflammation, and Energy Metabolism
Viral infection triggers increased mitochondrial function and sometimes, mitochondrial damage. In either case, increased ROS are produced. ROS, in turn, damage mtDNA and proteins, including those comprising the ETC, causing a decrease in ATP production (137, 138). In COVID-19, damage to endothelial cells results in chronic inflammation, thrombosis, atherosclerosis, and lung injury. Endothelial mitochondria modulate these inflammatory pathways via redox signaling, involving mtROS. However, continued elevation of mtROS leads to senescence, promoting inflammation and chronic endothelial dysfunction, culminating in vicious cycles that involve ROS, inflammation, and mitochondrial dysfunction (139, 140).
A mode by which mitochondrial dysfunction causes inflammation is by activating the NLRP3 inflammasome in immune cells. The NLRP3 inflammasome participates in the processing and release of inflammatory cytokines, such as IL-1β and IL-18 (141). Damaged ROS-generating mitochondria can elicit persistent inflammation via NLRP3 inflammasome-dependent inflammatory pathways (142). Additionally, damaged mitochondria mount inflammatory responses by releasing mtDNA into the cytosol; the DNA and its purinergic components function as a damage-activated molecular pattern to trigger the innate immune system (143, 144). Finally, mitochondria also modulate both adaptive and innate immune responses (143, 145, 146). In COVID-19, a dysregulation of the innate immune system has been observed, causing aberrant engagement of antiviral signaling cascades, which facilitates evasion of the host immune system and which is linked to mitochondrial function as described earlier (85, 86, 147). Thus, infection and inflammation are intimately linked to energy metabolism and redox imbalance (Fig. 2).
Fig. 2.
The interactions between redox imbalance, mitochondrial dysfunction, chronic inflammation, and related symptoms. As explained in the text, redox imbalance, mitochondrial dysfunction, and inflammation are bidirectionally related to each other and may cause some of the symptoms of both long COVID-19 and ME/CFS. The bidirectional connections mean that an initial abnormality in one component can trigger abnormalities in other components and can precipitate a persistent, self-reinforcing pathological process.
Hypometabolic State, the Cell Danger Response, and Integrated Stress Response
What might cause the hypometabolic state reported in ME/CFS? Dysregulated H2S production can induce a hypometabolic, torpor-like state in mice (73, 74). In addition, any of several stressors, including viral infection and oxidative stress, can trigger evolutionarily conserved protective responses that operate at the level of both the cell (the cell danger response) (148) and the whole organism (the integrated stress response, hibernation, and the state of dauer in Caenorhabditis elegans) (121, 149, 150). These protective responses generally are reversible when the stressor no longer is present. Since dysregulated H2S production can induce a hypometabolic, torpor-like state in mice (74), H2S production may be one potential switch (73). Since protein translation consumes considerable ATP, a reduction in protein translation thereby makes ATP available for repair of injury and preservation of vital functions.
The hypometabolic state seen in ME/CFS (and that may be seen in PASC) could be secondary to a persisting stressor (such as redox imbalance or viral infection), or it could result from a defect in the “switch” that turns off the protective state. Abnormalities in purinergic signaling secondary to mitochondrial damage (151, 152) and mitochondrial dysfunction of any cause (153) are often associated with cellular and organism stress responses, and each has been linked to a wide variety of neurological disorders.
Potential Redox-Based Therapeutics
Several therapies targeting redox imbalance already have been utilized or proposed for the treatment of disease. NO inhibits the replication of SARS-CoV-2 in vitro (154) and improves oxygenation in people with COVID-19 when administered by inhalation (155). Small studies of ubiquinol (156) and of a combination of NADH and CoQ10 (157) have reported clinical benefit. Many other potential treatments targeting redox imbalance also deserve consideration: for example, glutathione (and glutathione donors), N-acetyl cysteine, cysteamine, sulforaphane, ubiquinol, nicotinamide, melatonin, selenium, vitamin C, vitamin D, vitamin E, melatonin plus pentoxyfylline, disulfiram, ebselen, and corticosteroids. In two cases of acute COVID-19, glutathione administered therapeutically counteracted dyspnea associated with COVID-19 pneumonia and reduced pulmonary inflammation (158).
In rodents, administering H2S donors reduced inflammation and oxidative stress and attenuated ventilator-induced lung injury as well as injury induced by pneumonia (159, 160). In addition, the H2S donor, GYY4137, suppressed replication of enveloped RNA viruses like SARS-CoV-2 (161⇓–163). Additionally, the H2S donor, sodium hydrosulfide, inhibits platelet activation, NET formation, DNA, and ROS levels while decreasing SOD in the hyperhomocysteinemia (HHcy) group (164). Thus, treatment of acute COVID-19 with H2S donors may be efficacious (165).
A screen for inhibitors of the main protease of SARS-CoV-2 identified ebselen, an organoselenium compound, as a potential inhibitor for the protease, Mpro or NSP5, and a therapeutic agent for COVID-19 (166, 167).
In general, however, oral therapies directed at restoring redox balance have not produced dramatic improvements in conditions associated with redox imbalance (168). No single antioxidant can scavenge or neutralize the wide variety of ROS and RNS singlehandedly. Hence, up-regulating pathways that counteract multiple abnormalities and bolster antioxidant defense and balance may be more beneficial. The timing of intervention may also be critical.
Concluding Remarks
People with acute COVID-19 and people with ME/CFS share redox imbalance, systemic inflammation and neuroinflammation, impaired production of ATP and other abnormalities in common (Fig. 2), abnormalities that have bidirectional connections (169).
The syndrome of long COVID-19 that can develop in some COVID-19 survivors (people called “long haulers”) is very similar to ME/CFS, so it may well be that the group of abnormalities seen in acute COVID-19 and in ME/CFS also will be seen in long COVID-19. Presumably, redox abnormalities in COVID-19 are secondary to the infection with SARS-CoV-2. The same may be true among those ME/CFS patients whose illness began with an “infectious-like” illness.
Clearly, COVID-19–induced permanent damage to the lungs (chronic hypoxia), heart (congestive failure), and kidneys (fluid and acid-base abnormalities) could cause some of the persisting symptoms seen in long COVID-19. In both long COVID-19 and ME/CFS other symptoms (e.g., fatigue, brain fog) may be generated by neuroinflammation, reduced cerebral perfusion due to autonomic dysfunction, and autoantibodies directed at neural targets, as summarized elsewhere (170).
As many as 2.5 million people suffer from ME/CFS in the United States (6). The COVID-19 pandemic may generate a similar number of cases of long COVID-19 in the coming 1 to 2 y (5). It therefore is imperative that increased research be focused on both long COVID-19 and ME/CFS. Fortunately, the United States and several other countries have committed substantial funding to study chronic illnesses following COVID-19, one of which is long COVID-19. Two registries and associated biobanks of people with long COVID-19 and/or ME/CFS are available to aid research.* We suggest that the study of the connections between redox imbalance, inflammation, and energy metabolism in long COVID-19 and in ME/CFS may lead to improvements in both new diagnostics and therapies.
Association of Self-reported COVID-19 Infection and SARS-CoV-2 Serology Test Results With Persistent Physical Symptoms Among French Adults During the COVID-19 Pandemic
Joane Matta, PhD1; et al 11/8/21
Question Are the belief in having had COVID-19 infection and actually having had the infection as verified by SARS-CoV-2 serology testing associated with persistent physical symptoms during the COVID-19 pandemic?
Findings In this cross-sectional analysis of 26 823 adults from the population-based French CONSTANCES cohort during the COVID-19 pandemic, self-reported COVID-19 infection was associated with most persistent physical symptoms, whereas laboratory-confirmed COVID-19 infection was associated only with anosmia. Those associations were independent from self-rated health or depressive symptoms.
Meaning Findings suggest that persistent physical symptoms after COVID-19 infection should not be automatically ascribed to SARS-CoV-2; a complete medical evaluation may be needed to prevent erroneously attributing symptoms to the virus.
Importance After an infection by SARS-CoV-2, many patients present with persistent physical symptoms that may impair their quality of life. Beliefs regarding the causes of these symptoms may influence their perception and promote maladaptive health behaviors.
Objective To examine the associations of self-reported COVID-19 infection and SARS-CoV-2 serology test results with persistent physical symptoms (eg, fatigue, breathlessness, or impaired attention) in the general population during the COVID-19 pandemic.
Design, Setting, and Participants Participants in this cross-sectional analysis were 26 823 individuals from the French population-based CONSTANCES cohort, included between 2012 and 2019, who took part in the nested SAPRIS and SAPRIS-SERO surveys. Between May and November 2020, an enzyme-linked immunosorbent assay was used to detect anti–SARS-CoV-2 antibodies. Between December 2020 and January 2021, the participants reported whether they believed they had experienced COVID-19 infection and had physical symptoms during the previous 4 weeks that had persisted for at least 8 weeks. Participants who reported having an initial COVID-19 infection only after completing the serology test were excluded.
Main Outcomes and Measures Logistic regressions for each persistent symptom as the outcome were computed in models including both self-reported COVID-19 infection and serology test results and adjusting for age, sex, income, and educational level.
Results Of 35 852 volunteers invited to participate in the study, 26 823 (74.8%) with complete data were included in the present study (mean [SD] age, 49.4 [12.9] years; 13 731 women [51.2%]). Self-reported infection was positively associated with persistent physical symptoms, with odds ratios ranging from 1.39 (95% CI, 1.03-1.86) to 16.37 (95% CI, 10.21-26.24) except for hearing impairment (odds ratio, 1.45; 95% CI, 0.82-2.55) and sleep problems (odds ratio, 1.14; 95% CI, 0.89-1.46). A serology test result positive for SARS-COV-2 was positively associated only with persistent anosmia (odds ratio, 2.72; 95% CI, 1.66-4.46), even when restricting the analyses to participants who attributed their symptoms to COVID-19 infection. Further adjusting for self-rated health or depressive symptoms yielded similar results. There was no significant interaction between belief and serology test results.
Conclusions and Relevance The findings of this cross-sectional analysis of a large, population-based French cohort suggest that persistent physical symptoms after COVID-19 infection may be associated more with the belief in having been infected with SARS-CoV-2 than with having laboratory-confirmed COVID-19 infection. Further research in this area should consider underlying mechanisms that may not be specific to the SARS-CoV-2 virus. A medical evaluation of these patients may be needed to prevent symptoms due to another disease being erroneously attributed to “long COVID.”
Introduction
After infection by SARS-CoV-2, both hospitalized and nonhospitalized patients have an increased risk of various persistent physical symptoms that may impair their quality of life, such as fatigue, breathlessness, or impaired attention.1-3 Although the term “long COVID” has been coined to describe these symptoms4 and putative mechanisms have been proposed,3,5,6 the symptoms may not emanate from SARS-CoV-2 infection per se but instead may be ascribed to SARS-CoV-2 despite having other causes. In this study, we examined the association of self-reported COVID-19 infection and of serology test results with persistent physical symptoms. We hypothesized that the belief in having been infected with SARS-CoV-2 would be associated with persistent symptoms while controlling for actual infection.
Methods
The French CONSTANCES population-based cohort study7 received ethical approval and included approximately 200 000 volunteers who were aged 18 to 69 years between 2012 and 2019 and who consented to be followed up through annual questionnaires and linked administrative databases.8 A total of 35 852 volunteers responding to annual questionnaires through the internet were invited to take part in the nested Santé, Pratiques, Relations et Inégalités Sociales en Population Générale Pendant la Crise COVID-19 (SAPRIS) and SAPRIS-Sérologie (SERO) surveys.9,10 Ethical approval and written or electronic informed consent were obtained from each participant before enrollment in the original cohort. The SAPRIS survey was approved by the French Institute of Health and Medical Research ethics committee, and the SAPRIS-SERO study was approved by the Sud-Mediterranée III ethics committee. Electronic informed consent was obtained from all participants for dried-blood spot testing. No one received compensation or was offered any incentive for participating in this study. The present study is a cross-sectional analysis of data from the SAPRIS and SAPRIS-SERO surveys nested in the French CONSTANCES cohort.
Serologic Testing
Between May and November 2020, self-sampling dried-blood spot kits were mailed to each participant. Each kit included material (a dried-blood spot card, lancets, and a pad), printed instructions, and an addressed, stamped, and padded envelope to be returned with the card to a centralized biobank (CEPH Biobank). Received blood spots were visually assessed, registered, punched, and stored in tubes (0.5 mL, FluidX 96-Format 2D code; Brooks Life Sciences) at −30 °C. Eluates were processed with an enzyme-linked immunosorbent assay (Euroimmun) to detect anti–SARS-CoV-2 antibodies (IgG) directed against the S1 domain of the virus spike protein. A test was considered positive for SARS-CoV-2 when the results indicated an optical density ratio of 1.1 or greater (sensitivity, 87%; specificity, 97.5%).11 The participants received their serology test results by mail or email.
Self-reported COVID-19 Infection
Between December 2020 and January 2021, the participants answered this question from the fourth SAPRIS questionnaire: “Since March, do you think you have been infected by the coronavirus (whether or not confirmed by a physician or a test)?” Participants answered “Yes,” “No,” or “I don’t know.” At the time they answered this question, the participants were aware of their serology test results (eFigure in Supplement 1). A total of 2788 participants (7.8%) who answered “I don’t know” were excluded.
The participants who answered “Yes” additionally answered this question: “When did you get the coronavirus? Between March and June; In July or August; Between September and now.” Participants who indicated having been initially infected after serologic testing (n = 1312 [3.6%]) were excluded. The participants who answered “Yes” also answered this question: “Has this been confirmed? Yes, by virological or PCR test (based on nose swab; results provided after at least 24 hours); Yes, by antigenic test performed (based on nose swab; results provided within 1 hour); Yes, by serological test (based on a blood test; results provided after at least 24 hours); Yes, by rapid diagnostic test (based on blood test; results provided within 1 hour); Yes, by saliva test; Yes, by chest CT scan; Yes, by a physician (without testing); No, but I think I had it; I don’t know.”
Persistent Physical Symptoms
In the same questionnaire, symptoms were measured by the following question: “Since March 2020, have you had any of the following symptoms that you did not usually have before?” On the basis of the literature,1-3 the following symptoms were explored: sleep problems, joint pain, back pain, muscular pain, sore muscles, fatigue, poor attention or concentration, skin problems, sensory symptoms (pins and needles, tingling or burning sensation), hearing impairment, constipation, stomach pain, headache, breathing difficulties, palpitations, dizziness, chest pain, cough, diarrhea, anosmia, and other symptoms.
Two additional questions were asked for each symptom: “Has this symptom been present in the past 4 weeks?” Participants answered “Yes, but not present anymore,” “Yes, and still present,” or “No”; “How much time did this symptom last? Or how long has it been since you have had this symptom (if it is still present)?” with possible responses ranging from “Less than a week” to “More than 8 weeks.” To avoid considering symptoms that were no longer present or only transient and to limit recall bias, only participants who responded “Yes” and “More than 8 weeks” to these 2 questions were considered as having persistent symptoms. Because we aimed to compare participants who self-reported having had COVID-19 infection with those who did not, we did not distinguish between persistent symptoms that were similar to those experienced at the time of the initial episode and potentially new symptoms.
Participants who declared having any of the listed persistent symptoms also answered the following question: “Do you attribute the current symptoms to COVID-19?” and participants answered “Yes, all”; “Yes, only a few”; “No”; or “I don’t know.” Participants who answered “Yes, all” or “Yes, only a few” were considered to attribute their symptoms to COVID-19 infection.
Covariates
Age, sex, educational level, income, and self-rated health in 2019 were obtained from the inclusion questionnaire and the 2019 CONSTANCES questionnaire. Depressive symptoms during the pandemic were measured as part of the SAPRIS survey by using the Center for Epidemiologic Studies Depression Scale.12
Statistical Analysis
The crude prevalence of persistent physical symptoms was first calculated for 4 groups of participants according to both belief (ie, self-reported COVID-19 infection) and serology test results: belief negative and serology negative; belief positive and serology negative; belief negative and serology positive; and belief positive and serology positive. We used χ2 tests to search for between-group differences. To specifically test our hypothesis, we used separate logistic regressions for each persistent symptom as the outcome computed in models including either belief (model 1), serology test result (model 2), or both (model 3), adjusting for age, sex, income, and educational level. Additional models searched for belief by serology test result interactions. In sensitivity analyses, the models were further adjusted for self-rated health or depressive symptoms. Exploratory analyses were restricted to participants attributing their persistent symptoms to COVID-19 infection. A 2-sided value of P < .05 was considered statistically significant. All analyses were conducted using SAS, version 9.4 (SAS Institute Inc).
Results
Of 35 852 volunteers invited to participate in this cross-sectional analysis, a cohort of 26 823 (74.8%) with complete data were included (mean [SD] age, 49.4 [12.9] years; 13 731 women [51.2%]; and 13 092 men [48.8%]) (Table 1). The crude prevalence rates of persistent symptoms by belief and by serology test result categories are given in Table 2. Compared with participants in the CONSTANCES cohort, the participants in the present study were more likely to be older, men, more educated, have higher levels of income, and have better self-reported health (eTable 1 in Supplement 1). The prevalence of persistent physical symptoms ranged from 0.5% (146 participants with anosmia) to 10.2% (2729 participants with sleep problems). A total of 1091 participants had a serology test result positive for SARS-CoV-2, including 453 participants (41.5%) who subsequently reported having had COVID-19 infection before the serology test. A total of 914 participants reported having had COVID-19 infection before the serology test, including 453 (49.6%) with a serology test result positive for SARS-CoV-2 (Table 2). Differences in covariates according to the serology test results, the belief in having had COVID-19 infection, and both are reported in eTables 2, 3, and 4 in Supplement 1. Whether or not the diagnosis was confirmed by a laboratory test or by a physician among the participants with a positive belief is reported in eTable 5 in Supplement 1.
Before adjustment, the belief in having had COVID-19 infection was associated with 15 of 18 categories of persistent symptoms (Table 3, model 1), whereas a positive serology test result was associated with 10 categories of persistent symptoms (Table 3, model 2). After mutual adjustment, positive belief was significantly associated with higher odds of having all persistent symptoms, with odds ratios (ORs) ranging from 1.39 (95% CI, 1.03-1.86) to 16.37 (95% CI, 10.21-26.24) except for hearing impairment (OR, 1.45; 95% CI, 0.82-2.55) and sleep problems (OR, 1.14; 95% CI, 0.89-1.46) (Table 3, model 3). By contrast, a positive serology test result remained positively associated only with anosmia (OR, 2.72; 95% CI, 1.66-4.46) and was negatively associated with skin problems (OR, 0.49; 95% CI, 0.29-0.85) (Table 3, model 3). There was no significant interaction between belief and serology. Adjusting for self-rated health or depressive symptoms yielded similar results except for joint pain (OR, 1.31; 95% CI, 0.97-1.77) and back pain (OR, 1.29; 95% CI, 0.97-1.72), which were no longer associated with belief when adjusting for depressive symptoms (eTable 6 in Supplement 1).
Restricting the analyses to participants with a positive belief and attributing their persistent symptoms to COVID-19 showed a positive serology test result to be associated only with anosmia (OR, 2.97; 95% CI, 1.58-5.57) (eTable 7 in Supplement 1). Similarly, confirmation of the diagnosis by a laboratory test or by a physician (vs the response, “No, but I think I had it,” and excluding participants who answered “I don’t know”) was also associated only with anosmia (OR, 4.29; 95% CI, 1.92-9.58) (eTable 7 in Supplement 1).
Discussion
This cross-sectional analysis of data from a population-based cohort found that persistent physical symptoms 10 to 12 months after the COVID-19 pandemic first wave were associated more with the belief in having experienced COVID-19 infection than with having laboratory-confirmed SARS-CoV-2 infection.
In previous studies, the association between persistent symptoms and SARS-CoV-2 serology test results may be explained by the belief in having experienced COVID-19 infection.13 Furthermore, most previous studies assessing “long COVID” included only patients who had COVID-19 infection, thus lacking a control group of patients who did not have the infection.3,14 Indeed, our results showed that the persistent physical symptoms observed after COVID-19 infection were quite frequent in the general population. Because our study also included participants who reported not having had COVID-19 infection with either positive or negative serology test results, we were able to compare the prevalence of persistent physical symptoms according to these 2 variables. We were also able to perform analyses restricted to participants attributing their persistent symptoms to COVID-19 infection. Although our study did not assess long COVID per se because we also included participants without COVID-19 infection, these specific analyses may be more representative of the long COVID clinical issue in real-life settings15 than the picture provided by cohorts of patients with a laboratory-confirmed or physician-documented COVID-19 infection.
Although the participants were aware of the serology results when they reported having had COVID-19 infection or not, less than half of those with a positive serology test reported having experienced the disease. Conversely, among those who reported having had the disease, approximately half had a negative serology test result, consistent with some findings in clinical settings.15 These results, which allowed for disentangling the correlates of the serology test results from those of the belief in having had COVID-19 infection, were not unexpected. First, patients with a positive serology test result but no or only mild symptoms of COVID-19 infection may not believe that they had the disease. Because persistent symptoms may be more frequent among patients who experienced a higher number of acute COVID-19 symptoms,16 the severity of the initial episode may partially confound the association between the belief in having experienced COVID-19 infection and persistent symptoms among participants with positive serology test results. However, this belief was associated with persistent symptoms to a similar extent among participants with negative serology test results as shown by the lack of any interaction between belief and serology. Even if this belief could be explained by the experience of a COVID-19 infection–like episode among some of these participants, these results support the idea that persistent physical symptoms attributed to COVID-19 infection may not be specific to SARS-CoV-2. Second, patients who believe that they have had COVID-19 infection may reject a negative serology test result for several reasons, including perceptions about the frequency of false-negative tests and data suggesting that a weak anti–SARS-CoV-2 antibody response could be a risk factor of long COVID.17 Indeed, since the first definitions of long COVID, it has been proposed that the associated antibodies profile is “uncharacterized.”18 Among participants in the present study who believed that they had experienced COVID-19 infection, anosmia was the only symptom associated with the confirmation of the diagnosis by a laboratory test or a physician. In other words, those who responded, “No, but I think I had it” were 4 times less likely to have anosmia, with no differences regarding all other symptoms, further suggesting that these other symptoms were not specific to actual infection by SARS-CoV-2.
Two main mechanisms may account for our findings. First, having persistent physical symptoms may have led to the belief in having had COVID-19, especially in the context of a growing concern regarding long COVID. Although adjusting for self-rated health before the pandemic did not affect our results, another disease may underlie symptoms attributed to COVID-19 infection. Second, the belief in having had COVID-19 infection may have increased the likelihood of symptoms, either directly by affecting perception19,20 or indirectly by prompting maladaptive health behaviors, such as physical activity reduction or dietary exclusion. These mechanisms are thought to contribute to the long-described persistence of physical symptoms after acute infections.21
Strengths and Limitations
In addition to a large, population-based sample, the strengths of our study included the joint examination of self-reported COVID-19 infection and serology testing results while controlling for several covariates, including self-rated health—a robust indicator of physical health—and depressive symptoms.
This study had limitations. First, selection biases limit the representativeness of our sample. Second, our study may not have investigated all of the symptoms that patients with long COVID are reporting. However, the symptoms we studied were among those that are frequently explored in studies investigating long COVID3 and reported by patients with long COVID.22 Third, we analyzed persistent symptoms separately; different outcomes may be tested by clustering symptoms. In addition, because our study also included participants who did not report having had COVID-19 infection, we did not distinguish between symptoms that were experienced at the time of the initial episode of COVID-19 infection and new symptoms that occurred afterward. Fourth, we cannot exclude the possibility of misclassification regarding serology test results. On the basis of the present results, we estimate the prevalence of previous SARS-CoV-2 infection to be about 4%, and with a sensitivity of 87%, we would expect 139 participants to have false-negative results, which is less than 1% of those with negative serology test results. False-negative results were thus unlikely to have much influence on the associations between persistent symptoms and serology. In addition, the lack of any interaction between belief and serology test results suggests that persistent symptoms were associated with belief to a similar extent in participants with positive and negative serology test results. This finding makes our results unlikely to be explained solely by false-negative results. Furthermore, serology test results were associated only with persistent anosmia, a hallmark of COVID-19 infection, strengthening our confidence in the serology test results. This result held true even when restricting our analyses to participants attributing their symptoms to COVID-19 infection. Fifth, participants were aware of their serology test results when they reported having had COVID-19 infection or not. This factor may have reduced our ability to disentangle the associations of the 2 measures with persistent physical symptoms.
Conclusions
The results of this cross-sectional analysis of a large, population-based French cohort suggest that physical symptoms persisting 10 to 12 months after the COVID-19 pandemic first wave may be associated more with the belief in having experienced COVID-19 infection than with actually being infected with the SARS-CoV-2 virus. Although our study cannot determine the direction of the association between belief and symptoms, our results suggest that further research regarding persistent physical symptoms after COVID-19 infection should also consider mechanisms that may not be specific to the SARS-CoV-2 virus. From a clinical perspective, patients in this situation should be offered a medical evaluation to prevent their symptoms being erroneously attributed to COVID-19 infection and to identify cognitive and behavioral mechanisms that may be targeted to relieve the symptoms.23
Accepted for Publication: September 17, 2021.
Published Online: November 8, 2021. doi:10.1001/jamainternmed.2021.6454
Corresponding Author: Cédric Lemogne, MD, PhD, Service de Psychiatrie de l’adulte, Hôpital Hôtel-Dieu, 1 place du Parvis Notre-Dame, 75004 Paris, France ([email protected]).
The Science Suggests a Wuhan Lab Leak
The Covid-19 pathogen has a genetic footprint that has never been observed in a natural coronavirus.
By Steven Quay and Richard Muller
June 6, 2021
The possibility that the pandemic began with an escape from the Wuhan Institute of Virology is attracting fresh attention. President Biden has asked the national intelligence community to redouble efforts to investigate.
Much of the public discussion has focused on circumstantial evidence: mysterious illnesses in late 2019; the lab’s work intentionally supercharging viruses to increase lethality (known as “gain of function” research). The Chinese Communist Party has been reluctant to release relevant information. Reports based on U.S. intelligence have suggested the lab collaborated on projects with the Chinese military.
But the most compelling reason to favor the lab leak hypothesis is firmly based in science. In particular, consider the genetic fingerprint of CoV-2, the novel coronavirus responsible for the disease Covid-19.
In gain-of-function research, a microbiologist can increase the lethality of a coronavirus enormously by splicing a special sequence into its genome at a prime location. Doing this leaves no trace of manipulation. But it alters the virus spike protein, rendering it easier for the virus to inject genetic material into the victim cell. Since 1992 there have been at least 11 separate experiments adding a special sequence to the same location. The end result has always been supercharged viruses.
A genome is a blueprint for the factory of a cell to make proteins. The language is made up of three-letter “words,” 64 in total, that represent the 20 different amino acids. For example, there are six different words for the amino acid arginine, the one that is often used in supercharging viruses. Every cell has a different preference for which word it likes to use most.
In the case of the gain-of-function supercharge, other sequences could have been spliced into this same site. Instead of a CGG-CGG (known as “double CGG”) that tells the protein factory to make two arginine amino acids in a row, you’ll obtain equal lethality by splicing any one of 35 of the other two-word combinations for double arginine. If the insertion takes place naturally, say through recombination, then one of those 35 other sequences is far more likely to appear; CGG is rarely used in the class of coronaviruses that can recombine with CoV-2.
In fact, in the entire class of coronaviruses that includes CoV-2, the CGG-CGG combination has never been found naturally. That means the common method of viruses picking up new skills, called recombination, cannot operate here. A virus simply cannot pick up a sequence from another virus if that sequence isn’t present in any other virus.
Although the double CGG is suppressed naturally, the opposite is true in laboratory work. The insertion sequence of choice is the double CGG. That’s because it is readily available and convenient, and scientists have a great deal of experience inserting it. An additional advantage of the double CGG sequence compared with the other 35 possible choices: It creates a useful beacon that permits the scientists to track the insertion in the laboratory.
Now the damning fact. It was this exact sequence that appears in CoV-2. Proponents of zoonotic origin must explain why the novel coronavirus, when it mutated or recombined, happened to pick its least favorite combination, the double CGG. Why did it replicate the choice the lab’s gain-of-function researchers would have made?
Yes, it could have happened randomly, through mutations. But do you believe that? At the minimum, this fact—that the coronavirus, with all its random possibilities, took the rare and unnatural combination used by human researchers—implies that the leading theory for the origin of the coronavirus must be laboratory escape.
When the lab’s Shi Zhengli and colleagues published a paper in February 2020 with the virus’s partial genome, they omitted any mention of the special sequence that supercharges the virus or the rare double CGG section. Yet the fingerprint is easily identified in the data that accompanied the paper. Was it omitted in the hope that nobody would notice this evidence of the gain-of-function origin?
But in a matter of weeks virologists Bruno Coutard and colleagues published their discovery of the sequence in CoV-2 and its novel supercharged site. Double CGG is there; you only have to look. They comment in their paper that the protein that held it “may provide a gain-of-function” capability to the virus, “for efficient spreading” to humans.
There is additional scientific evidence that points to CoV-2’s gain-of-function origin. The most compelling is the dramatic differences in the genetic diversity of CoV-2, compared with the coronaviruses responsible for SARS and MERS.
Both of those were confirmed to have a natural origin; the viruses evolved rapidly as they spread through the human population, until the most contagious forms dominated. Covid-19 didn’t work that way. It appeared in humans already adapted into an extremely contagious version. No serious viral “improvement” took place until a minor variation occurred many months later in England.
Such early optimization is unprecedented, and it suggests a long period of adaptation that predated its public spread. Science knows of only one way that could be achieved: simulated natural evolution, growing the virus on human cells until the optimum is achieved. That is precisely what is done in gain-of-function research. Mice that are genetically modified to have the same coronavirus receptor as humans, called “humanized mice,” are repeatedly exposed to the virus to encourage adaptation.
The presence of the double CGG sequence is strong evidence of gene splicing, and the absence of diversity in the public outbreak suggests gain-of-function acceleration. The scientific evidence points to the conclusion that the virus was developed in a laboratory.
Dr. Quay is founder of Atossa Therapeutics and author of “Stay Safe: A Physician’s Guide to Survive Coronavirus.” Mr. Muller is an emeritus professor of physics at the University of California Berkeley and a former senior scientist at the Lawrence Berkeley National Laboratory.
Kurt Kirkpatrick 6/8/21 MedPage Today 6/12/21 re the ORIGIN
The Wall Street Journal carried an op-ed by 2 scientists 6/6/21 that studied the genome of Sars-Cov-2 and found an unnatural sequence of nucleotides coding for Arg-Arg. See excerpt that follows: “The pair noted that the double CGG sequence has never been found naturally among the entire group of coronaviruses that includes CoV-2, which causes COVID-19.
But, in what Steven Quay and Richard Muller called a “damning fact,” it was found in CoV-2.
“Proponents of zoonotic origin must explain why the novel coronavirus, when it mutated or recombined, happened to pick its least favorite combination, the double CGG,” they wrote. “Why did it replicate the choice the lab’s gain-of-function researchers would have made?
“At the minimum, this fact — that the coronavirus, with all its random possibilities, took the rare and unnatural combination used by human researchers — implies that the leading theory for the origin of the coronavirus must be laboratory escape.”
https://www.vanityfair.com/news/2021/06/the-lab-leak-theory-inside-the-fight-to-uncover-covid-19s-origins
This Covid-19/CoV2-19 corona virus was originally named for its site of ORIGIN (Wuhan, China) as was the Ebola (a river in Zaire) virus, German measles, Rocky Mount spotted fever, Norovirus (Norwalk, Connecticut), and Spanish flu, etc. Corona virus-19/CoV2-19 was first documented mid-November, 2019, in China. Although the Chinese government stated that the virus originated in the United States, almost certainly it originated in either what is called a live or “wet” market where wild animals are sold for food in Wuhan, China. Or the virus escaped the research Wuhan National Biosafety Laboratory close to Wuhan, China: the latter is considered a reasonable possibility. Suggesting that the latter consideration is correct, satellite photos of traffic and cell-phone tracking data are reported to show a lockdown around the Wuhan Virology Institute lab, a leading world center research institute studying corona viruses (I’ve always wanted to say the Latinate “viri” as the correct pleural for the singular “virus”) beginning October 19,2019, and thereafter: implying a dire event there that had to be contained. There are reports that the laboratory’s staff have been silenced, outside-of China investigators have been limited in who they were allowed to talk to or data they were allowed to exam, even the investigators themselves seem to have conflicts of interest that would limit their investigative integrity: if so, all this reasonably points to a laboratory accident within the Wuhan Virology Laboratory that may well be the very start of the current pandemic. These are reasonable, but uncertain, considerations. It is unlikely that this will be settled.
A Chines scientist from the South China University of Technology’s opinion piece on the origins of the SARS CoV2-19 Virus print Feb 2020:
https://img-prod.tgcom24.mediaset.it/images/2020/02/16/114720192-5eb8307f-017c-4075-a697-348628da0204.pdf
A bit of humor: fromMedPage Today 6/12/21:
Here is the VERY IMPORTANT LINK to the article by renowned SCIENCE WRITER/reporter Nicholas Wade regarding the above:
https://nicholaswade.medium.com/origin-of-covid-following-the-clues-6f03564c038
From that just above link/article: “ … We stand together to strongly condemn conspiracy theories suggesting that COVID-19 does not have a natural origin,” a group of virologists and others wrote in the Lancet on February 19, 2020, when it was really far too soon for anyone to be sure what had happened. Scientists “overwhelmingly conclude that this coronavirus originated in wildlife,” they said, with a stirring rallying call for readers to stand with Chinese colleagues on the frontline of fighting the disease.
Contrary to the letter writers’ assertion, the idea that the virus might have escaped from a lab invoked accident, is not conspiracy. It surely needed to be explored, not rejected out of hand. A defining mark of good scientists is that they go to great pains to distinguish between what they know and what they don’t know. By this criterion, the signatories of the Lancet letter were behaving as poor scientists: they were assuring the public of facts they could not know for sure were true.
It later turned out that the Lancet letter had been organized and drafted by Peter Daszak, president of the EcoHealth Alliance of New York. Dr. Daszak’s organization funded coronavirus research at the Wuhan Institute of Virology. If the SARS2 virus had indeed escaped from research he funded, Dr. Daszak would be potentially culpable. This acute conflict of interest was not declared to the Lancet’s readers. To the contrary, the letter concluded, “We declare no competing interests.”
Virologists like Dr. Daszak had much at stake in the assigning of blame for the pandemic. For 20 years, mostly beneath the public’s attention, they had been playing a dangerous game. In their laboratories they routinely created viruses more dangerous than those that exist in nature. They argued they could do so safely, and that by getting ahead of nature they could predict and prevent natural “spillovers,” the cross-over of viruses from an animal host to people. If SARS Co V2-19 had indeed escaped from such a laboratory experiment, a savage blowback could be expected, and the storm of public indignation would affect virologists everywhere, not just in China. “It would shatter the scientific edifice top to bottom,” an MIT Technology Review editor, Antonio Regalado, said in March, 2020.
A second statement which had enormous influence in shaping public attitudes was a letter (in other words an opinion piece, not a scientific article) published on March, 17, 2020 in the journal Nature Medicine. Its authors were a group of virologists led by Kristian G. Andersen of the Scripps Research Institute. “Our analyses clearly show that SARS-CoV-2 is not a laboratory construct or a purposefully manipulated virus,” the five virologists declared in the second paragraph of their letter.
Unfortunately this was another case of poor science, in the sense defined above. True, some older methods of cutting and pasting viral genomes retain tell-tale signs of manipulation. But newer methods, called “no-see-um” or “seamless” approaches, leave no defining marks. Nor do other methods for manipulating viruses such as serial passage, the repeated transfer of viruses from one culture of cells to another. If a virus has been manipulated, whether with a seamless method or by serial passage, there is no way of knowing that this is the case. Dr. Andersen and his colleagues were assuring their readers of something they could not know.
The discussion part their letter begins, “It is improbable that SARS-CoV-2 emerged through laboratory manipulation of a related SARS-CoV-like coronavirus”. But wait, didn’t the lead say the virus had clearly not been manipulated? The authors’ degree of certainty seemed to slip several notches when it came to laying out their reasoning.
The reason for the slippage is clear once the technical language has been penetrated. The two reasons the authors give for supposing manipulation to be improbable are decidedly inconclusive.
First, they say that the spike protein of SARS2 binds very well to its target, the human ACE2 receptor, but does so in a different way from that which physical calculations suggest would be the best fit. Therefore the virus must have arisen by natural selection, not manipulation.
If this argument seems hard to grasp, it’s because it’s so strained. The authors’ basic assumption, not spelled out, is that anyone trying to make a bat virus bind to human cells could do so in only one way. First they would calculate the strongest possible fit between the human ACE2 receptor and the spike protein with which the virus latches onto it. They would then design the spike protein accordingly (by selecting the right string of amino acid units that compose it). But since the SARS2 spike protein is not of this calculated best design, the Andersen paper says, therefore it can’t have been manipulated.
But this ignores the way that virologists do in fact get spike proteins to bind to chosen targets, which is not by calculation but by splicing in spike protein genes from other viruses or by serial passage. With serial passage, each time the virus’s progeny are transferred to new cell cultures or animals, the more successful are selected until one emerges that makes a really tight bind to human cells. Natural selection has done all the heavy lifting. The Andersen paper’s speculation about designing a viral spike protein through calculation has no bearing on whether or not the virus was manipulated by one of the other two methods.
The authors’ second argument against manipulation is even more contrived. Although most living things use DNA as their hereditary material, a number of viruses use RNA, DNA’s close chemical cousin. But RNA is difficult to manipulate, so researchers working on coronaviruses, which are RNA-based, will first convert the RNA genome to DNA. They manipulate the DNA version, whether by adding or altering genes, and then arrange for the manipulated DNA genome to be converted back into infectious RNA.
Only a certain number of these DNA backbones have been described in the scientific literature. Anyone manipulating the SARS2 virus “would probably” have used one of these known backbones, the Andersen group writes, and since SARS2 is not derived from any of them, therefore it was not manipulated. But the argument is conspicuously inconclusive. DNA backbones are quite easy to make, so it’s obviously possible that SARS2 was manipulated using an unpublished DNA backbone….”
“ … In Sept. 2020, Dr. Li-Meng Yan, a virologist and former postdoctoral fellow in virology at the University of Hong Kong, repeated the theory on TV saying, “I can present solid scientific evidence to our audience that this virus, COVID-19 SARS-CoV-2 virus, actually is not from nature. It is a man-made virus created in the lab … “
Scientist recovers coronavirus gene sequences secretly deleted last year in Wuhan
By Jeanna Bryner – Live Science Editor-in-Chief 6/25/21
He finds 13 sequences from some of the earliest cases in Wuhan. The SARS-CoV-2 virus invades human cells by attaching to ACE2 receptors on the surfaces of those cells. (Image credit: Shutterstock). Finding the origin story for SARS-CoV-2, the coronavirus responsible for nearly 3.9 million deaths worldwide, has been largely hampered by lack of access to information from China where cases first popped up.
Now, a researcher in Seattle has dug up deleted files from Google Cloud that reveal 13 partial genetic sequences for some of the earliest cases of COVID-19 in Wuhan, Carl Zimmer reported for The New York Times. The sequences don’t tip the scales toward or away from one of the many theories about how SARS-CoV-2 came to be — they do not suggest the virus leaked from a high-security lab in Wuhan, nor do they suggest a natural spillover event. But they do firm up the idea that the novel coronavirus was circulating earlier than the first major outbreak at a seafood market.
Related: 14 coronavirus myths busted by science
In order to determine exactly how and where the virus originated, scientists need to find the so-called progenitor virus, the one from which all other strains descended. Until now, the earliest sequences are primarily those sampled from cases at the Huanan Seafood Market in Wuhan, which was initially thought to be where the novel coronavirus first emerged at the end of December, 2019. However, cases from early December and as far back as November, 2019, had no ties to the market, indicating pretty early in the pandemic that the virus emerged from another spot.
There was one nagging issue with those first genetic sequences. Those from cases found at the market include three mutations that are missing in virus samples from cases that popped up weeks later outside of the market. The viruses missing those three mutations matched more closely with the coronaviruses found in horseshoe bats. Scientists are relatively certain that the novel coronavirus somehow emerged from bats, so it’s logical to assume the progenitor would also be missing those mutations.
And now, Jesse Bloom of the Howard Hughes Medical Institute in Seattle has found the deleted sequences — likely some of the earliest samples — also were devoid of those mutations. (Bloom is the lead author in a letter published in May in the journal Science urging an unbiased investigation into the origins of the coronavirus, Live Science reported.)
“They’re three steps more similar to the bat coronaviruses than the viruses from the Huanan fish market,” Bloom told The New York Times. This new data hints that the virus was circulating in Wuhan well before it showed up at the seafood market, Bloom said. “This fact suggests that the market sequences, which are the primary focus of the genomic epidemiology in the joint WHO-China report … are not representative of the viruses that were circulating in Wuhan in late December of 2019 and early January of 2020,” Bloom wrote in his paper uploaded June 22 to the preprint database bioRxiv.
According to Zimmer, about a year ago 241 genetic sequences from coronavirus patients had gone missing from an online database called Sequence Read Archive that’s maintained by the National Institutes of Health (NIH).
Bloom noticed the missing sequences when he came across a spreadsheet in a study published in May 2020 in the journal PeerJ in which the authors list 241 genetic sequences of SARS-CoV-2 through the end of March 2020; the sequences were part of a Wuhan University project called PRJNA612766 and were supposedly uploaded to the Sequence Read Archive. He searched the archive database for the sequences and got the message “No items found,” Bloom wrote in the bioRxiv paper, which has not been peer-reviewed.
Related: 11 (sometimes) deadly diseases that hopped across species
His sleuthing revealed that the deleted sequences had been collected by Aisu Fu and Renmin Hospital of Wuhan University, and a preprint of the research published from those sequences (referred to as Wang et al. 2020) suggested they came from nose swab samples from outpatients with suspected COVID-19 early in the epidemic. Bloom couldn’t find any explanation for why the sequences had been deleted, and his emails to both corresponding authors to inquire received no response.
“There is no plausible scientific reason for the deletion: the sequences are perfectly concordant with the samples described in Wang et al. (2020a,b),” Bloom wrote in bioRxiv. “There are no corrections to the paper, the paper states human subjects approval was obtained, and the sequencing shows no evidence of plasmid or sample-to-sample contamination. It therefore seems likely the sequences were deleted to obscure their existence.”
Bloom notes several limitations to his study, primarily that the sequences are only partial and include no information to give a clear date or place of collection — information crucial to tracing the virus back to its origin. Regardless, Bloom thinks that looking more deeply at archived data from the NIH and other organizations — and piecing together the sequences — could help to paint a clearer picture of both the origin and early spread of SARS-CoV-2, all without needing on-the-ground studies in China.
Read more about the deleted sequences at The New York Times. Originally published on Live Science.
March 27, 2020
Potential Effects of Coronaviruses on the Cardiovascular System: A Review
Mohammad Madjid, MD, et al 3/27/20
Coronaviruses contain 4 major structural proteins: the spike (S) protein (which mediates attachment to the host receptor and subsequent fusion of the virus and cell membrane), the nucleocapsid (N) protein, the membrane (M) protein, and the envelope (E) protein.8
Four CoV types account for 15% to 30% of common colds.11
Coronaviruses (CoVs) are single-stranded positive-sense RNA viruses,Coronaviruses contain 4 major structural proteins: the spike (S) protein (which mediates attachment to the host receptor and subsequent fusion of the virus and cell membrane), the nucleocapsid (N) protein, the membrane (M) protein, and the envelope (E) protein.8
The SARS-CoV outbreak began in the Guangdong Province in southern China in November 2002, and was most likely linked to a zoonotic event in the wild-animal markets in China (HRS believes that there is a significant possibility that the virus leaked from the local virology institute). Soon after the isolation of SARS-CoV, SARS-CoV–like viruses were found in Himalayan palm civets and raccoon dogs, with 99.8% nucleotide homology to human SARS-CoV.14
The SARS-CoV belongs to the β-CoVs group and binds to the zinc peptidase angiotensin-converting enzyme 2 (ACE2), a surface molecule that is localized on the endothelial cells of arteries and veins, arterial smooth muscle, respiratory tract epithelium, epithelia of the small intestine, respiratory tract epithelium, and immune cells, to enter the host cell.15-17
Further studies of wild animals proved robust evidence that SARS-CoV might have originated in bats, when a SARS-like CoV was identified in Chinese horseshoe bats with a sequence similarity of 87% to 92% with human SARS-CoV, and it is believed that palm civets and raccoon dogs provided the intermediate amplification host for SARS-CoV before transmitting it to animal handlers in the animal market.14
In Practice Update b1/29/21 by Jonathan Temte, MD, PhD-condensed by HRS
SARS-CoV-2 Variants and Why They Matter
A concise overview of viral “variants”, has been provided by Lauring and Hodcroft.1 Mutation within a genome is the rule and not the exception, and that rates of change are governed by internal (biochemical) and external (selection) processes.
Coronaviruses—by the very nature of their RNA-based genome—have an intrinsically higher rate of mutation than DNA-based viruses. That said, they manifest a lower rate of mutation than influenza viruses due to the presence of genetic code for an enzyme that corrects some transcription errors. Understanding the nomenclature of a changing genome:
• Mutation is any substitution in a nucleotide within genome sequence
◦ can result in an amino acid change in protein synthesis
◦ can be neutral without affecting the protein structure
• Variant describes a group of viruses with change in the genomic sequence
◦ can involve a single mutation or many mutations
• Strain is used to describe a variant that imparts a phenotypic change in a virus
◦ transmissibility
◦ virulence (higher morbidity or mortality)
◦ antigenicity
As variants arise, other forces come into play. Arriving in a population with high susceptibility, there can be the “founder effect,” wherein the variant takes off as the only virus around. Natural selection can favor viruses with higher transmissibility, or the ability to escape from existing host immunity. Purifying selection results in the removal of mutations that are deleterious in hosts.
The concerned is mainly about those strains that have the potential to be more transmissible, have higher virulence, or alter the immune response gained from previous infection or vaccination. Three strains have come under scrutiny lately.2 In addition, all three have now been identified in the United States:
• UK (B.1.1.7) appears to have emerged with a large number of mutations in southeastern UK and has spread rapidly, implying heightened levels of transmissibility.
• South Africa (1.351) has multiple mutations in the spike protein, the antigenic basis for current vaccines.
• Brazil (P.1) emerged with 17 unique mutations, including three in the receptor-binding domain of the spike protein
1. Lauring AS, Hodcroft EB. Genetic variants of SARS-CoV-2—what do they mean? [published online ahead of print, 2021 Jan 6]. JAMA. 2021 Jan 6. doi: 10.1001/jama.2020.27124. Online ahead of print. https://jamanetwork.com/journals/jama/fullarticle/2775006
2. CDC. Emerging SARS-CoV-2 variants. Updated January 15, 2021. https://www.cdc.gov/coronavirus/2019-ncov/more/science-and-research/scientific-brief-emerging-variants.htm
Understanding immunity and viral waves: Why are COVID cases in India decreasing, despite the low double vaccination rate?
by Rajib Dasgupta, The Conversation 11/19/21
COVID continues to slow down in India. The festival season, which includes Durga Puja and Diwali where large groups of Indians gather, did not lead to a surge in cases. Epidemiological modelers had earlier predicted a third wave peaking during October and November.
Daily new cases have dropped from a peak of more than 400,000 per day in May 2021 to currently below 10,000 cases a day. And while antibody tests might give us a clue as to why, we can’t get complacent about vaccination rates.
Signals from recent antibody tests
In India, “serosurveys” have been regularly conducted since the pandemic began. This is where blood is tested from large numbers of people to check for the presence of COVID antibodies—the things our bodies make after being infected with COVID or receiving a COVID vaccine.
The fourth national survey in July reported present 67.6% of people across India had COVID antibodies, providing them with a level of immunity against the virus. At that time 24.8% of people were immunized with a single dose of vaccine and 13% were fully vaccinated. This means a large proportion of those with antibodies had actually been infected with COVID.
Delhi reported 97% of people were positive for antibodies in October, including 80% of children. Some 95.3% of those immunized with the Indian version of the Astrazeneca vaccine Covishield had developed antibodies, as did 93% of those who received India’s own vaccine Covaxin.
The state of Haryana’s serosurvey in October found antibodies in 76.3% of adults, upwards of 70% among children, and negligible difference between urban and rural populations. Kerala had the lowest sero-prevalence of 44.4% in the fourth national serosurvey in July, but in October it had risen to 82.6% among the general population and 85.3% among residents of urban slums.
What does this mean for a ‘third wave’ in India?
A third wave in India is an unlikely scenario with these high levels of antibodies, and vaccination levels continuing to rise.
It’s now recognized those who becoming naturally infected with COVID and recover before vaccination develop better immunity than those who only have antibodies from vaccination. This is referred to as “hybrid immunity”—those with previous SARS-CoV-2 infection mount unusually potent immune responses to the COVID vaccines.
The Centers for Disease Control in the US notes that both the fully vaccinated individuals and previously infected groups have a low risk of subsequent infection for at least six months.Results of the most recent national serosurvey in India reflect the seroprevalence during the third week of June 2021; the Delta-led second wave had bottomed out at that time. Though about 30% of the population remained susceptible, subsequent serosurveys and an absence of any post-festival surge confirm continuing high levels of protection.
“Patchwork vaccination” areas, where there are pockets of low coverage of vaccination among areas with high levels of coverage, run the risk of small outbreaks, but are unlikely to be large enough to be of any major epidemiological concern.
With high seropositivity among adults, many of the new cases can now be expected among children, particularly with the reopening of educational institutions. But high levels of immunization among teachers (upwards of 90%) and the emerging evidence that reopening schools has not been associated with significant increases in community transmission, are reassuring.
The WHO’s chief scientist said in late August that India seems to be “entering some stage of endemicity.” Endemic refers to the constant presence or usual prevalence of a disease in a population within a geographic area, where disease spread and rates are predictable.
Could a new variant, such as the Delta Plus subvariant first detected in India in April 2021 threaten the current relative stability? While it has been said it might be about 10–15% more transmissible than the Delta variant, the evidence from Europe suggests it has not yet been able to establish any dominance over Delta.
Is vaccination on track?
Of India’s 1.4 billion people, 26.9% are fully vaccinated and 54.9% have received at least one dose so far. But 35 million fewer women have been vaccinated compared to men and independent analyses show tribal and rural districts continue to lag.
There are two current targets: achieving 90% coverage of the first dose by the end of November and timely administration of the second dose. While the first is likely to be achieved, there are widespread reports of complacency regarding the second dose. A campaign is underway to encourage people to complete the schedule.
Delivering the billion plus doses has convincingly demonstrated vaccine confidence. But convincing people to take a vaccine when for many it seems like the risk has passed is a difficult task. Prior infection-induced immunity protects against reinfection but this acquired immunity wanes over time. Hence the recommendation for COVID vaccination for all eligible persons, including those who have been previously infected.
Districts with relatively low vaccine coverage require greater outreach efforts to reduce prevailing inequities. India’s immunization program has demonstrated its strengths in polio eradication and measles-rubella elimination campaigns. We need to borrow some of those techniques to ensure all Indians are protected against COVID.
Natural immunity compared to vaccine-induced immunity: The definitive summary
by Sl Attkisson 8/27/21
Updated Aug. 27, 2021, large Israel study that finds dramatically better protection from natural immunity than vaccination Updated Aug. 24 with Israel changing policies with recognition that the Pfizer vaccine allegedly wears off after several months.
Updated Aug. 6, 2021, with CDC analysis of Kentucky (unvaccinated Kentuckians had “2.34 times the odds of reinfection” compared with fully vaccinated) and national analysis in Israel (vaccinated Israelis were 6.72 times more likely to get infected after the shot than after natural infection). .
While it’s impossible to know, public health officials are grappling with the reality of an increasing number of fully-vaccinated Americans coming down with Covid-19 infections, getting hospitalized, and even dying of Covid. The Centers for Disease Control (CDC) insists vaccination is still the best course for every eligible American. But many are asking if they have better immunity after they’re infected with the virus and recover, than if they’re vaccinated.
Why does CDC seem to be “ignoring” natural immunity?
In fact, some medical experts have said they’re confounded by public health officials’ failure to factor natural and virus-acquired immunity into the Covid equation.
• Covid-19 Natural Immunity: The Definitive Summary
• Covid-19 Vaccine Concerns Summary
• Covid-19 Vaccine Analysis: Common Adverse Events
• Report a Possible Vaccine Adverse Event
ADVERSE EVENTS REPORT 10/4/21: https://justthenews.com/politics-policy/coronavirus/mon78-most-common-adverse-events-reported-after-covid-19-vaccination?utm_source=daily&utm_medium=email&utm_campaign=newsletter&utm_medium=email&utm_campaign=Daily%20Newsletter%20October%204%202021%20125421%20615b31bdcbe87&utm_content=Daily%20Newsletter%20October%204%202021%20125421%20615b31bdcbe87+CID_263517962d87af31f06f2518dc6fbdb3&utm_source=newsletter
However, vaccination rates alone tell little about a population’s true immune-status. And where high Covid case counts occur, it ultimately means a larger segment of that community ends up better-protected, vaccines aside. That’s according to virologists who point out that fighting off Covid, even without developing any symptoms, leaves people with what’s thought to be more robust and longer-lasting immunity than the vaccines confer.
The vaccine immunity problem
Hard data counters widespread public misinformation that claimed “virtually all” patients hospitalized and dying of Covid-19 are unvaccinated. Pfizer and Moderna had claimed their vaccines were “100% effective” at preventing serious illness. Many in the media even popularized a phrase designed to push more people to get vaccinated: “pandemic of the unvaccinated.”
Not so, says CDC and other data.
Listen to podcast with Dr. Jay Bhattacharya, who addresses the strength of natural immunity
Recent CDC data found that 74% of those who tested positive for Covid-19 in a Massachusetts analysis had been fully-vaccinated. Equally as troubling for those advocating vaccination-for-all: four out of five people hospitalized with Covid were fully-vaccinated. The CDC said “viral load” — indicating how able the human host is to spread Covid-19 — is about the same among the vaccinated and unvaccinated. Contrary to the misinformation by CDC Director Rochelle Walensky last May, vaccinated people can— and are— spreading Covid. (CDC officials later corrected Walensky’s false claim.)
Below: CDC’s data with light blue showing fully vaccinated. Dark blue is unvaccinated (but may include some fully vaccinated.)
CDC’s newest findings on so-called “breakthrough” infections in vaccinated people are mirrored by other data releases.
Illinois health officials recently announced more than 160 fully-vaccinated people have died of Covid-19, and at least 644 been hospitalized; ten deaths and 51 hospitalizations counted in the prior week. Israel’s Health Ministry recently said effectiveness of the Pfizer-BioNTech vaccine has fallen to 40 percent. Last month, 100 vaccinated British sailors isolated on a ship at sea reportedly came down with Covid seven weeks into their deployment. In July, New Jersey reported 49 fully vaccinated residents had died of Covid; 27 in Louisiana; 80 in Massachusetts. In Iceland there is a spike in cases, mostly among the vaccinated, among a highly-vaccinated population that had previously claimed to have defeated Covid-19. Of 116 cases diagnosed in one day, 73 were among the vaccinated; 43 were unvaccinated.
Nationally, as of July 12, 2021, the CDC said it was aware of more than 4,400 people who got Covid-19 after being fully vaccinated and had to be hospitalized; and 1,063 fully vaccinated people who died of Covid. But health officials still argue that vaccinated people make up only a small fraction of the seriously ill. Critics counter that CDC’s recent Massachusetts data demonstrates that is not so.
Update August 6: CDC has released a small analysis in Kentucky and indicated that it’s proof that vaccines are more effective than natural immunity. Read the analysis here. According to CDC, among the reinfected sample, 20% had been fully vaccinated. Among those who had Covid and were not later reinfected, 66% were unvaccinated. “Kentucky residents who were not vaccinated had 2.34 times the odds of reinfection compared with those who were fully vaccinated.” The data goes back and forth or is unclear.
Updated August 6: In Israel, more than 7,700 new cases of the virus have been detected during the most recent wave starting in May, but just 72 of the confirmed cases were reported in people who were known to have been infected previously – that is, less than 1% of the new cases. Roughly 40% of new cases – or more than 3,000 patients – involved people who had been infected despite being vaccinated. By contrast, Israelis who were vaccinated were 6.72 times more likely to get infected after the shot than after natural infection, with over 3,000 of the 5,193,499, or 0.0578%, of Israelis who were vaccinated getting infected in the latest wave.
https://sharylattkisson.com/2021/08/report-israel-vaccination-provides-far-less-protection-than-previous-covid-infection/
Updated August 27: A large study in Israel finds natural immunity confers longer lasting and stronger protection against infection, symptomatic disease and hospitalization for the Delta variant of Coronavirus compared to two-doses of Pfizer vaccine.
According to the study, after three months, risk of infection was 13.06 times higher among vaccinated patients and they were 27 times more likely to experience symptoms than unvaccinated people who’d had Covid previously.
Participants who were double jabbed were 5.96 times more likely to be infected and 7.13 times more likely to experience symptoms including cough, fever and shortness of breath.
September 15, 2021
Vaccine effectiveness high for preventing COVID-19 hospitalization
(HealthDay)—Vaccine effectiveness (VE) is high for preventing COVID-19-associated hospitalization, even during predominance of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) delta strain, according to two studies published in the Sept. 10 early-release issue of the U.S. Centers for Disease Control and Prevention Morbidity and Mortality Weekly Report.
Shaun J. Grannis, M.D., from the Regenstrief Institute in Indianapolis, and colleagues examined medical encounters from 187 hospitals and 221 emergency departments and urgent care clinics across nine states during June to August 2021. The researchers found that among adults hospitalized with COVID-19-like illness, SARS-CoV-2 infections were identified among 18.9 and 3.1 percent of unvaccinated and fully vaccinated patients, respectively. VE was 86 percent against COVID-19 hospitalization overall and was significantly lower among adults aged 75 years and older versus those aged 18 to 74 years (76 versus 89 percent). VE was highest for Moderna versus Pfizer-BioNTech and Janssen recipients across all ages (95 percent versus 80 and 60 percent, respectively). VE against COVID-19 emergency department/urgent care encounters were 82 percent overall and was highest for Moderna versus Pfizer-BioNTech and Janssen vaccine recipients (92 percent versus 77 and 65 percent, respectively).
Kristina L. Bajema, M.D., from the CDC COVID-19 Response Team, and colleagues examined mRNA VE against COVID-19-associated hospitalizations among 1,175 U.S. veterans aged 18 years or older at five Veterans Affairs Medical Centers during Feb. 1 to Aug. 6, 2021. The researchers found that the overall adjusted VE was 86.8 percent against COVID-19-associated hospitalization and was similar before and during delta prevalence (84.1 and 89.3 percent during Feb. 1 to June 30 and July 1 to Aug. 6, respectively). Among adults aged 65 years or older and those aged 18 to 64 years, VE was 79.8 and 95.1 percent, respectively.
Vaccine immunity may not really be waning much — which means universal booster shots may do little good. NYT 8/30/21: by David Leonhardt
August 30, 2021
By David Leonhardt
Vaccine immunity may not really be waning much — which means universal booster (HRS INSERTS HERE: AND MAY BE UNNECESSARY IN MOST) shots may do little good.
Late last month, researchers in Israel released some alarming new Covid-19 data. The data showed that many Israelis who had been among the first to receive the vaccine were nonetheless catching the Covid virus. Israelis who had been vaccinated later were not getting infected as often.
The booster-industrial complex
The study led to headlines around the world about waning immunity — the idea that vaccines lose their effectiveness over time. In the U.S., the Israeli study accelerated a debate about vaccine booster shots and played a role in the Biden administration’s recent recommendation that all Americans receive a booster shot eight months after their second dose.
But the real story about waning immunity is more complex than the initial headlines suggested. Some scientists believe that the Israeli data was misleading and that U.S. policy on booster shots has gotten ahead of the facts. The evidence for waning immunity is murky, these scientists say, and booster shots may not have a big effect.
After returning from an August break last week, I have spent time reaching out to scientists to ask for their help in understanding the current, confusing stage of the pandemic. How worried should vaccinated people be about the Delta variant? How much risk do children face? Which parts of the Covid story are being overhyped, and which deserve more attention?
I will be trying to answer these questions in the coming weeks. (I’d also like to know what questions you want answered; submit them here.)
One of the main messages I’m hearing from the experts is that conventional wisdom about waning immunity is problematic. Yes, the immunity from the Covid vaccines will wane at some point. But it may not yet have waned in a meaningful way.
“There’s a big difference between needing another shot every six months versus every five years,” Dr. David Dowdy, an epidemiologist at Johns Hopkins University, told me. “So far, looking at the data we have, I’m not seeing much evidence that we’ve reached that point yet.”
Simpson strikes again
At first glance, the Israeli data seems straightforward: People who had been vaccinated in the winter were more likely to contract the virus this summer than people who had been vaccinated in the spring.
Yet it would truly be proof of waning immunity only if the two groups — the winter and spring vaccine recipients — were otherwise similar to each other. If not, the other differences between them might be the real reason for the gap in the Covid rates.
As it turns out, the two groups were different. The first Israelis to have received the vaccine tended to be more affluent and educated. By coincidence, these same groups later were among the first exposed to the Delta variant, perhaps because they were more likely to travel. Their higher infection rate may have stemmed from the new risks they were taking, not any change in their vaccine protection.
Statisticians have a name for this possibility — when topline statistics point to a false conclusion that disappears when you examine subgroups. It’s called Simpson’s Paradox.
This paradox may also explain some of the U.S. data that the C.D.C. has cited to justify booster shots. Many Americans began to resume more indoor activities this spring. That more were getting Covid may reflect their newfound Covid exposure (as well as the arrival of Delta), rather than any waning of immunity over time.
‘Where is it?’
Sure enough, other data supports the notion that vaccine immunity is not waning much.
The ratio of positive Covid tests among older adults and children, for example, does not seem to be changing, Dowdy notes. If waning immunity were a major problem, we should expect to see a faster rise in Covid cases among older people (who were among the first to receive shots). And even the Israeli analysis showed that the vaccines continued to prevent serious Covid illness at essentially the same rate as before.
“If there’s data proving the need for boosters, where is it?” Zeynep Tufekci, the sociologist and Times columnist, has written.
Part of the problem is that the waning-immunity story line is irresistible to many people. The vaccine makers — Pfizer, Moderna and others — have an incentive to promote it, because booster shots will bring them big profits. The C.D.C. and F.D.A., for their part, have a history of extreme caution, even when it harms public health. We in the media tend to suffer from bad-news bias. And many Americans are so understandably frightened by Covid that they pay more attention to alarming signs than reassuring ones.
The bottom line
Here’s my best attempt to give you an objective summary of the evidence, free from alarmism — and acknowledging uncertainty:
Immunity does probably wane modestly within the first year of receiving a shot. For this reason, booster shots make sense for vulnerable people, many experts believe. As Dr. Céline Gounder of Bellevue Hospital Center told my colleague Apoorva Mandavilli, the C.D.C.’s data “support giving additional doses of vaccine to highly immunocompromised persons and nursing home residents, not to the general public.”
The current booster shots may do little good for most people. The vaccines continue to provide excellent protection against illness (as opposed to merely a positive Covid test). People will eventually need boosters, but it may make more sense to wait for one specifically designed to combat a variant. “We don’t know whether a non-Delta booster would improve protection against Delta,” Dr. Aaron Richterman of the University of Pennsylvania told me.
While Americans are focusing on booster shots, other policies may do much more to beat back Covid, including more vaccine mandates in the U.S.; a more rapid push to vaccinate the world (and prevent other variants from taking root); and an accelerated F.D.A. study of vaccines for children.
As always, we should be open to changing our minds as we get new evidence. As Richterman puts it, “We have time to gather the appropriate evidence before rushing into boosters.”
Implications of SARS-CoV-2 Viral Load in ChildrenGetting Back to School and Normal
Christina A. Rostad, MD1,2; Satoshi Kamidani, MD1,2; Evan J. Anderson, MD1,2,3
JAMA Pediatr. 2021;175(10):e212022. doi:10.1001/jamapediatrics.2021.2022
My email to the author 10/9/21: https://jamanetwork.com/journals/jamapediatrics/fullarticle/2780964?guestAccessKey=ff6bf21a-95ad-4fc0-bd4d-18df76495b6d&utm_source=silverchair&utm_campaign=jama_network&utm_content=healthpolicy_highlights&cmp=1&utm_medium=email
EJA: As you well know, in medicine and life, there is a time and place for virtually everything. You recommend the injection of a foreign protein into (vaccination of) children. Such is with benefit as well as some danger. Maybe I did not read your article closely enough, but what is the incidence of infection and infectivity of K-12 children that justifies your position? My general position in an adult double vaccinated world, even if not 100% vaccinated, but including those who have natural immunity, is that the usual risk-free people under the age of 35 do not need vaccination—in all reasonable probability. Moreover, I am experienced in the outpatient CoV2-19 treatment which protocol you can review at the below website. That 6 day early treatment obviates the need for hospitalization or a worse result in my inner city and small series of 35 adults. BTW, your article is so well done, it will be entered in to the ‘Summary Corona Virus Update’ located at that website. PS: I am an fellow Emory alum (Cardiology under Willis Hurst/Bruce Logue/Bob Schlant). Bob Silverstein
Original Investigation Editirail 6/11/21
Comparison of Symptoms and RNA Levels in Children and Adults With SARS-CoV-2 Infection in the Community Setting
Erin Chung, MD; Eric J. Chow, MD, MS, MPH; Naomi C. Wilcox, MPH; Roy Burstein, PhD; Elisabeth Brandstetter, MPH; Peter D. Han, MS; Kairsten Fay, BS; Brian Pfau, BS; Amanda Adler, MS; Kirsten Lacombe, RN, MSN; Christina M. Lockwood, PhD; Timothy M. Uyeki, MD, MPH, MPP; Jay Shendure, MD, PhD; Jeffrey S. Duchin, MD; Mark J. Rieder, PhD; Deborah A. Nickerson, PhD; Michael Boeckh, MD, PhD; Michael Famulare, PhD; James P. Hughes, PhD; Lea M. Starita, PhD; Trevor Bedford, PhD; Janet A. Englund, MD; Helen Y. Chu, MD, MPH
The SARS-CoV-2 pandemic has impacted the medical, economic, social, and political landscape worldwide. Despite public health measures to contain the virus, including masks, hand hygiene, school closures, and stay-at-home orders, COVID-19 was the third leading cause of death in the US in 2020. Although early data suggested that children accounted for only 2% of COVID-19 cases,1 more recent reports have indicated that 13% of diagnosed cases in the US have occurred in children. Additionally, the number of COVID-19 hospitalizations among children is similar to that observed in a typical influenza season, and COVID-19 deaths in children exceed the total observed in any single influenza season.2,3 In March 2020, in an attempt to curb the exponential growth of the COVID-19 pandemic, kindergarten through 12th-grade schools closed in all 50 US states, affecting 57 million students.4 The effects of these school closures have been far reaching, including marked social disruption and psychological and educational impact. Nevertheless, the resumption of in-person learning has proven controversial, as officials and communities weigh the risks of potential transmission of SARS-CoV-2 in schools against the benefits of in-person learning. To adequately assess these risks, it is important to address 3 key questions: (1) What is the role of children in community transmission of infectious pathogens? (2) What is the role of children specifically in SARS-CoV-2 transmission? and (3) What can be done to get children safely back in daycare, preschool, and school?
In addressing the first question, it is critical to recognize that interacting with children carries inherent exposure to and risk of infectious diseases for daycare workers, teachers, parents, or grandparents. Children play a large role in community transmission of multiple infectious pathogens, including hepatitis A, respiratory syncytial virus, cytomegalovirus, influenza, rotavirus, and invasive pneumococcal disease. For some of these pathogens, the extent of child-to-adult transmission was identified only after pediatric vaccination affected adult disease burden.5 Closure of schools and stay-at-home orders in the spring of 2020 had a marked impact on respiratory syncytial virus and influenza community transmission well before masking was routinely recommended. These observations have provided a framework to understand how the home, school, and community interface to influence transmission dynamics of infectious diseases. In this way, they have also helped inform strategies to mitigate transmission of SARS-CoV-2.
So what is known about the role of children in transmission of SARS-CoV-2 to other children and to adults? Transmission likely depends on multiple factors, including symptoms (ie, type, severity, and duration), the duration and timing of exposure (ie, presymptomatic or symptomatic), the amount of virus in the infected individual (viral load), the viral variant (eg, B.1.1.7), and host factors, such as baseline susceptibility and immune responses in the exposed individual. Furthermore, the risk of developing symptoms (eg, COVID-19) once infected with SARS-CoV-2 may depend on host factors, including age and comorbidities. Any such factors may differ between children and adults. Thus, determining the role of children in the transmission of SARS-CoV-2 is challenging.
Chung et al6 performed a cross-sectional, countywide, community-based surveillance study in King County, Washington, of SARS-CoV-2 symptoms and viral RNA levels among children and adults. The authors analyzed more than 37 000 nasal swab samples to identify 123 children and 432 adults with SARS-CoV-2 infection. They found that children were less frequently symptomatic (61.8% of children vs 92.8% of adults), had fewer symptoms (mean [SD], 1.6 [2.0] among children vs 4.5 [3.1] among adults), and had shorter duration of symptoms (mean [SD], 3.8 [3.8] among children vs 4.9 [4.1] among adults). Compared with asymptomatic individuals, symptomatic individuals had lower SARS-CoV-2 cycle threshold (Ct) values, which corresponds to higher viral RNA levels. Importantly, there was no difference in Ct values between symptomatic children and symptomatic adults, nor between asymptomatic children and asymptomatic adults. Although viral RNA levels were measured using Ct values, which are a semiquantitative surrogate of viral load, a recent study demonstrated correlation between both Ct values and RNA viral loads with cultivable SARS-CoV-2 virus from the nasopharynx.7 These data corroborate recent studies of asymptomatic children and studies of viral load by age.6 To put these data into the context of transmission risk, a recent study demonstrated that SARS-CoV-2 Ct values almost linearly inversely correlated with transmission.8 Furthermore, a meta-analysis found that the risk of asymptomatic transmission is significantly lower than that of symptomatic transmission (relative risk, 0.58; 95% CI, 0.34-0.99; P = .047).9 Taken together, these findings suggest that children may be less likely to transmit SARS-CoV-2 because of reduced frequency and severity of symptoms, which are associated with reduced viral load.
While the relative risk of transmission from children with SARS-CoV-2 infection remains uncertain, it is clear that children can transmit the virus. Early data from a study in South Korea suggested that children older than 10 years had a high risk of transmitting COVID-19.10 Subsequent studies have indicated a low but appreciable SARS-CoV-2 transmission risk from children. In a meta-analysis of household transmission dynamics, 3.8% of transmission clusters had a pediatric index case, and secondary infection rates of pediatric household contacts were lower than adult household contacts (relative risk, 0.62; 95% CI, 0.42-0.91).11 In schools, transmission typically follows trends in community transmission, rather than preceding or augmenting them.12 Schools have not been associated with frequent outbreaks13 or substantial increases in community transmission as measured by COVID-19–associated hospitalizations.14 Recent data demonstrate that although the risk of COVID-19 is greater among children who are attending school in person, this risk disappears with layered prevention measures.15 The collective implications of these studies are that children appear to be less likely to transmit SARS-CoV-2 than adults, and that transmission in schools can be mitigated.
What then can be done to get kids back to daycare, preschool, and school safely? Our increasing understanding of transmission dynamics among children informs decisions about in-person learning. The US Centers for Disease Control and Prevention emphasizes that kindergarten through 12th-grade schools can resume in-person learning through phased reopening,16 and prioritization of the correct and consistent use of masks and physical distancing. Layering prevention strategies, including teacher masking, daily symptom screens, and appropriate isolation and quarantine, further reduces transmission risk in schools. The study by Chung et al6 underscores the potential transmission risk of symptomatic children and the importance of isolating while symptomatic. Keeping symptomatic children home would have benefits for prevention not only of SARS-CoV-2, but also of influenza, respiratory syncytial virus, and other infectious pathogens. Additionally, extracurricular activities are important in SARS-CoV-2 transmission, and measures to prevent transmission should be used.16
Finally, to get back to something resembling the normal life of 2019, vaccination of children is needed. As has been observed for other pathogens, local vaccine hesitancy can result in sustained transmission of vaccine-preventable infections.5 Without vaccination, children will continue to serve as a reservoir of SARS-CoV-2 infections, experiencing preventable morbidity and delaying the development of herd immunity.17 The Pfizer-BioNTech BNT162b2 vaccine has received emergency use authorization by the US Federal Drug Administration (FDA) for administration to adults and adolescents 12 years and older, and Moderna has released data demonstrating the safety and efficacy of its vaccine (mRNA-1273) for adolescents 12 years and older. Clinical trials are also underway to assess the safety and immunogenicity of BNT162b2 and mRNA-1273 for children 6 months and older (NCT04816643 and NCT04796896, respectively). Expanding vaccine eligibility for children and addressing vaccine hesitancy will provide greater opportunities to layer protection and reduce transmission risk in schools. Additionally, encouraging uptake of vaccines among educators and school staff is critical to reduce their risk of occupational acquisition (eg, from children or other staff) and transmission of SARS-CoV-2 to others. As in-school transmission events parallel community transmission, we must all remain vigilant in our commitment to public health prevention strategies, flexible in our approach to an evolving virus, and resilient in our efforts to safely provide the best learning opportunities for our children.
Published Online: June 11, 2021. doi:10.1001/jamapediatrics.2021.2022
From the prestigious journal ‘Nature’ 9/2021: Older Israelis (those with bone marrow transplants, dementia, cancer chemotherapy, mental retardation, and nursing home residents were most who profited from the third immunization) who have received a third dose of a COVID-19 vaccine are 20 times less likely to test positive for SARS-CoV-2 or to develop severe COVID-19 than are those who have had only two injections, according to a highly anticipated study published on 15 September1.
The standard regimen for messenger RNA-based COVID-19 vaccines is two doses, but some governments, including Israel’s, have started administering third ‘booster’ shots. The latest study evaluated 1.1 million Israelis over the age of 60 who had received their first two doses at least five months earlier. Twelve or more days after receiving a third jab, participants were about 19.5 times less likely to have severe COVID-19 than were people in the same age group who had received only two jabs and were studied during a similar time period.
The bright side of recovering from Covid-19
But there’s promising news to be found within natural and acquired immunity statistics, according to virologists. As of May 29, CDC estimated more than 120 million Americans— more than one in three— had already battled Covid. While an estimated six-tenths of one-percent died, the other 99.4% of those infected survived with a presumed immune status that appears to be superior to that which comes with vaccination.
If doctors could routinely test to confirm who has fought off and become immune to Covid-19, it would eliminate the practical need or rationale for those protected millions to get vaccinated. It would also allow them to avoid even the slight risk of serious vaccine side effects.
Read more about Israel cases here
Unfortunately, virologists say no commonly-used test can detect with certainty whether a person is immune. A common misconception is that antibody tests can make that determination. But experts say immunity after infection or exposure often comes without a person producing or maintaining measurable antibodies.
Because of that reality, people who have had asymptomatic infections — infections where they suffered no symptoms — have no easy way to know that they’re immune. However, a growing body of evidence indicates that the millions who know they got Covid can be assured they’re unlikely to suffer reinfection, for at least as long of a time period that scientists have been able to measure. Possibly far beyond.
Update Aug. 24, 2021: With evidence of seriously-waning immunity at the five- to six-month mark, CDC is now recommending a third “booster” shot. In highly-vaccinated Israel, recognition that vaccination likely does not confer protection beyond a few months, the country has reimagined its vaccine passports. They will only apply to people who have had three shots, and only be good for a six month period of time.
The immunity-after-Covid-infection studies
The following are some of the data and studies regarding immunity acquired after Covid infection.
Comparing SARS-CoV-2 natural immunity to vaccine-induced immunity: reinfections versus breakthrough infections Aug. 24, 2021
This large study in Israel shows “natural immunity confers longer lasting and stronger protection against infection, symptomatic disease and hospitalization caused by the Delta variant of SARS-CoV-2, compared to the BNT162b2 two-dose vaccine-induced immunity.”
Longitudinal analysis shows durable and broad immune memory after SARS-CoV-2 infection with persisting antibody responses and memory B and T cells, July 20, 2021
This study followed 254 Covid-19 patients for up to 8 months and concluded they had “durable broad-based immune responses.” In fact, even very mild Covid-19 infection also protected the patients from an earlier version of “SARS” coronavirus that first emerged around 2003, and against Covid-19 variants. “Taken together, these results suggest that broad and effective immunity may persist long-term in recovered COVID-19 patients,” concludes the study scientists.
Associations of Vaccination and of Prior Infection With Positive PCR Test Results for SARS-CoV-2 in Airline Passengers Arriving in Qatar, June 9, 2021
This study of airline passengers in Qatar found that both vaccination and prior infection were “imperfect” when it comes to preventing positive Covid-19 test results, but that the incidence of reinfection is similarly low in both groups. (In an emil to HRS, aVERYexperienced PhD vaccine researcher sent me this IMPORTANT IF NOT CRUCIAL information: “ …The PCR test faulty at 35 cycles. All the PCR tests overseen by the CDC are set at 37 to 45 cycles. The CDC admits that any tests over 28 cycles are not admissible for a positive reliable result. This alone invalidates over 90% of the alleged covid cases “infections” tracked by the use of this faulty test …” !!!!
Necessity of COVID-19 vaccination in previously infected individuals, June 1, 2021
This study followed 52,238 employees of the Cleveland Clinic, the premier cardiology institution if the USA Health System in Ohio.
For previously-infected people, the cumulative incidence of re-infection “remained almost zero.” According to the study, “Not one of the 1,359 previously infected subjects who remained unvaccinated had a [Covid-19] infection over the duration of the study” and vaccination did not reduce the risk. “Individuals who have had [Covid-19] infection are unlikely to benefit from COVID-19 vaccination,” concludes the study scientists.
SARS-CoV-2 specific memory B-cells from individuals with diverse disease severities recognize SARS-CoV-2 variants of concern, May 29, 2021
This study found strong immune signs in people who had previously been infected with Covid-19, including “those [who] experienced asymptomatic or mild disease.” The study concludes there is “reason for optimism” regarding the capacity of prior infection “to limit disease severity and transmission of variants of concern as they continue to arise and circulate.”
A population-based analysis of the longevity of SARS-CoV-2 antibody seropositivity in the United States, May 24, 2021
This study of real world data extended the timeframe of available data indicating that patients have strong immune indicators for “almost a year post-natural infection of COVID-19.” The study concludes the immune response after natural infection “may persist for longer than previously thought, thereby providing evidence of sustainability that may influence post-pandemic planning.”
SARS-CoV-2 infection induces long-lived bone marrow plasma cells in humans, May 24, 2021
This study examined bone marrow of previously-infected patients and found that even mild infection with Covid-19 “induces robust antigen-specific, long-lived humoral immune memory in humans.” The study indicates “People who have had mild illness develop antibody-producing cells that can last lifetime.”
People who have had mild illness develop antibody-producing cells that can last lifetime.
World Health Organization (WHO) scientific brief, May 10, 2021
This scientific brief issued by WHO states that after natural infection with Covid-19, “available scientific data suggests that in most people immune responses remain robust and protective against reinfection for at least 6-8 months.”
Detection of SARS-CoV-2-Specific Humoral and Cellular Immunity in COVID-19 Convalescent Individuals, May 3, 2020
This study found humoral and cellular immunity in recovered Covid patients. “Production of S-RBD-specific antibodies were readily detected in recovered patients. Moreover, we observed virus-neutralization activities in these recovered patients,” wrote the study authors.
The adaptive immune system consists of three major lymphocyte types: B cells (antibody producing cells), CD4+ T cells (helper T cells), and CD8+ T cells (cytotoxic, or killer, T cells
From: Antigen-Specific Adaptive Immunity to SARS-CoV-2 in Acute COVID-19 and Associations with Age and Disease Severity
Protection of previous SARS-CoV-2 infection is similar to that of BNT162b2 vaccine protection: A three-month nationwide experience from Israel, April 24, 2021
This study from Israel found a slight advantage to natural infection over vaccination when it comes to preventing a reinfection and severe illness from Covid-19.
The study authors concluded, “Our results question the need to vaccinate previously-infected individuals.”
A 1 to 1000 SARS-CoV-2 reinfection proportion in members of a large healthcare provider in Israel: a preliminary report, March 6, 2021
This study found a rare Covid-19 positive test “reinfection” rate of 1 per 1,000 recoveries.
Lasting immunity found after recovery from COVID-19, Jan. 26, 2021
Research funded by the National Institutes of Health and published in Science early in the Covid-19 vaccine effort found the “immune systems of more than 95% of people who recovered from COVID-19 had durable memories of the virus up to eight months after infection,” and hoped the vaccines would produce similar immunity. Experts say tthis dise not seem to be the case.
SARS-CoV-2 reinfection in a cohort of 43,000 antibody-positive individuals followed for up to 35 weeks, Jan. 15, 2021
This study found Covid-19 natural infection “appears to elicit strong protection against reinfection” for at least seven months. “Reinfection is “rare,” concludes the scientists.
Immunological memory to SARS-CoV-2 assessed for up to eight months after infection, Nov. 1, 2020
This study confirmed and examined “immune memory” in previously-infected Covid-19 patients.
Negligible impact of SARS-CoV-2 variants on CD4+ and CD8+ T cell reactivity in COVID-19 exposed donors and vaccinees, Nov. 1, 2020
This study concluded “T cell” immune response in former Covid-19 patients likely continues to protect amid Covid-19 variants.
Orthogonal SARS-CoV-2 Serological Assays Enable Surveillance of Low-Prevalence Communities and Reveal Durable Humoral Immunity, Oct. 13, 2020
This study found that “neutralizing antibodies are stably produced for at least 5–7 months” after a patient is infected with Covid-19.
SARS-CoV-2-specific T cell immunity in cases of COVID-19 and SARS, and uninfected controls, July 25, 2020
This study found that all patients who recently recovered from Covid-19 produced immunity-strong T cells that recognize multiple parts of Covid-19.
They also looked at blood samples from 23 people who’d survived a 2003 outbreak of a coronavirus: SARS (Cov-1). These people still had lasting memory T cells 17 years after the outbreak. Those memory T cells, acquired in response to SARS-CoV-1, also recognized parts of Covid-19 (SARS-CoV-2).
Much of the study on the immune response to SARS-CoV-2, the novel coronavirus that causes COVID-19, has focused on the production of antibodies. But, in fact, immune cells known as memory T cells also play an important role in the ability of our immune systems to protect us against many viral infections, including—it now appears—COVID-19.
“Immune T Cells May Offer Lasting Protection Against COVID-19”
Estimated US Infection- and Vaccine-Induced SARS-CoV-2 Seroprevalence Based on Blood Donations, July 2020-May 2021
Jefferson M. Jones, MD, MPH1; et al
JAMA. Published online 9/2/21. doi:10.1001/jama.2021.15161
•
Change in Donor Characteristics and Antibodies to SARS-CoV-2 in Donated Blood in the US, June-August 2020
Roger Y. Dodd, PhD; Meng Xu, MPH; Susan L. Stramer, PhD
•
Change in Antibodies to SARS-CoV-2 Among Health Care Personnel in Nashville, Tennessee
Manish M. Patel, MD; Natalie J. Thornburg, PhD; William B. Stubblefield, MD; H. Keipp Talbot, MD, MPH; Melissa M. Coughlin, PhD; Leora R. Feldstein, PhD, MSc; Wesley H. Self, MD, MPH
•
Seroprevalence of SARS-CoV-2 Antibodies in the US Adult Asymptomatic Population
Robert L. Stout, PhD; Steven J. Rigatti, MD
•
Prevalence of SARS-CoV-2 Antibodies From a Serosurveillance of Kenyan Blood Donors, January to March 2021
Sophie Uyoga, PhD; Ifedayo M. O. Adetifa, MD, PhD; Mark Otiende, MSc; Christine Yegon, BSc; Ambrose Agweyu, MD, PhD; George M. Warimwe, MRCVS, PhD; J. Anthony G. Scott, MD
Question Based on blood donations in the US from July 2020 through May 2021, how did infection- and vaccine-induced SARS-CoV-2 seroprevalence vary over time by demographic group and by geographic region?
Findings In this repeated cross-sectional study that included 1 443 519 blood donation specimens from a catchment area representing 74% of the US population, estimated SARS-CoV-2 seroprevalence weighted for differences between the study sample and general population increased from 3.5% in July 2020 to 20.2% for infection-induced antibodies and 83.3% for combined infection- and vaccine-induced antibodies in May 2021. Seroprevalence differed by age, race and ethnicity, and geographic region of residence, but these differences changed over the course of the study.
Design, Setting, and Participants In a repeated cross-sectional study conducted each month during July 2020 through May 2021, 17 blood collection organizations with blood donations from all 50 US states; Washington, DC; and Puerto Rico were organized into 66 study-specific regions, representing a catchment of 74% of the US population. For each study region, specimens from a median of approximately 2000 blood donors were selected and tested each month; a total of 1 594 363 specimens were initially selected and tested. The final date of blood donation collection was May 31, 2021.
Main Outcomes and Measures … both spike and nucleocapsid antibodies. Combined infection- and vaccination-induced seroprevalence was defined as the prevalence of the population with spike antibodies. The seroprevalence estimates were compared with cumulative COVID-19 case report incidence rates.
Results Among 1 443 519 specimens included, 733 052 (50.8%) were from women, 174 842 (12.1%) were from persons aged 16 to 29 years, 292 258 (20.2%) were from persons aged 65 years and older, 36 654 (2.5%) were from non-Hispanic Black persons, and 88 773 (6.1%) were from Hispanic persons. The overall infection-induced SARS-CoV-2 seroprevalence estimate increased from 3.5% (95% CI, 3.2%-3.8%) in July 2020 to 20.2% (95% CI, 19.9%-20.6%) in May 2021; the combined infection- and vaccination-induced seroprevalence estimate in May 2021 was 83.3% (95% CI, 82.9%-83.7%). By May 2021, 2.1 SARS-CoV-2 infections (95% CI, 2.0-2.1) per reported COVID-19 case were estimated to have occurred.
Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection
• David S. Khoury, et al 5/17/21 Nature Medicine volume 27, pages 1205–1211 (2021)Cite this article
Abstract
Predictive models of immune protection from COVID-19 are urgently needed to identify correlates of protection to assist in the future deployment of vaccines. To address this, we analyzed the relationship between in vitro neutralization levels and the observed protection from severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection using data from seven current vaccines and from convalescent cohorts. We estimated the neutralization level for 50% protection against detectable SARS-CoV-2 infection to be 20.2% of the mean convalescent level (95% confidence interval (CI) = 14.4–28.4%). The estimated neutralization level required for 50% protection from severe infection was significantly lower (3% of the mean convalescent level; 95% CI = 0.7–13%, P = 0.0004). Modeling of the decay of the neutralization titer over the first 250 d after immunization predicts that a significant loss in protection from SARS-CoV-2 infection will occur, although protection from severe disease should be largely retained. Neutralization titers against some SARS-CoV-2 variants of concern are reduced compared with the vaccine strain, and our model predicts the relationship between neutralization and efficacy against viral variants. Here, we show that neutralization level is highly predictive of immune protection, and provide an evidence-based model of SARS-CoV-2 immune protection that will assist in developing vaccine strategies to control the future trajectory of the pandemic.
Main
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has spread globally over the past year, infecting an immunologically naive population and causing significant morbidity and mortality. Immunity to SARS-CoV-2 induced either through natural infection or vaccination has been shown to afford a degree of protection against reinfection and/or reduce the risk of clinically significant outcomes. Seropositive recovered subjects have been estimated to have 89% protection from reinfection1, and vaccine efficacies from 50 to 95% have been reported2. However, the duration of protective immunity is presently unclear, primary immune responses are inevitably waning3,4,5, and there is ongoing transmission of increasingly concerning viral variants that may escape control by both vaccine-induced and convalescent immune responses6.
A critical challenge at present is to identify the immune correlate(s) of protection from SARS-CoV-2 infection and thereby predict how changes in immunity will be reflected in clinical outcomes. A defined correlate of protection will permit both confidence in opening up economies and facilitate rapid improvements in vaccines and immunotherapies. In influenza infection, for example, a hemagglutination inhibition (HAI) titer of 1:40 is thought to provide 50% protection from influenza infection7 (although estimates range from 1:17 to 1:110, refs. 8,9). This level was established over many years using data from a standardized HAI assay10 applied to serological samples from human challenge and cohort studies. This assay is used to predict vaccine efficacy and to assist in the annual reformulation of seasonal influenza vaccines. At present, however, there are few standardized assays for assessing SARS-CoV-2 immunity, little data comparing immune levels in susceptible versus resistant individuals, and no human challenge model11.
The data currently available for SARS-CoV-2 infection include immunogenicity data from phase 1 and 2 studies of vaccines, and data on protection from preliminary reports from phase 3 studies and from seropositive convalescent individuals (Supplementary Tables 1 and 2). Although antiviral T and B cell memory certainly contribute some degree of protection, strong evidence of a protective role for neutralizing serum antibodies exists. For example, passive transfer of neutralizing antibodies can prevent severe SARS-CoV-2 infection in multiple animal models,12,13 and Regeneron has recently reported similar data in humans14. We therefore focus our studies on in vitro virus neutralization titers reported in studies of vaccinated and convalescent cohorts. Unfortunately, the phase 1 and 2 studies all use different assays for measuring neutralization. Normalization of responses against a convalescent serum standard has been suggested to provide greater comparability between the results from different assays15. Although all studies compare immune responses after vaccination against the responses in convalescent individuals, the definition of convalescence is not standardized across studies. Similarly, among phase 3 studies, the timeframes of study and the case definitions of infection also vary (Supplementary Table 2). Recognizing these limitations, our aim was to investigate the relationship between vaccine immunogenicity and protection.
Results
Identification of neutralization titer as a correlate of immune protection
To compare neutralization titers across studies, we determined the mean and standard deviation (on a log scale) of the neutralization titer in published data from seven vaccine studies (mRNA-1273, NVX-CoV2373, BNT162b2, rAd26-S+rAd5-S, ChAdOx1 nCoV-19, Ad26.COV2.S and CoronaVac) and one convalescent study3,16,17,18,19,20,21,22 (Supplementary Table 1). Because of the different assays used in each study, neutralization titers were normalized to the mean convalescent titer using the same assay in the same study (noting that the definition of convalescence was also not standardized across studies and a variable number of convalescent samples are analyzed in each study). We then compared this normalized neutralization level in each study against the corresponding protective efficacy reported from the seven phase 3 clinical trials19,23,24,25,26,27,28,29 (detailed in Supplementary Table 2). Despite the known inconsistencies between studies, comparison of normalized neutralization levels and vaccine efficacy demonstrates a remarkably strong non-linear relationship between mean neutralization level and the reported protection across different vaccines (Spearman r = 0.905; P = 0.0046) (Fig. 1a).
Fig. 1: Understanding the relationship between neutralization and protection.
a, Relationship between neutralization level and protection from SARS-CoV-2 infection. The reported mean neutralization level from phase 1 and 2 trials and the protective efficacy from phase 3 trials for seven vaccines, as well as the protection observed in a seropositive convalescent cohort, are shown (details of data sources are given in Supplementary Tables 1 and 2). The 95% CIs are indicated as vertical and as horizontal whiskers. The red solid line indicates the best fit of the logistic model and the red shading indicates the 95% predictive interval of the model. The mean neutralization level and protective efficacy of the Covaxin vaccine are indicated as a green circle (data from this study were available only after modeling was complete and did not contribute to fitting). b, Schematic illustration of the logistic approach to identifying the protective neutralization level. The data for each study include the distribution of the measured in vitro neutralization titer against SARS-CoV-2 in vaccinated or convalescent subjects (as a proportion of the mean titer in convalescent subjects (dashed line)) (blue/red bell curve), accompanied by a level of protective efficacy for the same regimen. The efficacy is illustrated by the proportions of the bell curve ‘protected’ (blue) and ‘susceptible’ (red) for individual studies. The modeling fits the optimal 50% protective neutralization level (blue solid line, the shaded area indicates the 95% CI) that best estimates the correct levels of protection observed across the different studies. c, Predictions of the leave-one-out analysis. Modeling was repeated multiple times using all potential sets of the seven vaccination studies and the convalescent study to predict the efficacy of the eighth study. The diagonal dashed line indicates the position of a 1:1 correlation (i.e., the relationship if the model were completely accurate). The horizontal whiskers indicate 95% CIs and the vertical whiskers indicate 95% predictive intervals.
Full size image
Estimation of the protective neutralization level against COVID-19
To further dissect the relationship between immunogenicity and protection in SARS-CoV-2 we considered the parallels with previous approaches to estimating a ‘50% protective titer’ in influenza infection. These historic studies in influenza involved comparison of HAI titers in infected versus uninfected subjects (in either natural infection or human challenge studies) and used logistic or receiver operating characteristic approaches to identify an HAI titer that provided protection7,8,9,30,31. We adapted these approaches to analyze the existing data on reported ‘mean neutralization level’ in different studies and the observed level of protection from infection (details of statistical methods are provided in the Methods).
We first fitted a logistic model to estimate the ‘50% protective neutralization level’ (across all studies) that best predicted the protective effect observed in each study (consistent with the use of a logistic function to model protection in influenza serological studies30,31). We found that this model provided a good explanation of the relationship between mean neutralization level and protection across the studies, and determined that the estimated 50% protective neutralization level was 20.2% (95% confidence interval (CI) = 14.4–28.4%) of the mean convalescent level (Fig. 1a,b). Given that different neutralization assays were used for each study11 (see above and Supplementary Table 1), a 50% protective neutralization level equivalent to 20% of the mean titer in the convalescent subjects equates to a measured in vitro neutralization titer of between 1:10 and 1:30 in most of the assays reported (although up to 1:200 in one assay), or we estimate approximately 54 international units (IU)/ml (95% CI 30–96 IU/ml) (Supplementary Table 4). Given that the model is dependent on the mean and distribution of neutralization levels, we also estimated these using different approaches, which led to similar estimates (see Methods and Extended Data Fig. 1).
To relax the assumption that neutralization levels are normally distributed in the above model, we also estimated the protective level using a distribution-free approach and applied this to the raw data for individual neutralization levels reported in the studies. We refer to this as the ‘protective neutralization classification model’. Although this approach may be slightly unrealistic in applying a protected or unprotected cut-off in a binary fashion (unlike the logistic approach), it has the advantage of being independent of any assumptions of the distribution of neutralization titers. Using this classification approach the estimated protective threshold was 28.6% (95% CI = 19.2–29.2%) of the mean convalescent level. As expected, the estimated protective level using the classification method was slightly higher than the 50% protective level estimated using the logistic method, given that the classification method essentially estimates a level of 100% protection instead of 50% protection.
This analysis suggests that the mean in vitro neutralization level of a vaccine measured early after vaccination is predictive of the subsequent protective efficacy measured in phase 3 trials, and estimates that the 50% neutralization level for SARS-CoV-2 is approximately 20% of the mean convalescent titer. To test the potential utility of this in predicting the protective efficacy of an unknown vaccine, we repeated our analysis using a leave-one-out approach. That is, we repeated our analysis by removing one of the datasets and fitting the model to the remaining seven vaccine or convalescent studies. We then used the parameters obtained from this fitting to predict the efficacy of the eighth (removed) dataset. We repeated this by removing each dataset one at a time. Figure 1c shows the results of using the logistic model of protection to predict the efficacy of each vaccine from the results of the other seven. In addition, after fitting the model to the data for eight vaccine or convalescent studies, the phase 3 efficacy results of another vaccine (BBV152) were released in a press release on 3 March 2021 (ref. 32). Using the observed neutralization level (a mean of 79.2% of the convalescent titer in that study33 (Supplementary Table 1), the predicted efficacy of the new vaccine was 79.6% (95% predictive interval: 76.2–83.0%), which is in very close agreement with the reported efficacy of 80.6% (ref. 32) and suggests good predictive value of the model (Fig. 1a).
Modeling of the duration of immune protection after vaccination
Recent studies have identified a decline in neutralization titer with time for up to 8 months after SARS-CoV-2 infection3,4,5. A major question is whether vaccine-induced responses may be more durable than those measured following infection. Limited studies have analyzed the trajectory of neutralization titer after vaccination34. To compare decay in neutralization titer we fitted a model of exponential decay to equivalent time periods in data from either convalescent3 or messenger RNA vaccination34 cohorts. Comparing neutralization titers measured between 26 and 115 d (the longest time period available for vaccination) after either mRNA-based vaccination34 or symptom onset for post-infection sera3, we found similar half-lives (65 d versus 58 d, respectively; P = 0.88, likelihood ratio test; Extended Data Fig. 2a). Although this comparison relies on limited data, it suggests that decay of vaccine-induced neutralization is similar to that observed after natural SARS-CoV-2 infection.
If the relationship between neutralization level and protection that we observe cross-sectionally between different vaccines is maintained over time, we can use the observed relationship between neutralization and protection to predict how the observed waning of neutralization titers might affect vaccine effectiveness. Important caveats to this modeling are that (1) it assumes that neutralization is a major mechanism of protection (or that the mechanism of protection remains correlated with neutralization over time), although B cell memory and T cell responses may be more durable3,4,5,35 and may play a larger role later after infection or vaccination; (2) it applies the decay of neutralization observed in convalescence to the vaccine data; and (3) it assumes that the decay in titer is the same regardless of the initial starting titer (whereas others have suggested either faster36 or slower37 decay for higher initial levels). These limitations notwithstanding, we analyzed the half-life of neutralization titer using published data from a study of convalescent subjects up to 8 months after infection (using a mixed-effects model with censoring) and estimated that neutralization titer decayed with a half-life of 108 d over this period (Extended Data Fig. 2b)5. We also tried alternative models of decay such as bi-exponential decay (consistent with rapid early decay slowing over time), but found that these did not provide a better fit to the available data. We then used this half-life of 108 d to model the decay of neutralization and protection over the first 250 d after vaccination (Fig. 2a). Our model predicts that even if the waning of neutralization titer over time is the same for different vaccines, this decay will have non-linear effects on the level of protection from SARS-CoV-2 infection, depending on initial vaccine efficacy. For example, a vaccine starting with an initial efficacy of 95% would be expected to maintain 77% efficacy by 250 d. However, a response starting with an initial efficacy of 70% would be predicted to drop to 33% efficacy after 250 d. This analysis can also be used to estimate how long it would take a response of a given initial efficacy to drop to 50% (or 70%) efficacy, which may be useful in predicting the time until boosting is required to maintain a minimum level of efficacy (Fig. 2b). Clearly, data generated from standardized assays are needed to track the long-term decay of post-vaccination immune responses and their relationship to clinical protection. However, this model provides a framework that can be adapted to predict outcomes as further immune and protection data become available. Indeed, if a disconnect between the decay of neutralization titer and protection is observed, this may be a direct pointer to the role of non-neutralizing responses in protection.
Fig. 2: The effects of waning neutralization titer on protection.
a, Prediction of the effects of declining neutralization titer. Assuming that the observed relationship between neutralization level and protection is consistent over time, we estimate the decline in efficacy for vaccines with different levels of initial efficacy. The model assumes a half-life of the neutralization titer of 108 d over the first 250 d (as observed in a convalescent cohort5). b, Modeling of the time for efficacy to drop to 70% (red line) or 50% (blue line) for scenarios with different initial efficacy. For example, for a group starting with an initial protective efficacy of 90%, the model predicts that 70% efficacy will be reached after 201 d and 50% efficacy will not be reached before 250 d. c, Estimation of the impact of viral antigenic variation on vaccine efficacy. In vitro studies have shown that neutralization titers against some SARS-CoV-2 variants are reduced compared with titers against wild-type virus. If the relationship between neutralization and protection remains constant, we can predict the difference in protective efficacy against wild-type and variant viruses from the difference in neutralization level. The dashed line indicates equal protection against wild-type and variant strains. Details of the data and modeling are provided in the Methods.
Full size image
Modeling of the effect of viral variation on immune protection
In addition to the effect of declining neutralization titer over time, reduced neutralization titers and reduced vaccine efficacy to different viral variants have also been observed6,38,39,40,41. For example, it has been reported that the neutralization titer against the B.1.351 variant in vaccinated individuals is between 7.6-fold and 9-fold lower compared with the early Wuhan-related Victoria variant42. Our model predicts that a lower neutralization titer against a variant of concern will have a larger effect on vaccines for which protective efficacy against the wild-type virus was lower (Fig. 2c). For example, a fivefold lower neutralization titer is predicted to reduce efficacy from 95% to 77% in a high efficacy vaccine, but from 70% to 32% for a vaccine with lower initial efficacy.
Estimation of the 50% protective level against severe infection
The analysis above investigates vaccine (and convalescent) protection against symptomatic SARS-CoV-2 infection (using the definitions provided in the different phase 3 and convalescent studies, Supplementary Table 2). However, it is thought that the immune response may provide greater protection from severe infection than from mild infection. To investigate this, we also analyzed data on the observed level of protection from severe infection when these were available. It is important to note that as with symptomatic infection, the definition of severe infection was not consistent across studies (the definitions for each study are detailed in Supplementary Table 3). Given that there have been under 100 severe infections reported across all the phase 3 trials combined, the 95% confidence intervals on the level of protection from severe infection are broad. The neutralization level for 50% protection from severe infection was 3.0% of the mean convalescent level (95% CI = 0.71–13%), which was significantly lower than the 20% level required for protection from any symptomatic infection (P = 0.00039, likelihood ratio test, Supplementary Table 5) (Fig. 3a). An important caveat to this analysis is the implicit assumption that neutralization titer itself confers protection from severe infection. However, it is possible that T cell responses or recall of memory B cell responses may also be important in protection from severe disease43,44,45,46.
” …Our work uses available data on immune responses and protection to model both the protective titer and the long-term behavior of SARS-CoV-2 immunity. It suggests that neutralization titer will be an important predictor of vaccine efficacy in the future as new vaccines emerge. …”
COVID-19 spread in households after camp outbreak
Reuters Health Medical News|July 23, 2021
A new look at how COVID-19 spreads in households has concluded that the disease can easily be passed to parents and extended family members, but social distancing and mask wearing at home can dramatically reduce that spread.
The analysis, published online by the New England Journal of Medicine, may help give doctors a clearer picture of how COVID-19 spreads within family units.
The estimates come from an outbreak at an overnight summer camp in Georgia involving 224 children age 7 to 19, all of whom tested positive for the virus. Eighty eight percent eventually developed symptoms (HRS wonders what were the variables that caused such an unusually high % of symptomatics and 141 fell ill after they returned home from camp.
The team led by Dr. Victoria Chu of the Centers for Disease Control and Prevention in Atlanta: … household contacts usually developed symptoms 5 days after the child felt sick.
Ten percent of the adults who became sick ended up in the hospital, typically for 5 to 11 days: 72% of adults were tested.
Compared with the child’s siblings, parents were 2.3 times more likely to test positive for COVID-19 and extended family members, particularly grandparents, had an infection rate 6.6 times higher.
Sharing meals was 3.9 times riskier, being within six feet of the infected child for 15 minutes or more upped the risk 5.4-fold, being face-to-face with the child was 4.6 times more hazardous and persons having direct physical contact with the sick child was 3.3 times more.
Symptomatic children were more than 5 times as likely to spread the disease to a household member than if they were not symptomatic.
The chance of spread was reduced by 60% if the child practiced social distancing at home and by 50% if the child wore a mask while around other people.
“Characteristics of, and Important Lessons From, the Coronavirus Disease 2019 (CoV2-19) Outbreak in China: Summary of a Report of 72,314 Cases From the Chinese Center for Disease Control and Prevention. By Zunyou Wu, MD, PhD. The Chinese Center for Disease Control and Prevention recently published the largest case series to date of coronavirus disease 2019 (COVID-19) in mainland China (72 314 cases, updated through February 11, 2020).1This Viewpoint summarizes key findings from this report and discusses emerging understanding of and lessons from the COVID-19 epidemic. Epidemiologic Characteristics of the COVID-19 Outbreak.
Most case patients were 30 to 79 years of age (87%), 1% were aged 9 years or younger, 1% were aged10 to 19 years, and 3% were age 80 years or older. Most cases were diagnosed in Hubei Province (75%)and most reported Wuhan-related exposures (86%;ie, Wuhan resident or visitor or close contact with Wuhan resident or visitor). Most cases were classified as mild (81%; i.e., non-pneumonia and mild pneumonia). However, 14% were severe (i.e., dyspnea, respiratoryfrequency30/min, blood oxygen saturation93%, partial pressure of arterial oxygen to fraction of inspired oxygen ratio <300, and/or lung infiltrates >50% within 24 to 48 hours), and 5% were critical (ie, respiratory failure, septic shock, and/or multiple organ dysfunction or failure) (Box).1The overall case-fatality rate (CFR) was 2.3% (1023 deaths among 44 672 confirmed cases). No deaths occurred in the group aged 9 years and younger, but cases in those aged 70 to 79 years had an 8.0% CFR and cases in those aged 80 years and older had a 14.8% CFR. No deaths were reported among mild and severe cases (HRS FINDS THE LATTER STATEMENT A BIT UNBELIEVALBE OR IS A TYPING ERROR FOR “NON-SEVERE”). The CFR was 49.0% among critical cases. CFR was elevated among those with preexisting comorbid conditions—10.5% for cardiovascular disease, 7.3% for diabetes (” … From the NIH F Collins 6/8/21: beta cells and other cell types in the pancreas express the ACE2 receptor protein, the TMPRSS2 enzyme protein, and neuropilin 1 (NRP1), all of which SARS-CoV-2 depends upon to enter and infect human cells …”), 6.3% for chronic respiratory disease, 6.0% for hypertension, 5.6 % for cancer & 3.8% for health care workers.
5/29/20 A S Zubair JAMA Neurol “Currently, there are 7 Corona viruses that can infect humans, including human coronavirus (HCoV)–229E, HCoV-NL63, HCoV-HKU1, HCoV-OC43, MERS-CoV, SARS-CoV-1, and SARS-CoV-2.8 Beta coronaviruses SARS-CoV-2, SARS-CoV-1, and MERS-CoV are associated with severe disease in humans.1,3,8 Although HCoV are typically associated with respiratory tract disease, 3 HCoV have been shown to infect neurons: HCoV-229E, HCoV-OC43, and SARS-CoV-1.”
Medical Xpress 7/14/20: “An estimated 60% of known infectious diseases and 75% of all new, emerging, or re-emerging diseases in humans have animal origins. SARS-CoV-2 is the newest of seven coronaviruses found in humans, all of which came from animals, either from bats, mice or domestic animals. Bats were also the source of the viruses causing Ebola, rabies, Nipah and Hendra virus infections, Marburg virus disease, and strains of Influenza A virus.”
The fascinating sequence of VIRAL ESCAPE resulting in HUMAN INFECTION is that corona virus infected bat meat whether from the research labs in Wuhan, China, or just wild living bats may have gone through intermediate hosts such as snakes, Malayan/Sunda pangolins (scaly anteaters), and now dogs are also recognized as a possible vector. Ed Yong in the 4/29/20 The Atlantic (magazine) “…scientists have also identified about 500 other corona viruses among China’s many bat species. There will be many more—I think it’s safe to say tens of thousands,” said Peter Daszak of the EcoHealth Alliance, who has led that work. Laboratory experiments show that some of these new viruses could potentially infect humans. SARS-CoV-2 likely came from a bat, too.
It seems unlikely that a random bat virus should somehow jump into a susceptible human. But when you consider millions of people, in regular contact with millions of bats, which carry tens of thousands of new viruses, vanishingly improbable events become probable ones. In 2015, Daszak’s team found that 3 percent of people from four Chinese villages that are close to bat caves had antibodies that indicated a previous encounter with SARS-like coronaviruses…”
N Zhu NEJM 2020;382:727- Phylogenetic analysis of 2019-nCoV, the Severe Acute Respiratory Syndrome Corona Virus 2 (SARS-CoV-2), indicated that it is different but related to SARS-CoV-1 (∼ 80% nucleotide identity) that appeared during the autumn of 2002 in the province of Guangdong, China, spreading itself into 29 countries infecting 8,422 and killing 916 individuals: M Murakami at Hokkaido University’s Institute for Genetic Medicine and T Hirano from the National Institutes for Quantum and Radiological Science and Technology, reviewed two recent studies by Zhou et al and Hoffmann. SARS-CoV-2 enters human cells by attaching to a cell protein/enzyme called ACE2 utilizing a human enzyme called TMPRSS2 (Type II transmembrane serine protease = TMPRSS2 is crucial for its gaining entry). SARS-CoV-2 is known to be engulfed into the human cell along with the ACE2 receptor & neuropilin-1 it had combined with. “This reduces the number of ACE2 receptors on cells, leading to an increase of a polypeptide, called angiotensin II, in the blood,” says Murakami, “ACE2 is expressed in airway epithelia, kidney cells, small intestine, lung parenchyma, and vascular endothelia throughout the body and widely throughout the CNS”: JAMA Neurol 5/29/20. Angiotensin II triggers an inflammatory pathway involving NF-κB and IL-6-STAT3 particularly in nonimmune cells including endothelial cells and epithelial cells.” This pathway forms a positive feedback cycle, named IL-6 amplifier, resulting in its excessive activation and therefore the cytokine storm and ARDS,” says Hirano, a pioneer in IL-6 research. “Targeting these pathways, such as with the anti-IL-6 receptor antibody called tocilizumab, could disrupt this life-threatening inflammatory reaction in COVID-19 patients,” Hirano added. International J of Inf Dis 8/6/20: N Potere “Disease progression was experienced by none of the (low dose) tocilizumb-treated patients vs by 5 (50%) patients in the standard of care group. Overall, experts concluded that in hospitalized patients with moderate COVID-19 and hyperinflammation” tocilizumab would be helpful.
https://www.vumedi.com/video/sars-cov-2-infection-after-vaccination-how-common-is-it-can-vaccine-efficacy-data-be-maintained-outs/?token=afa3f3b9-4e66-4953-b2d9-1c4ef36b78d9&utm_source=COVID%20Interests%20Criteria_81037&utm_medium=Video&utm_campaign=%28COVID%29%20How%20Common%20Is%20SARS-CoV-2%20Infection%20After%20Vaccination%3F&utm_content=SARS-CoV-2%20Infection%20After%20Vaccination%3A%20How%20Common%20Is%20It%3F%20Can%20Vaccine%20Efficacy%20Data%20Be%20Maintained%20Outside%20the%20Trial%20Setting%3F&utm_term=COVID%20Vaccines&link_data=eyJidWxrX21haWxfYWN0aW9uIjoiYyIsInJlY2lwaWVudF9pZCI6MTIyMTQ2OTAyMCwibWFpbF9pZCI6ODEwMzd9%3A1lagQJ%3AWNTn57yazdeFHJYjPtT3qCU_j3A&mail_id=81037
ROCHE’s Actemra (tocilizumab) – 5/31/21 A research article, published in JAMA, found this arthritis drug, an anti-IL-6, reduced the risk of death in patients with moderate-tosevere Covid-19-associated pneumonia who also had a high CRP level 90 days after treatment. At Day 14, 18% of tocilizumab patients with a CRP>15 mg/dL died or required ventilation vs. 57% of placebo patients.
Angiotensin Converting Enzyme 2: A Double-Edged Sword
Kaiming Wang, BSc; Mahmoud Gheblawi, BSc; Gavin Y. Oudit, MD, PhD, FRCP(C)
Circulation. 2020;142(5):426-428.
Angiotensin converting enzyme 2 (ACE2) has garnered much attention given the current coronavirus disease 2019 (COVID-19) pandemic as the cellular receptor for severe acute respiratory syndrome coronavirus-2 (SARS–CoV-2). ACE2 was discovered 20 years ago based on approaches searching for ACE homologues and was initially cloned from human heart failure ventricular and lymphoma cDNA libraries.[1] Since then, 2 major functions have been identified for ACE2: (1) an endogenous counter-regulator of the renin-angiotensin system (RAS), and (2) a cellular receptor for SARS-CoV and SARS–CoV-2 viruses.
ACE2 is ubiquitously expressed with highest levels detected in the cardiovascular system, gut, kidneys, and lungs. In the cardiovascular system, ACE2 is expressed in cardiomyocytes, epicardial adipose tissue, cardiac fibroblasts, vascular smooth muscle, and endothelial cells.[1,2] ACE2 is a type I transmembrane protein that functions as a monocarboxypeptidase with a catalytically active ectodomain exposed to the circulation that hydrolyzes various peptides, including angiotensin II and angiotensin I, generating angiotensin 1–7 and angiotensin 1–9, respectively.[1] A soluble form of ACE2 can be released from the membrane through proteolytic cleavage mediated by ADAM17 (ADAM metallopeptidase domain 17) resulting in loss of ACE2 protection against tissue RAS and increased plasma ACE2 activity, a known marker of adverse prognosis in patients with cardiovascular disease.
The discovery of ACE2 introduced an alternative protective arm, ACE2/angiotensin 1–7/Mas receptor axis, to counterbalance the more renowned pathogenic ACE/angiotensin II/angiotensin II receptor type 1 (AT1) receptor axis that predominates in disease states as a result of RAS overactivation (Figure A). Cleavage of angiotensin I by ACE generates angiotensin II, which is the primary effector peptide of the ACE/angiotensin II/AT1 receptor axis, triggering potent vasoconstriction, inflammation, cell proliferation, hypertrophy, fibrosis, and tissue remodeling. ACE2 cleaves angiotensin II into the cardioprotective angiotensin 1–7, which acts through Mas receptors to counterbalance the detrimental effects of angiotensin II signaling. Therefore, ACE2 protects against RAS-induced injuries through 2 processes: (1) degrading angiotensin I and angiotensin II to limit substrate availability in the adverse ACE/angiotensin II/AT1 receptor axis, and (2) generating angiotensin 1–7 to increase substrate availability in the protective ACE2/angiotensin 1–7/Mas receptor axis.
(Enlarge Image)
Figure.
Role of angiotensin converting enzyme 2 (ACE2) in the renin-angiotensin system (RAS) and proposed mechanism for severe acute respiratory syndrome coronavirus-2 (SARS–CoV-2)–induced downregulation of cell surface ACE2 expression.
A, ACE2 balances the 2 axes of the RAS, increased ACE2 promotes the protective ACE2/angiotensin 1–7 (Ang 1–7)/Mas receptor axis (MASR), and loss of ACE2 results in a shift towards diseased states characterized by overactivity in the ACE/angiotensin II (Ang II)/Ang II receptor type 1 (AT1) receptor axis (AT1R). B, Viral spike glycoprotein of SARS–CoV-2 interacts with cell surface ACE2 and becomes internalized together through endocytosis, resulting in decreased surface ACE2 expression. The endocytic event upregulates ADAM17 (ADAM metallopeptidase domain 17) activity, which cleaves ACE2 from the cell membrane, perpetuating the loss of ACE2 from tissue RAS. Loss of ACE2 leads to accumulation of Ang II which, through AT1 receptors, also upregulates ADAM17, resulting in further cleavage of cell surface ACE2. Soluble recombinant human ACE2 (rhACE2) is a promising therapeutic for SARS–CoV-2 through its ability to (1) sequester viral particles to prevent their interaction and subsequent entry through cell surface ACE2 and (2) limit activities of angiotensin II and increase levels of protective angiotensin 1–7. Ang I indicates angiotensin I.
Loss-of-function experiments using ACE2 knockout mice and ACE2 inhibitors have revealed increased susceptibility to myocardial infarction, hypertension, and angiotensin II–induced myocardial hypertrophy, microvascular complications, inflammation, fibrosis, diastolic and systolic dysfunction, and oxidative stress.[1,2] Importantly, partial loss of ACE2, as seen in human hearts explanted from patients with heart failure and dilated cardiomyopathy, is sufficient to enhance the susceptibility to heart disease.[1] Conversely, gain-of-function experiments with recombinant ACE2, overexpression of ACE2, and supplemental angiotensin 1–7 have shown protective roles in various models of cardiovascular disease including hypertension, diabetes mellitus, and heart failure with preserved ejection fraction.[1,2] Pharmacological antagonists of the RAS, such as ACE inhibitors (reduce hypertension-induced immune cell activation Nature Biotechnology (2020). DOI: 10.1038/s41587-020-00796-1) and angiotensin II receptor blockers, protect the cardiovascular system partly by increasing ACE2 levels in disease states. Clinical trials with intravenous infusion of recombinant human ACE2 in patients with pulmonary arterial hypertension and acute lung injury reported immediate decreases in plasma angiotensin II/angiotensin 1–7 ratios, reflecting ACE2 functions and its therapeutic effects.
Binding and entry of both SARS-CoV and SARS–CoV-2 into human cells is facilitated by the interaction between receptor-binding domain of the S1 subunit on viral spike glycoproteins with the ectodomain of ACE2.[3] Endocytosis of ACE2 alongside viral particles into endosomes reduces surface ACE2 expression which represents an initial insult toward ACE2-mediated tissue protection. Of particular concern are the positive feedback pathways in place to facilitate further downregulation of ACE2 expression after the initial endocytotic event, perpetuating tissue damage and imbalance of the tissue RAS from SARS–CoV-2 infections (Figure B). Viral entry is also facilitated by ADAM17 activity, which is upregulated by SARS-CoV, a process dependent on the ACE2 cytoplasmic domain. Upregulation in ADAM17 protease activity perpetuates loss of ACE2 from the cell surface, resulting in a shift away from the protective ACE2/angiotensin 1–7/Mas receptor axis towards the disease state and accumulation in angiotensin II. Angiotensin II further upregulates ADAM17 activity in a well-characterized positive feedback loop leading to the shedding of its regulator, ACE2, through the AT1 receptors and downstream extracellular signal-related kinase/p38 mitogen-activated protein kinase signaling pathways as a sequela to SARS–CoV-2 receptor binding. Furthermore, ADAM17 also mediates the liberation of membrane bound precursors of tumor necrosis factor α, interferon γ, and interleukin 4 proinflammatory cytokines into the circulation, giving rise to its alternative name, tumor necrosis factor converting enzyme (TACE). These cytokines, namely interleukin 4 and interferon γ, downregulate cell surface expression of ACE2, and reduce ACE2 mRNA levels leading to another pathway for ACE2 loss from SARS–CoV-2–induced systemic and tissue inflammation. (The NIH reported that autoantibodies against interferons are associated with severe Covid-19. P Bastard et al. Auto-antibodies against type I IFNs in patients with life-threatening COVID-19. Science DOI: 10.1126/science.abd4585 (2020). Those autoantibodies are more common in men.)
In lung injury, deregulation of RAS through downregulation of ACE2 increases vascular permeability, pulmonary edema, and severity of injury in SARS-CoV infections though actions of angiotensin II that are attenuated by AT1 receptor blockade. In postmortem autopsy samples of heart tissue from patients who succumb to SARS, increased myocardial fibrosis, inflammation, and reduced myocardial ACE2 expression have been reported, along with detectable viral SARS-CoV genome, providing suggestive evidence for myocardial injury from SARS-CoV.[4] Despite the predominance of respiratory symptoms, acute cardiac and kidney injuries, myocarditis, arrhythmias, and gut and liver abnormalities occurs in COVID-19 patients,[5] consistent with the widespread expression of ACE2. The loss of ACE2-mediated protection from the cardiovascular systems after SARS–CoV-2 infection could contribute to the cardiovascular events observed in COVID-19 patients.[5]
Recombinant human ACE2 has entered into clinical trial in a cohort of 24 patients in China. Systemic delivery of recombinant human ACE2 (0.4 mg/kg intravenous twice a day for 7 days) will hopefully sequester viral SARS–CoV-2 particles in the circulation, preventing their interaction and subsequent internalization through endogenous ACE2 receptors while also activating the systemic protective axis of the RAS.
In summary, the bifunctional role of ACE2 as a double-edged sword turns off the RAS system and leads to beneficial effects but also mediates unique susceptibility to lung and cardiovascular disease in COVID-19 patients by serving as the SARS–CoV-2 receptor. The ACE2 double-edged sword can be carefully wielded to provide potential novel therapeutics for cardiovascular disease but also for COVID-19. Moreover, the long-term sequelae of COVID-19 survivors and their possible increased risk for lung and cardiovascular disease requires careful monitoring and follow-up informed by knowledge of ACE2 biology.
https://jamanetwork.com/journals/jama/fullarticle/2770758?guestAccessKey=30d84bfd-6f0a-4f92-81e9-f606284668b4&utm_source=silverchair&utm_medium=email&utm_campaign=article_alert-jama&utm_content=olf&utm_term=091120
Three peptides could be causing severe COVID-19 complications
by Texas Tech University Health Sciences Center5/5/21
Credit: CC0 Public Domain
A study published in July 2020 hypothesized a link between the presence of bradykinin, a well-known peptide, and severe cases of COVID-19. Vardan Karamyan, Ph.D., an associate professor and vice chair for the Texas Tech University Health Sciences Center (TTUHSC) Jerry H. Hodge School of Pharmacy Department of Pharmaceutical Sciences, had not previously conducted or evaluated any research related to COVID-19. However, he found the article intriguing because it discussed bradykinin, one of three specific peptides with which his lab has much well-published experience.
The paper received a lot of attention in both the media and scientific literature, but as Karamyan read through it, he felt it failed to address an equally important part of a bigger picture: the likely involvement of two other bioactive peptides, known as substance P and neurotensin, in the same processes and mechanisms proposed by the authors.
“I decided to write a focused review article on the topic and looked for journals which announced special issues devoted to COVID-19,” Karamyan recalled.
Karamyan learned that the American Physiological Society (APS) had two journals with ongoing special issues focusing on COVID-19. Because APS has a membership base of highly accomplished scientists and a wide audience, Karamyan submitted the manuscript of his focused review to Physiological Reports, one of APS’s peer-reviewed journals that published Karamyan’s review in its March 2021 issue.
In the original article, which included the author’s data and additional information from other research, Karamyan saw that the researchers had postulated that bradykinin was the peptide at the center of many of the complications experienced by patients with severe COVID-19 cases. Peptides are short chains composed of at least two and no more than 50 amino acids, which also are the building blocks of proteins.
“What they were saying makes sense, and it is really beautifully done; they branch out quite a bit and they put things together in a very systematic way,” Karamyan explained. “But what was very surprising is that if their proposed mechanism was true, then two other peptides (substance P and neurotensin) should also be next to bradykinin; yet there was not even a single word about those two peptides.”
Karamyan said it was obvious the original study arrived at some important findings, but he also knew that a basic tenant of science says the better any disease is understood, the better the chances of curing or managing it.
“That was their motivation too,” Karamyan asserted. “Their study tells us, ‘Look, maybe this is what is happening,’ and that made me think that I really need to try to bring awareness that there are these two other peptides that could be equally important.”
Almost since the pandemic began, research has shown that many of the worst cases of COVID-19 are related to cytokine storms, which is when the body overproduces cytokines in an effort to fight the virus. Cytokines are substances such as proteins, peptides or glycoproteins that are released by cells within the immune system to affect other cells. For instance, cytokines help the body manage conditions such as immunity, inflammation and hematopoiesis (the production of blood cells and platelets). When they are overproduced, however, the resulting cytokine storm can have deleterious effects.
“In the beginning, it’s a compensatory reaction when the cytokines are produced, but when it is overdone, this makes things much worse,” Karamyan added.
Using their own data and that put forward by previous investigators, the researchers who authored the original paper concluded it was the overexpression of bradykinin, or a bradykinin storm rather than a cytokine storm that was adversely affecting COVID-19 patients.
“As I noted, it makes sense,” Karamyan said. “It’s not that cytokines don’t do anything, but I agree with them that bradykinin could make things worse.”
To make their case, the authors of the original paper showed there were mechanisms that would make bradykinin appear to be upregulated, or increased within the body, while other mechanisms meant to degrade bradykinin are downregulated, or decreased. Karamyan said those same mechanisms also would affect substance P and neurotensin in the same manner at least 80% of the time.
“Those same mechanisms, or the majority of them, would lead to elevated levels of not just bradykinin, but also substance P and neurotensin,” Karamyan said. “That’s one fact; the second is that if this inflammation is going on, if things are deteriorating in a COVID-19 patient, in the same way that bradykinin is making things worse, these peptides are known to do the same thing, and the number one thing is where the vasculature becomes leaky and we have edema formation and so on.”
Karamyan said the third fact he pointed out in his review is from his own collaborative and preliminary study with TTUHSC colleague Abraham Al-Ahmad, Ph.D., where they showed that these three peptides together—bradykinin, substance P and neurotensin—make more profound changes in vascular permeability than each of them produce alone. Vascular permeability refers to a blood vessel wall’s capacity to allow other molecules and cells to pass through.
“When they’re together, bradykinin, substance P and neurotensin kind of have this synergetic or combined effect,” Karamyan said.
If those three peptides are concurrently available, Karamyan continued, they could adversely affect COVID-19 patients and lead to some of the conditions the authors mentioned such as vascular permeability, oxidative stress, inflammation, edema and others. He coined the term “vasoactive peptide storm” to describe the process.
“It’s a made-up term, yes, but all three are vasoactive peptides that would do similar things,” Karamyan said. “That’s why I suggested a vasoactive peptide storm.”
In the original paper, the authors proposed using drugs known as receptor blockers that can prevent or reduce the impact of bradykinin. Karamyan agreed, but suggested adding receptor blockers for substance P and neurotensin to yield better results. In fact, Karamyan’s lab may already have an enzyme that can inactivate all three, though he said the approach is speculative at this point.
“An enzyme that we are working with called neurolysin actually degrades all three peptides,” Karamyan said. “It inactivates bradykinin, neurotensin and substance P. An alternative approach—again highly speculative—could perhaps be instead of using three different drugs to block all three peptides, possibly using one enzyme to get rid of all three peptides.”
Whether Karamyan decides to investigate the significance and impact of the so-called vasoactive peptide storm, or leave it for others more accustomed to working with COVID-19, Karamyan believes he may have found another piece to the pandemic puzzle. He and several colleagues have discussed testing the ability of neurolysin to deactivate all three peptides.
“These reviewers were telling me this is something that makes sense and is worthy of trying experimentally,” Karamyan said. “Obviously, it was very pleasant to hear this and it gave me confidence. Since then, I’m kind of on the border. It would be some deviation from what I do in regards to the disease itself. In regards to the mechanisms, no, because this is my field: peptides and enzymes. As of now I don’t have anything planned yet, but it is possible that I may, or we may collectively as a group look into it.”
More information: Vardan T. Karamyan, Between two storms, vasoactive peptides or bradykinin underlie severity of COVID‐19?, Physiological Reports (2021). DOI: 10.14814/phy2.14796
COVID-19 and the Path to Immunity
David S. Stephens, MD1; M. Juliana McElrath, MD, PhD2 9/11/20
The emergence of adaptive immunity in response to the novel Betacoronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), occurs within the first 7 to 10 days of infection.1-3
A robust memory B-cell and plasmablast expansion is detected early in infection,2,4 with secretion of serum IgM and IgA antibodies by day 5 to 7 and IgG by day 7 to 10 from the onset of symptoms. In general, serum IgM and IgA titers decline after approximately 28 days (Figure), and IgG titers peak at approximately 49 days. Simultaneously, SARS-CoV-2 (CoV2-19) activates T cells in the first week of infection, and virus-specific memory CD4+ cells and CD8+ T cells reportedly peak within 2 weeks but remain detectable at lower levels for 100 or more days of observation. Grifoni et al1 and others5,6 have identified CoV2-19–specific memory CD4+ T cells in up to 100% and CD8+ T cells in approximately 70% of patients recovering from COv2-19. Although severe CoV2-19 is characterized by high-viral titers, dysregulated innate inflammatory cytokine and chemokine responses and prolonged lymphopenia, antibody-dependent enhancement or dominant CD4+ TH2-type cytokines (eg, IL-4, IL-5, IL-13) do not appear to contribute to acute COVID-19 severity.
Generalized model of T-cell and B-cell (plasmablast, antibody) responses to Co V2-19 infection projected over 1 year following infection. Neutralizing antibodies, memory B cells, and CD4+ and CD8+ memory T cells to CoV2-19, which are generated by infection, vaccination, or after re-exposure, are key to the path to immunity. The dotted lines represent peak B-cell, T-cell, and antibody responses following infection.
The magnitude of the anti–CoV2-19 IgG and IgA titers to the spike protein correlates in convalescing patients with CD4+ T-cell responses1; and the magnitude of IgG1 and IgG3 RBD enzyme-linked immunosorbent assay (ELISA) titers correlates strongly with viral neutralization.2,3
The generation of neutralizing antibodies directed at the spike protein is a basis of multiple human vaccines in clinical trials7 to counteract CoV2-19, and virus neutralization is the basis of potential therapeutic and preventive monoclonal antibodies also currently in human clinical trials. Such virus neutralizing antibodies are protective in animal models of SARS-CoV-2 infection, at least in the short Term of several months. Potent neutralizing antibodies and TH1-biased CD4+ T-cell responses to the spike protein protect against CoV2-19 infection in the lungs and nasal mucosa of nonhuman primates without evidence of immunopathological changes.8 The RBD region of the CoV2-19 spike protein shows little sequence homology to the seasonal coronaviruses.2 Although variants in the CoV2-19 spike (S) protein (e.g., D614G) may be a concern, CoV2-19 such variants have not been shown to reduce the recognition of RBD epitopes important for antibody neutralization. 10/2020: an entirely NEW approach that generates antibodies and T-cell activation is that of Dr. Patrick Soon-Shiong’s Immunity Bio.
Recent reports have demonstrated a decline in IgG neutralizing antibodies to SARS-CoV-2 in convalescence. Antibody levels always decline after the acute phase of infection because most of the plasmablasts, the “effector” response of B cells, induced during the first weeks after infection are short-lived. A similar pattern is seen with the effector CD8+ T-cell response. After this reduction, serological memory is maintained by the smaller number of long-lived plasma cells that reside in the bone marrow and constitutively secrete antibody in the absence of antigen. The antibody recall response comes from this pool of memory B cells that are also long-lived. In fact, rare circulating memory cells have been shown to produce highly potent neutralizing antibodies when serum neutralizing titers are low.3 Thus, an early decline of neutralizing antibody levels should not be of concern.
Following experimental re-challenge with the same HCoV 229E strain at 1 year, no individuals who had been previously infected developed a cold and all had a shorter duration of detectable virus shedding. Thus, at least strain-specific immunity to clinical coronavirus disease may be preserved despite rapid waning of antibodies. In nonhuman primates, CoV2-19 infection protects against reinfection.10 (7 months of immunity duration noted in IgG antibody positive patients as published by S F Lumley in the NEJM 12/2020 Memory B-Cells last at least 8 months as published by M van Zelm in Science Immunology 2020).
CoV2-19–specific CD4+ and CD8+ memory T cells are also generated in asymptomatic to severe disease,1,5,6 which exhibit cytotoxic activities and express antiviral cytokines, features that may control viral replication and prevent recurrent severe infections. Moreover, investigations have focused on circulating T-cell responses in acute COVID-19, often during periods of marked lymphopenia4.
Substantial data now demonstrate the presence of preexisting T-cell immunity to CoV2-19 in blood donors either prior to the CoV2-19 pandemic or more recently among those without infection.1,5,6 Memory CD4+ T cells are found in higher frequencies than are CD8+ T cells, and these likely represent responses induced by previous infection with other human endemic beta-coronaviruses known to cause the common cold. Such T cells can recognize known or predicted epitopes1 within the nucleocapsid (N protein) and spike structural proteins as well as the nonstructural proteins (NSPs), NSP7 and NSP13.5 SARS-CoV-2 reactive T cells are also seen in household contacts of patients infected with CoV2-19.
Seroprevalence data (antibodies to the CoV2-19 spike protein) estimate that there may be 10 times more SARS-CoV-2 infections than the number of reported cases. Thus, it is possible that 40 million to 50 million (12% to 15% of the US population) to date may have been infected with a detectable serological response to SARS-CoV-2.
Antibodies Fade Faster After Vaccine vs Actual Infection
By Reuters Staff 8/24/21
Protective antibody levels decline faster in recipients of the mRNA COVID-19 vaccine from Pfizer/BioNTech than in COVID-19 survivors, according to doctors at one of Israel’s largest HMOs. The research team tracked antibody levels in 2,653 adults who received two doses of the vaccine and in 4,361 COVID-19 survivors who were never vaccinated. Antibody levels fell by up to 40% per month in vaccinated participants, versus less than 5% per month in so-called convalescents.
After six months, about 84% of vaccine recipients still had detectable antibodies, whereas roughly 90% of convalescents still had detectable antibodies after nine months. Dr. Ariel Israel of Leumit Health Services, coauthor of a reported posted on medRxiv ahead of peer review, noted that antibodies are not the immune system’s only weapon against the virus. Still, he said, the data suggests that antibody protection in Pfizer vaccine recipients wanes at a higher rate than in COVID-19 survivors.
Leumit researchers had previously reported that breakthrough infection rates increase starting about five months after vaccination. Dr. Israel said the combined data argues for a booster shot five months after the second injection, especially for high-risk individuals.
SOURCE: https://bit.ly/2XP2Dwi medRxiv, online August 22, 2021.
Experts push back against rush for COVID booster shots
by Dennis Thompson
(HealthDay)—Opposition is mounting among U.S. and international health experts against President Joe Biden’s push to make COVID-19 booster shots available later this month.
The scientific evidence simply isn’t there to support booster shots, and those doses would be better used in the arms of the unvaccinated around the world to prevent future mutations of COVID, infectious disease experts said in an interview with HealthDay Now.
“The important thing to remember is this is being driven by the unvaccinated. That’s what’s in the hospital right now. It’s not vaccinated people that are the issue in this pandemic,” said Dr. Amesh Adalja, a senior scholar with the Johns Hopkins Center for Health Security. “The whole debate over boosters needs to be framed by that, because putting third doses into highly vaccinated populations isn’t going to change what’s happening in the United States.”
Unfortunately, the Biden Administration’s advocacy for booster shots has created a rush by some Americans to get a third dose—even though the additional jabs haven’t yet been approved by federal regulators, said Dr. Camille Kotton, an infectious disease specialist with Massachusetts General Hospital in Boston.
“I will say that I’m shocked at the number of people that have gone out and just helped themselves to booster doses,” Kotton said. “It’s a little bit greedy to do something that’s not really recommended yet, just because they’ve read the newspaper and thought that was a good idea for themselves.”
Last month, Biden told the nation that booster shots would be available the week of Sept. 20 for the earliest recipients of the COVID-19 vaccines, although he added that the plan was pending approval from the U.S. Food and Drug Administration and the U.S. Centers for Disease Control and Prevention.
The FDA will hold an advisory board meeting on Sept. 17 to review data from Pfizer regarding booster shots for its vaccine, the agency announced this week.
WHO, EU don’t back booster shots
Both the World Health Organization and the European Union have counseled against booster shots, urging that nations instead focus on getting shots into the arms of the unvaccinated. That’s the approach Adalja and some other health experts are promoting.
“The longer this virus is unchecked anywhere in the world, the more likely we are to see variants. Until we control this pandemic in all corners of the globe, we’re still going to have variants,” Adalja said. “The way we control this pandemic is by getting first doses, second doses into people’s arms all around the world, even in the United States.”
Kotton and Adalja both noted that at this point, the scientific evidence doesn’t seem to support the need for booster shots in fully vaccinated people.
“Really what we’re seeing is higher rates of breakthrough disease, which is still generally mild symptoms,” said Kotton, who serves on the CDC’s Advisory Committee on Immunization Practices (ACIP), the board that will review booster shots prior to their approval. “People aren’t going to the hospital the vast majority of the time. They just have a little bit of a viral syndrome.”
ACIP recently approved a third dose of vaccine for people with compromised immune systems, but Kotton noted that the dose isn’t a booster. Rather, people who are immune-compromised will receive a three-dose vaccine series rather than a two-dose series.
“This is not actually a booster dose,” Kotton said. “This is a third dose that is considered part of their primary vaccine series.”
In that case, medical evidence showed that people with immune system deficiencies—solid organ recipients, bone marrow transplant patients, folks fighting cancer—needed a three-dose series to achieve sufficient protection against COVID, Kotton said.
But for the general population, the evidence continues to show that the vaccines protect people against severe illness and hospitalization, Kotton and Adalja said.
“Primarily we’ve been trying to prevent this virus from causing severe disease, hospitalization and death,” Adalja said. “By that standard, the vaccines—even when you see antibody levels wane or you see breakthrough infections occurring—they’re still performing off the charts, because that’s what they were designed to do.”
Let scientists make the call
“Vaccines are not bug zappers. They’re not force fields. They’re not meant to stop every breakthrough infection,” Adalja continued. “Because the breakthrough infections are generally mild, I don’t know that we want to be in the business of chasing them with booster shots when this is not a virus that’s ever going to go away.”
Adalja said he’s “very eager” to see the ACIP debate the data and discuss when healthy people might need a booster.
“This is something we want to be proactive about. We want to have a plan in case it’s necessary,” Adalja said. “But I think giving a date certain that [is] six months or eight months or Sept. 20, that doesn’t really strike me as something that’s evidence-based. I think that’s why you see many infectious disease doctors question the clinical need for these boosters.
“These types of decisions shouldn’t be announced by the White House,” Adalja noted. “They should be announced by the ACIP and the CDC. If we reverse this process, we kind of go back to the old days of the pandemic where you had politicians making decisions about what treatments were effective or were not effective.”
In the meantime, Kotton cautions people not to seek out a booster until it’s been formally approved. She noted that doctors or pharmacists could wind up in trouble for providing a booster, since it’s “a significant violation of federal policy,” Kotton said.
In addition, people might be on the hook for the cost of their booster dose, since it hasn’t been approved. “I would really not recommend that people do that of their own accord until such time as we have good scientific evidence to support that,” Kotton said.
More information: The U.S. Centers for Disease Control and Prevention has more on COVID-19 vaccines.
Neutralizing antibodies to SARS-CoV-2 remain high for up to 13 months following infection: Study
By Denise Baez 8/6/21
Neutralizing titres against the spike protein of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) remained relatively high for up to 13 months post infection, with only modest declines in neutralizing titres, according to a study published in the Journal of General Internal Medicine.
Patients with more severe disease showed higher rates of positivity over time suggesting a more robust antibody response, reported Juan P. Wisnivesky, MD, Icahn School of Medicine at Mount Sinai, New York, New York, and colleagues.
“Understanding immunological memory post-COVID-19 [coronavirus disease 2019] can provide insights into the risk for reinfection and the potential durability of vaccines,” the authors wrote. “While early reports suggested that antibody titres may rapidly decrease, other studies found high antibody titres several months after acute COVID-19 infection. However, many of these studies included relatively small samples and reported overall rates of positivity at titres that may not confer neutralising activity. We extend these results by showing persistent rates of a positive antibody response — at potentially neutralizing titres — for up to 13 months after acute infection.”
The study cohort consisted of 620 adults (mean age, 49 years; 64% females; 53% white; 20% Latinx) who had recovered from COVID-19 and were enrolled in a prospective research registry at the Mount Sinai Health System between July 20, 2020, and April 13, 2021. All participants had laboratory-confirmed COVID-19 infection and had not received COVID-19 vaccination before enrolment or during the follow-up period. The participants contributed a total of 1,195 antibody tests.
The researchers used an enzyme-linked immunosorbent assay based on the stabilised full-length spike protein to assess SARS-CoV-2 antibodies from at least 30 days to up to 13 months after acute infection. A titer ≥1:320 (≥16 AU/mL) has been correlated with ≥90% neutralising activity in a micro-neutralisation assay and has been used as a criterion for convalescent plasma donation.
Overall, 90% of participants had positive antibody tests 6 months post-infection, and 83% had positive antibody tests 13 months post-COVID-19. Antibody titers were relatively stable over 13 months post infection.
The adjusted probability of having a positive antibody test at 13 months was 71% for outpatients, 75% for patients who presented to the emergency department, and 84% for patients who required hospitalization.
“Rates of positive antibody tests against SARS-CoV-2 remained high up to 13 months post-COVID-19,” the authors wrote. “Patients treated in the hospital had significantly higher rates of positive results compared with those who were managed in the outpatient setting. There were no significant differences among patients treated in the emergency department or managed as outpatients for acute COVID-19.”
The researchers noted that they did not have information on severity of outpatient illness, therefore, they were not able to compare the immune response in asymptomatic patients compared with patients with mild or moderate COVID-19.
Reference: https://link.springer.com/article/10.1007/s11606-021-07057-0
Protective immunity after recovery from SARS-CoV-2 infection
• Noah Kojima
• Jeffrey D Klausner
Published:November 08, 2021DOI:https://doi.org/10.1016/S1473-3099(21)00676-9
Previous ArticleClinical outcomes and bacterial characteristics of …
The SARS-CoV-2 pandemic is now better controlled in settings with access to fast and reliable testing and highly effective vaccination rollouts. Several studies have found that people who recovered from COVID-19 and tested seropositive for anti-SARS-CoV-2 antibodies have low rates of SARS-CoV-2 reinfection. There are still looming questions surrounding the strength and duration of such protection compared with that from vaccination.
We reviewed studies published in PubMed from inception to Sept 28, 2021, and found well conducted biological studies showing protective immunity after infection (panel). Furthermore, multiple epidemiological and clinical studies, including studies during the recent period of predominantly delta (B.1.617.2) variant transmission, found that the risk of repeat SARS-CoV-2 infection decreased by 80·5–100% among those who had had COVID-19 previously (panel). The reported studies were large and conducted throughout the world. Another laboratory-based study that analysed the test results of 9119 people with previous COVID-19 from Dec 1, 2019, to Nov 13, 2020, found that only 0·7% became reinfected.
In a study conducted at the Cleveland Clinic in Cleveland, OH, USA, those who had not previously been infected had a COVID-19 incidence rate of 4·3 per 100 people, whereas those who had previously been infected had a COVID-19 incidence rate of 0 per 100 people. Furthermore, a study conducted in Austria found that the frequency of hospitalisation due to a repeated infection was five per 14 840 (0·03%) people and the frequency of death due to a repeated infection was one per 14 840 (0·01%) people. Due to the strong association and biological basis for protection clinicians should consider counselling recovered patients on their risk for reinfection and document previous infection status in medical records.
Panel
Although those studies show that protection from reinfection is strong and persists for more than 10 months of follow-up,
it is unknown how long protective immunity will truly last. Many systemic viral infections, such as measles, confer long-term, if not lifelong, immunity, whereas others, such as influenza, do not (due to changes in viral genetics).
We are limited by the length of current reported follow-up data to know with certainty the expected duration that previous infection will protect against COVID-19. Encouragingly, authors of a study conducted among recovered individuals who had experienced mild SARS-CoV-2 infection reported that mild infection induced a robust antigen-specific, long-lived humoral immune memory in humans.
It important to note that antibodies are incomplete predictors of protection. After vaccination or infection, many mechanisms of immunity exist within an individual not only at the antibody level, but also at the level of cellular immunity.
It is known that SARS-CoV-2 infection induces specific and durable T-cell immunity, which has multiple SARS-CoV-2 spike protein targets (or epitopes) as well as other SARS-CoV-2 protein targets. The broad diversity of T-cell viral recognition serves to enhance protection to SARS-CoV-2 variants, with recognition of at least the alpha (B.1.1.7), beta (B.1.351), and gamma (P.1) variants of SARS-CoV-2.
Researchers have also found that people who recovered from SARS-CoV infection in 2002–03 continue to have memory T cells that are reactive to SARS-CoV proteins 17 years after that outbreak.
Additionally, a memory B-cell response to SARS-CoV-2 evolves between 1·3 and 6·2 months after infection, which is consistent with longer-term protection.
Some people who have recovered from COVID-19 might not benefit from COVID-19 vaccination.
In fact, one study found that previous COVID-19 was associated with increased adverse events following vaccination with the Comirnaty BNT162b2 mRNA vaccine (Pfizer–BioNTech).
In addition, there are rare reports of serious adverse events following COVID-19 vaccination.
In Switzerland, residents who can prove they have recovered from a SARS-CoV-2 infection through a positive PCR or other test in the past 12 months are considered equally protected as those who have been fully vaccinated.
Although longer follow-up studies are needed, clinicians should remain optimistic regarding the protective effect of recovery from previous infection. Community immunity to control the SARS-CoV-2 epidemic can be reached with the acquired immunity due to either previous infection or vaccination. Acquired immunity from vaccination is certainly much safer and preferred. Given the evidence of immunity from previous SARS-CoV-2 infection, however, policy makers should consider recovery from previous SARS-CoV-2 infection equal to immunity from vaccination for purposes related to entry to public events, businesses, and the workplace, or travel requirements.
NK has received consulting fees from Curative. JDK serves as an independent medical director of Curative.
References
1. 1.
◦ Dan JM, et al
Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection.
Science. 2021; 371eabf4063
August 28, 2021
Landmark mass study finds vaccinated people 13 times more likely to catch COVID-19 than those who have recovered and have natural immunity
By T Lifson 8/28/21
A study of 800,000 people in Israel has found that natural immunity in people who have recovered from an earlier COVID-19 infection is vastly superior to the immunity acquired by vaccination using two of the major vaccines in use. The conclusion:
This analysis demonstrated that natural immunity affords longer lasting and stronger protection against infection, symptomatic disease and hospitalization due to the delta variant.
Bloomberg summarizes:
People who recovered from a bout of Covid-19 during one of the earlier waves of the pandemic appear to have a lower risk of contracting the delta variant than those who got two doses of the vaccine from Pfizer Inc. and BioNTech SE.
The largest real-world analysis comparing natural immunity — gained from an earlier infection — to the protection provided by one of the most potent vaccines currently in use showed that reinfections were much less common. The paper from researchers in Israel contrasts with earlier studies, which showed that immunizations offered better protection than an earlier infection, though those studies were not of the delta variant. (snip)
The analysis also showed that protection from an earlier infection wanes with time. The risk of a vaccine-breakthrough delta case was 13-fold higher than the risk of developing a second infection when the original illness occurred during January or February 2021. That’s significantly more than the risk for people who were ill earlier in the outbreak.
Israel dealing with a severe outbreak of the delta variant:
Source.
A. Berenson details the body’s natural immune response and cites other studies as well indicating the effectiveness of natural immunity, a position that was contradicted by some earlier studies.
Marty Makary, MD, of Johns Hopkins Medical School made the point that the study is not a reason to avoid vaccination. But Dr. Makary also stated that widespread distrust of medical authorities is a response to the behavior of so many over the course of the pandemic-a position that HRS agrees with.
Age-Dependent Neutralization of SARS-CoV-2 and P.1 Variant by Vaccine Immune Serum Samples
Timothy A. Bates, Bsc1; Hans C. Leier, BA1; Zoe L. Lyski, MS1; et al James R. Goodman, MD, PhD2; Marcel E. Curlin, MD3; William B. Messer, MD, PhD1; Fikadu G. Tafesse, PhD1
JAMA. Published online July 21, 2021. doi:10.1001/jama.2021.11656
Vaccination with 2 doses of the BNT162b2 vaccine (Pfizer-BioNTech) reportedly provides 95% protection from COVID-19.1 However, patient age is known to contribute to the risk of COVID-19 incidence and severity.2 We examined the relationship between age and neutralizing antibody titers against the early SARS-CoV-2 USA-WA1/2020 strain and the P.1 variant of concern after 2 doses of the BNT162b2 vaccine.
… In this study, initial vaccine-elicited neutralizing antibody titers were negatively associated with age, resulting in a diminished ability to neutralize SARS-CoV-2 in vitro. Neutralizing titers against P.1 were reduced across all ages, although the magnitude of the age-dependent difference was smaller. Interim clinical trial data did not identify age as a contributing factor to overall vaccine efficacy.1 However, recent studies in vaccinated populations have found a measurable increase in COVID-19 cases among vaccinated older adults.3,4 The data from the current study are consistent with neutralizing antibody levels playing an important role in this observation.
Neutralizing antibody titers are thought to be strongly correlated with protection from infection; however, the threshold of this protection has not yet been precisely determined.5 … the emerging SARS-CoV-2 variants of concern, including P.1, B.1.1.7, and B.1.351, have been widely reported to be less well-neutralized by vaccine-induced antibodies and are responsible for a majority of breakthrough infections, according to a May, 2021, report.6 HRS comments: most breakthrough infections occur in those with some form of compromised immunity.
Rare ‘breakthrough’ COVID infections in vaccinated are milder: study
by Dennis Thompson 7/6/21 Read the study here
People who suffer a rare “breakthrough” coronavirus infection after getting the Pfizer or Moderna vaccine will not get as sick and, importantly, are much less likely to pass the coronavirus on to others, a new study shows.
It’s very unlikely that a person will become infected with COVID-19 after getting one of the messenger RNA (mRNA) vaccines, which provided 91% effective protection among the vaccinated people included in this study.
But those who got COVID-19 despite their vaccination wound up having milder symptoms over a shorter period of time compared to those who weren’t inoculated, researchers reported 7/1/21 in the New England Journal of Medicine.
Vaccinated people who caught COVID-19 also had a 40% lower viral load during their infection, compared with unvaccinated people said Dr. Jefferey Burgess, associate dean for research at the University of Arizona’s College of Public Health, in Tucson.
According to Dr. Amesh Adalja, a senior scholar with the Johns Hopkins Center for Health Security, in Baltimore, the findings “should give people a lot of confidence about COVID-19 vaccines. When the very rare breakthrough infections occur they are really not clinically meaningful, as the severity and infectiousness is greatly attenuated—even in not fully vaccinated individuals.”
The researchers tracked all these people from mid-December to mid-April.
156 unvaccinated people became infected with COVID-19, compared with only five fully vaccinated and 11 partially vaccinated people. A full two-dose course provided 91% protection, and even just one dose gave 81% protection, the researchers calculated. If a vaccinated person did get infected with COVID-19, they were 58% less likely to suffer a fever or chills, the results showed.
Instead, they usually had cold-like symptoms (such as the sniffles), spent two fewer days sick in bed, on average, and had an overall length of illness that was six days shorter than folks who eschewed vaccination.
This study took place before the advent of the Delta variant, which is 50 to 80 times more transmissible than the original Alpha strain of COVID-19, noted Dr. Tina Tan, a professor specializing in pediatric infectious diseases at Northwestern University’s Feinberg School of Medicine, in Chicago.
“I do not believe the Delta variant poses a problem for the vaccines, and those who develop rare breakthrough infections post-vaccination are likely to have clinically insignificant disease that is not contagious,” Adalja said.
Study suggests relatively higher neutralizing response against Delta variant in vaccinated individuals with breakthrough or prior COVID-19 infection
8/6/21: Findings from a study published in the Journal of Travel Medicine demonstrated that individuals with prior coronavirus disease 2019 (COVID-19) infection who received one dose or two doses of Covishield vaccine as well as individuals with breakthrough COVID-19 infection had relatively higher neutralizing responses against the Delta (B.1.617.2) variant in comparison to the infection-naive individuals who received either one or two doses of Covishield.
“NAbs in breakthrough participants and the COVID-19 recovered individuals with one or two-dose vaccination had relatively higher protection against Delta in comparison to the [infection-naive] vaccinees with one or two dose vaccination,” the authors wrote. “NAb titers against B.1 and Delta variants were highest among breakthrough participants which may be due to spike-specific T-cell responses.”
Reference: https://academic.oup.com/jtm/advance-article/doi/10.1093/jtm/taab119/6338089
Written by Jonathan Temte MD, PhD
COVID-19 Vaccine Effectiveness Against the Delta Variant
Viral evolution is a given. With the unfortunate continuation of thousands of daily transmission events, the generation of variants—some with alterations in transmissibility—is inevitable. If we view North America as one front in the battle against SARS-CoV-2, we have witnessed this ongoing transition. In late May 2021, 45% of sequenced viruses were assigned to the B.1.1.7 clade (Alpha variant); at that time B.1.617.2 (Delta variant) represented about 15%. Now, 2 months later, Alpha has diminished to 8% of cases and Delta has surged to 68%.1 Much of this replacement is due to transmissibility. R-naught (Ro) of the original SARS-CoV-2 was estimated at about 2.5; the Alpha variant came in at 4 to 5; Delta has topped that at 5 to 8. And with the higher Ro comes a higher herd immunity threshold.
Because of rapid expansion of the Delta variant, we are very interested in any differential effectiveness of our current vaccines across the variants. The good news is that two vaccines, one available in the United States (BNT162b2; Pfizer/BioNTech) and one available elsewhere (ChAdOx1; Astra Zeneca), maintain high levels of effectiveness against the Alpha and Delta variants.2
There are, however, some important caveats. First, there is a slight drop in overall effectiveness of vaccines for Delta compared with Alpha. Second, two doses of either vaccine are needed to achieve reasonable levels of protection. Finally, this study evaluated effective against symptomatic disease as an endpoint; it does not inform us on the protection from asymptomatic disease and the associated specter of inadvertent transmission when one is not showing signs of illness.
Take-home lessons for primary care clinicians are:
• COVID-19 vaccines protect against the common variants of SARS-CoV-2, including Delta.
• Ensuring that individuals receive all recommended doses of a COVID vaccine is paramount to protection.
• Even when vaccinated, individuals carry a slight risk for symptomatic COVID-19, and a higher risk for asymptomatic infection; vaccinated individuals may spread SARS-CoV-2.
• Use of a face mask, even if vaccinated, is warranted if among individuals at high risk.
References
1. Nextstrain. Genomic epidemiology of novel coronavirus – North America-focused subsampling. Accessed August 2, 2021. https://nextstrain.org/ncov/gisaid/north-america
2. Lopez Bernal J, Andrews N, Gower C, et al. Effectiveness of Covid-19 Vaccines against the B.1.617.2 (Delta) Variant. N Engl J Med. 2021Jul 21. doi/10.1056/NEJMoa2108891. Online ahead of print. https://www.nejm.org/doi/10.1056/NEJMoa2108891
Delta variant makes contact tracing so much harder
by C Raina MacIntyre, The Conversation 7/23/21
Delta is the most contagious of all variants known.
One study found the amount of virus shed from people infected with Delta to be over 1,000 times greater than from the original 2020 strain identified in Wuhan. Another study, which hasn’t yet been peer reviewed, showed Delta is more than twice as likely to cause hospitalization, ICU admission and death.
One detailed study showed the average time from exposure to becoming infected was six days in 2020, but four days with Delta.
NSW Health reports that when they start contact tracing, they are finding almost 100% of household members already infected, compared with about 30% last year. In South Australia it was reported people are getting infected and already infectious within 24 hours of exposure. (HRS QUESTIONS THE TRUTH OF THESE ASSERTIONS, BUT HAS NOT REVIEWED THE DATA).
© Medical Xpress 2011 – 2021 powered by Science X Network
Science vs. bureaucracy NYT 10/15/21
” … The latest example of a recurring Covid problem. Again and again, government officials have chosen to follow pre-existing bureaucratic procedures even when doing so has led to widespread public confusion and counterproductive behavior.
Officials often defend this approach by saying they merely “follow the science,” but that’s not quite accurate. When there is a conflict between scientific evidence and bureaucratic protocols, science often takes a back seat. Consider:
• At the start of the pandemic, health officials around the country were desperate to conduct Covid-19 tests, but the C.D.C. sometimes kept those officials from developing their own tests — even as the C.D.C.’s own initial test was a failure.
• Around the same time, federal officials discouraged the public from wearing masks, saying there was not enough evidence to support them — despite masks’ longtime effectiveness in Asia and inside hospitals.
• For much of this year, the F.D.A. refused to grant full authorization to any Covid vaccine — even as its top leaders were saying the shots were safe and effective and urging Americans to get vaccinated.
• The F.D.A. has been slow to approve rapid Covid tests, which helps explain why Britain, France and Germany are awash in the tests — but your local drugstore may not have any.
The repeated slowness of U.S. officials stems from a worthy goal. They want to consider the scientific evidence carefully before making decisions. They want to avoid confusing the public or, even worse, promoting less than optimal medical treatments.
Yet in their insistence on following procedures that were not written with a global pandemic in mind, officials have often ended up doing precisely what they sought to avoid. They have perplexed the public and encouraged medically dubious behavior.
During a public-health crisis, officials are most effective when they are “first, right and credible,” as the sociologist and Times Opinion columnist Zeynep Tufekci has written. They are least effective when they offer “mixed messaging, delays and confusion.” HRS says that approach is wrong as it is wrong in EVERY WAR: one must use the best information and then with humility. the best guess: that is leadership and what wins (or loses) wars.The USA Corona virus officials (FDA, NIH/Fauci.Birx) not only guessed wrong & hesitanted when, like General George Patton, in WW II,they should have been attacking.
From HealthTap 8/7/21: “The likelihood of symptomatic COVID-19 infection is extremely low, if fully vaccinated. In all reporting states, researchers found that much less than 1% of fully vaccinated people experienced breakthrough COVID-19 cases, from 0.01% in Connecticut to 0.29% in Alaska. The hospitalization rate of fully vaccinated people with COVID-19 was even lower, from 0.00% in multiple states to 0.06% in Arkansas (and hospitalization may or may not be due to COVID-19). Fortunately, the death rate for fully vaccinated people with COVID-19 was 0.00% in all reporting states but Arkansas and Michigan, where the death rate was 0.01% (and these deaths may or may not have been due to COVID-19).”
I agree with 95% +of this, but not all. My comments are in ALL CAPS
From the ‘The Washington Times’
“Biden team’s misguided and deadly COVID-19 vaccine strategy. Vaccination ‘arms race’ could prove dangerous to the American public. HRS SAYS, IT IS ALSO BENEFICIAL
By Dr. Robert Malone (HRS SAYS READ HIS AMAZING CREDENTIALS BELOW) and Peter Navarro 8/5/21
The Biden administration’s strategy to universally vaccinate in the middle of the pandemic is bad science and badly needs a reboot. HRS DISAGREES WITH THE ABSOLUTE NATURE OF THAT STATEMENT.
This strategy will likely prolong the most dangerous phase of the worst pandemic since 1918 and almost assuredly cause more harm HRS DISAGREES SIMILARLY than good – even as it undermines faith in the entire public health system. Four flawed assumptions drive the Biden strategy. The first is that universal vaccination can eradicate the virus and secure economic recovery by achieving herd immunity throughout the country (and the world). HRS AGREES WITH THIS/DISAGREES WITH THE AUTHORS HERE However, the virus is now so deeply embedded in the world population that, unlike polio and smallpox, eradication is unachievable. HRS SAYS IT CAN BE DEALT WITH FOR ALL PRACTICAL PURPOSES BY TAKING THE VACCINATIONS. SARS-CoV-2 and its myriad mutations will likely continually circulate, much like the common cold and influenza.
The second assumption is that the vaccines are (near) perfectly effective. However, our currently available vaccines are quite “leaky.” While good at preventing severe disease and death THAT IS ADEQUATE FOR HRS, they only reduce, not eliminate, the risk of infection, replication, and transmission. As a slide deck from the Centers for Disease Control has revealed, even 100% acceptance of the current leaky vaccines combined with strict mask compliance will not stop the highly contagious Delta variant from spreading.
The third assumption is that the vaccines are safe. HRS AGREES WITH THE AUTHORS THAT THE SAFETY IS UNCERTAIN, BUT THIS IS LIKE A WAR AND MOST WILL BE SAFE, A FEW WOUNDED Yet scientists, physicians, and public health officials now recognize risks that are rare YES, RARE but by no means trivial. Known side effects include serious cardiac and thrombotic conditions, menstrual cycle disruptions, Bell’s Palsy, Guillain Barre syndrome, and anaphylaxis. THAT SENTENCE IS TRUE
Unknown side effects which virologists fear may emerge include existential reproductive risks A POSSIBILITY-I HAVE NOT MADE UP MY MIND ABOUT THIS YET, additional autoimmune conditions, and various forms of disease enhancement, i.e., the vaccines can make people more vulnerable to reinfection by SARS-CoV-2 or reactivation of latent viral infections and associated diseases such as shingles. HRS SAYS “+/-“. With good reason, the FDA has yet to approve the vaccines now administered under Emergency Use Authorization.
The failure of the fourth “durability” assumption is the most alarming and perplexing. It now appears our current vaccines are likely to offer a mere 180-day window of protection HRS SAYS THESE AUTHORS SPEAK PREMATURELY– a decided lack of durability underscored by scientific evidence from Israel and confirmed by Pfizer, the Department of Health and Human Services, and other countries.
Here, we are already being warned of the need for universal “booster” shots at six-month intervals THIS IS NOT CERTAIN for the foreseeable future. The obvious broader point that militates for individual vaccine choice is that repeated vaccinations, each with a small risk, can add up to a big risk. It’s an arms race with the virus.
The most important reason why a universal vaccination strategy is imprudent tracks to the collective risk associated with how the virus responds when replicating in vaccinated individuals. Here, basic virology and evolutionary genetics tell us the goal of any virus is to infect and replicate in as many people as possible. A virus can’t efficiently spread if, like with Ebola, it quickly kills its hosts.
The clear historical tendency for viruses crossing over from one species to another is to evolve in a way that makes them both more infectious and less pathogenic over time. However, a universal vaccination policy deployed in the middle of a pandemic can turn this normal Darwinian taming process into a dangerous vaccine arms race. HRS LIKES THAT PHRASE”VACCINE ARMS RACE”
The essence of this arms race is this: The more people you vaccinate, the greater the number of vaccine-resistant mutations you are likely to get, the less durable the vaccines will become, ever more powerful vaccines will have to be developed, and individuals will be exposed to more and more risk.
Science tells us here that today’s vaccines, which use novel gene therapy technologies, generate powerful antigens that direct the immune system to attack specific components of the virus. Thus, when the virus infects a person with a “leaky” vaccination, the viral progeny will be selected to escape or resist the effects of the vaccine.
If the entire population has been trained via a universal vaccination strategy to have the same basic immune response, then once a viral escape mutant is selected, it will rapidly spread through the entire population – whether vaccinated or not.
A far more optimal strategy is to vaccinate only the most vulnerable. This will limit the amount of vaccine-resistant mutations and thereby slow, if not halt, the current vaccine arms race. CERTAINLY, THE VULNERABLE (&THERE ARE MANY) NEED IT
Fortunately, those most vulnerable represent a relatively small number; and these cohorts have already achieved high levels of vaccine acceptance. They include senior citizens, for whom the risk of serious disease or death increases exponentially with age, and those with significant comorbidities such as obesity, lung, and heart disease.
For much of the rest of the population, there’s nothing to fear but fear of the virus itself. HRS SAYS “ONLY TO A POINT. This is particularly true if we have lawful outpatient access to a growing arsenal of scientifically proven prophylactics and therapeutics. AS YOU MIGHT SUSPECT, HRS IS CHEERLEADER FOR THIS!
For example, there has been much controversy over ivermectin or hydroxychloroquine HRS USES EITHER IN HIS MEDICAL TREATMENT “COCKTAIL/SOUP” OF 8 CONSTITUENTS for 5 days. Yet, with the emergence of a growing body of scientific evidence, we can be assured these two medicines are safe and effective in prophylaxis and early treatment when administered under a physician’s supervision. Numerous other useful treatments range from famotidine/celecoxib, fluvoxamine, and apixaban to various anti-inflammatory steroids, Vitamins C & D, quercetin, selenium, and zinc.
The broader goal when administering these agents is to moderate symptoms and take death off the table, particularly for the unvaccinated. Unlike vaccines, these agents are generally not dependent on specific viral properties or mutations but instead mitigate or treat the inflammatory symptoms of the disease itself. (Pfizer is now actively marketing its own antiviral therapeutic – tacit admission Pfizer’s own vaccine is incapable of eradicating the virus.)
We are not “anti-vax.” One of us (Dr. Malone) invented the core mRNA technology being used by Pfizer and Moderna to produce their vaccines and has spent his entire professional career developing and advancing novel vaccine technologies, vaccines, and other medical countermeasures. The other (Mr. Navarro) played a key role at the Trump White House in jump starting Operation Warp Speed and ensuring timely delivery of the vaccines.
We are simply saying that just because you have a big vaccine hammer, it is not necessarily wise to use it for every nail. The American people deserve better than a universal vaccination strategy under the flag of bad science and enforced through authoritarian measures.
• Dr. Robert Malone is the discoverer of in-vitro and in-vivo RNA transfection and the inventor of mRNA vaccines while he was at the Salk Institute in 1988. Peter Navarro served in the Trump White House as the Defense Production Act Policy Coordinator.
Just below is the link to a 3/4/21 JAMA article saying that ivermectin is of no value in the treatment of the coronavirus: this is typical of the incorrect and misleading research articles that have consistently been published “against” virtually all of the available and I believe effective treatments such as colchicine, low-dose aspirin, statins, Asmanex steroid inhaler, Singulair, ivermectin, azithromycin, hydroxychloroquine, fenofibrate, lactoferrin that should be used IN COMBINATION and not as isolated treatments as is done in the treatment of diabetes, hypertension, cancer, congestive heart failure, asthma, etc. The power in these repudating comments is impressive. From the National Institues of Health/NIH website 2/11/21: “ … ivermectin acts by inhibiting the host importin alpha/beta-1 nuclear transport proteins, which are part of a key intracellular transport process that viruses hijack to enhance infection by suppressing the host’s antiviral response.4,5 In addition, ivermectin docking may interfere with the attachment of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike protein to the human cell membrane.6 Ivermectin is thought to be a host-directed agent, which may be the basis for its broad-spectrum activity in vitro against the viruses that cause dengue, Zika, HIV, and yellow fever.4,7-9 …”
https://jamanetwork.com/journals/jama/fullarticle/2777389?guestAccessKey=6fd047d5-20c0-4ecd-82aa-9f3a8255dd98&utm_source=silverchair&utm_medium=email&utm_campaign=article_alert-jama&utm_term=mostread&utm_content=olf-widget_08092021
15 Comments for this article
Study Endpoint 3/4/21
Peter Yim, PhD | Virtual Scalpel, Inc.
After initiation of the study the primary endpoint, “worsening by 2 points on the 8-category ordinal scale,” was “substantially lower” than expected. At that point the primary study endpoint was modified to be “time from randomization to complete resolution of symptoms”. Two questions to the authors:
1. Did the authors consider the following modification to the primary endpoint: “worsening by 1 point on the 8-category ordinal scale”?
2. Did the authors consider using one of the original secondary endpoints as the primary endpoint?
And, the title of the study protocol uses the term “D11AX22 Molecule” instead of “ivermectin.” Did the informed consent form also use “D11AX22 Molecule” to refer to ivermectin?
March 4, 2021
Questions
Eric Osgood, MD | St. Francis Medical Center
1. “Having received ivermectin within the previous 5 days,” was an exclusion criterion. Thee drug is used prophylactically monthly or every 2 weeks. Do we know if participants used it during a window outside the 5 days but recently enough where residual levels could have effects? If it does have benefit, couldn’t this explain why deterioration was so much rarer than anticipated based on the literature?
2. What measures were taken to ensure the placebo arm did not receive active drug prior to 9/29/20? Shouldn’t all placebo subjects have had serum ivermectin levels drawn?
3. Is “total symptom resolution” a validated metric?
4. Why do authors propose initial clinical deterioration rate of 18% was not even close to being met, and only 3.5% in the placebo arm worsened by 2 points? Could an explanation be what I proposed in question 1? With a placebo arm doing this well, would a statistically significant benefit of experimental arm even be mathematically possible?
5. Bioavailability of ivermectin is much greater if taken with a lipid-rich meal. Why were participants instructed to take it on an empty stomach?
6. Why was there no virological assessment?
Clinical Advisor to Frontline COVID-19 Critical Care Alliance (FLCCC), which has published an At-Home Outpatient Treatment Protocol with ivermectin
March 6, 2021
Methodological Flaws
H. Robert Silverstein, MD | Preventive Medicine Center, Hartford
The article states: “On October 20, 2020, the lead pharmacist observed that a labeling error had occurred between September 29 and October 15, 2020, resulting in all patients receiving ivermectin and none receiving placebo during this time frame. The study blind was not unmasked due to this error. The data and safety monitoring board recommended excluding these patients from the primary analysis but retaining them for sensitivity analysis. The protocol was amended to replace these patients to retain the originally calculated study power. The primary analysis population included patients who were analyzed according to their randomization group, but excluded patients recruited between September 29 and October 15, 2020, as well as patients who were randomized but later found to be in violation of selection criteria. Patients were analyzed according to the treatment they received in the as-treated population (sensitivity analysis).”
To me this is an error that should have stopped the study. It is my belief, and I suspect that of many others, that the editors erred allowing publication of this article.
March 5, 2021
Changing Statistical Design
Binh Ngo, M.D. | Keck USC School of Medicine
The authors did not acknowledge the most significant limitation of their study: the original power calculation was based on a 20% worsening of 2 points in their ordinal scale.
“According to the literature, 20% of patients will develop the primary outcome (worsening of 2 or more points in the 7-point ordinal scale). Thus, we will need to include 400 patients (72 total events plus 10% lost events) in order to detect a hazard ratio of 0.5 of ivermectin vs. placebo in time to deterioration, with a power of 80% and alpha of 0.05 “
In this very young population, only 12 patients reached that outcome. So they decided to change the primary outcome parameter to “resolution of symptoms”. Yet they retained the original power calculation. So they decided to simply terminate the study if “there was no indication of benefit”.
Their approach is fundamentally flawed. It is clear that all parameters were trending to favor ivermectin at the time they truncated their study. It is of concern that this issue was not highlighted by the reviewers.
Binh Ngo, M.D.
Keck USC School of Medicine
March 5, 2021
Study Power for Negative Outcome
Adriaan de Haan, B.Eng | Independent
Can the authors provide
– The study power calculations for the updated primary outcome of complete symptom resolution.
– The Type II error P value; for a negative outcome this value is as important as the Type I error P value normally published for positive outcomes.
– Information on how it was determined that the placebo arm did not take ivermectin outside of the study confines, either in the weeks leading up to the study or even during the study. It has been widely reported that South America has seen widespread adoption of ivermectin through self-medication and that this fact complicated running good studies for Ivermectin, yet I do not see any mechanisms put in place to ensure that placebo arm did not have ivermectin in their system. In fact, it was found that a large percentage was accidentally given ivermectin, casting doubt on whether the same mistake might have been made in other participants.
Also, in secondary outcomes, the Escalation of Care since Randomization for Ivermectin arm = 4.
In the Post-Hoc Outcomes, the Escalation of Care occurring >= 12 hours since Randomization for the Ivermectin arm also = 4.
This would imply that all 4 patients that had Escalation of Care since Randomization in the ivermectin arm occurred > 12 h after Randomization, hence the same 4 patients would be in both of these analyses.
However, for some reason the “Duration, median (IQR) d” for these same patients are different in the Secondary Outcomes and Post-Hoc Outcomes.
For Secondary Outcomes: 13 (3.5-21)
For Post Hoc Outcomes: 6.5 (4.5-21)
Can the authors explain how it is possible for the duration of Escalation of Care to be different for these same 4 patients in these two cases?
March 5, 2021
My concerns with this Study
Nick Arrizza, BASc, M.D.(ret’d) | Retrired Physician from Private Practice
Looking at this study they used “symptom profile” as an outcome, a crude measure in my view as these are variable from start to finish (while also treating “symptoms” of both groups with NSAIDS, steroids, and so on). A negative PCR test should have been the primary outcome measure and in my view is one of the significant weaknesses of this study. So are we simply looking at the effectiveness of the complementary drugs (present in both groups) to treat ‘symptoms’ or are we actually seeing the potential effects of ivermectin on disease resolution? In addition there appears to be an imbalance among treatment & placebo groups in the numbers of male and female members which if significant could be confounders.
March 5, 2021
Findings Seem to Favor Ivermectin
Kevin Tomera, MD | Beloit Health System
i find it interesting that viral clearance, hospitalization, fever, time with fever, and clinical deterioration all favor ivermectin.
To expect total resolution of all covid symptoms within 21 days has not been achieved by any treatment. So why did the authors and editors choose this lofty goal ?
Bias
Hector Carvallo, Professor of Medicine | Universidad Abierta Interamericana Argentina
It is no secret that young COVID patients will, fortunately, develop few symptoms (if any) and will recover far sooner than aged ones, not only because of age difference but also because of the lack of co-morbidities. What this trial missed is the chance to investigate viral load in those subjects; if so, they would have found out something important: those young patients surely ceased to provoke contagions. Is it evidence-based medicine or just bias? I foresee we will witness lots of articles like this in the near future.
G-Med Global 11/6/21: “ …See ivmmeta.com. This is a met analysis of 64 studies on ivermectin’s benefits including 31 RCTs. There is scientific literature showing that ivermectin decreases replication of the virus through decreased attachment, inhibition of RNA dependent RNA polymerase, inhibition of importin related transport of the virus into the nucleus (which is responsible for decreasing local interferon response), and inhibiting 3 C like protease which is needed to break apart the different proteins for reassembly. It also modulates the immune response by inhibiting Nuclear Factor Kappa Beta. Furthermore it is extremely safe if your patients have an intact blood brain barrier. IT IS CONTRAINDICATED IN THOSE WITHOUT A BLOOD BRAIN BARRIER, CHILDREN UNDER 2, PREGNANT WOMEN, AND THOSE WITH AN ACTIVE LOA LOA INFECTION. With overwhelming literature support, numerous beneficial results from thousands of physicians, well delineated mechanisms of action, a dramatic decrease in severe disease with its widespread use, and an extremely safe and inexpensive medication, I believe that it should absolutely be used …”
September 5, 2021
Fake News: The strange campaign against ivermectin escalates
By Monica Showalter
The campaign against ivermectin?
It’s not just that the FDA has put out a quackish tweet (shown here by AT contributor Dr. Brian Joondeph, M.D.) scolding viewers that they are not horses or cows. That’s in reference to the fact that in some high-dose forms, ivermectin is used as a horse dewormer, begging the question about its effectiveness when it is dosed and formulated properly as a human medical treatment for COVID, prescribed off-label. The news reports repeatedly call ivermectin a dangerous drug, implying that all uses of the drug are dangerous, even when taken properly.
It’s now a lot of nonsense about mass injuries from people who take the horse dewormer form of the drug on their own to beat COVID, as if there is anyone out there advocating such an improper use of the medicine.
Here are a few of the screaming headlines from multiple news outlets:
Gunshot Victims Left Waiting as Horse Dewormer Overdoses Overwhelm Oklahoma Hospitals, Doctor Says -Rolling Stone
Gunshot victims left to wait as Oklahoma hospitals overwhelmed with horse dewormer overdoses, doctor says -Fox59
(Funny that similarity, almost like the same guy wrote those headlines?)
Overdoses from anti-parasite drug ivermectin overwhelm rural Oklahoma hospitals – leaving gunshot victims waiting for emergency rooms -Daily Mail
Doctor says gunshot victims forced to wait for treatment as Oklahoma hospitals fill up with people overdosing from ivermectin -Independent
One problem: The story is fake.
The hospital in Oklahoma that was said to be at the epicenter of all those many gunshot victims (got questions about that, too) left waiting for their emergency care, is explicitly denying that it’s overwhelmed by horse-ivermectin overdose cases and the lone doctor making the claim, Dr. Jason McElyea, is not an employee of the hospital at all, he’s a contractor, and hasn’t worked at the hospital for months. In fact, it hasn’t treated any horse-ivermectin cases at all.
Here’s the statement, as reported by Rolling Stone:
UPDATE: Northeastern Hospital System Sequoyah issued a statement: Although Dr. Jason McElyea is not an employee of NHS Sequoyah, he is affiliated with a medical staffing group that provides coverage for our emergency room. With that said, Dr. McElyea has not worked at our Sallisaw location in over 2 months. NHS Sequoyah has not treated any patients due to complications related to taking ivermectin. This includes not treating any patients for ivermectin overdose. All patients who have visited our emergency room have received medical attention as appropriate. Our hospital has not had to turn away any patients seeking emergency care. We want to reassure our community that our staff is working hard to provide quality healthcare to all patients. We appreciate the opportunity to clarify this issue and as always, we value our community’s support.”
Which pretty well discredits the whole hysterical narrative being spread around by multiple news outlets. It’s fake news, based on shoddy reporting, from a claim of one source with zero firsthand knowledge, yet a whole host of news outlets picked up the fake story and spread it around to scare people.
Now, obviously, the hospital would have an interest in seeing patients coming in for treatment and a report like that probably would have kept a lot of them away.
But at the same time, a detailed denial like that is pretty firm and the hospital would be in trouble if it were not true. They said they’re treated no one for taking horse dewormer, no one at all.
So the news outlets have made huge errors in judgment and trashed their own credibility based on that now-denied report they could have gotten the answer to, and it was one after another running themselves off the cliff, none of them really checking, all of them taking the word of one doctor, who could have had any agenda. I suspect CNN might have run a version on the story based on multiple claims on Twitter and the fact that a Google search with CNN, Oklahoma, and hospital keywords turns up with CNN’s name in news pickups from others such as Yahoo!, but all clicks to the story come up empty. If so, it’s got to be bad.
It follows from another claim that multiple news agencies manipulated a photo of famous podcaster Joe Rogan, who had COVID and took a people-version of ivermectin, prescribed off-label by a doctor, and was cured quickly of his illness. The news photos showed a picture of his face with a yellow filter and slight blurring in a bid to make him look kind of green and sick. The original photos showed him looking perfectly healthy.
There’s some kind of campaign ramping up against ivermectin, a drug whose inventor won the Nobel prize in 2015 for the first version of, and which has been endorsed by the Japan Medical Association, as well as in many studies which have found it to be an effective early treatment for COVID. There’s no President Trump involved, so it’s something very funny going on. Could it be the work of Big Pharma which has far more expensive medications for COVID to sell? It’s terrible if true, given that there’s a lot of evidence out there that ivermectin does indeed cure the illness.
But that’s no issue to the press it seems, and other than Rolling Stone, none have published the denial. What’s going on here? Why this ramped up campaign of lies and manipulations?
NIH support for ivermectin wi references:
U.S. Department of Health & Human Services
Search
• Everything
• News
• Videos
• Clinical Trials
• Este tema en español
Top of Form
Enter your search term
Bottom of Form
Search results
Powered by Bing
Ivermectin | COVID-19 Treatment Guidelines
www.covid19treatmentguidelines.nih.gov/therapies/antiviral-therapy/ivermectin/
Ivermectin is a Food and Drug Administration (FDA)-approved antiparasitic drug that is used to treat several neglected tropical diseases, including …
A five-day course of ivermectin for the treatment of COVID …
pubmed.ncbi.nlm.nih.gov/33278625/
Ivermectin, a US Food and Drug Administration-approved anti-parasitic agent, was found to inhibit severe acute respiratory syndrome coronavirus 2 …
Ivermectin for Prevention and Treatment of COVID-19 …
www.ncbi.nlm.nih.gov/pmc/articles/PMC8248252/
Ivermectin is not a new and experimental drug with an unknown safety profile. It is a WHO “Essential Medicine” already used in several different …
Ivermectin for Prevention and Treatment of COVID-19 …
pubmed.ncbi.nlm.nih.gov/34469921/
Ivermectin for Prevention and Treatment of COVID-19 Infection: A Systematic Review, Meta-analysis, and Trial Sequential Analysis to Inform Clinical …
The mechanisms of action of Ivermectin against SARS-CoV-2 …
www.ncbi.nlm.nih.gov/pmc/articles/PMC8203399/
Ivermectin is enlisted in the World Health Organization’s Model List of Essential Medicines . Drug repurposing, drug redirecting, or drug reprofiling …
Review of the Emerging Evidence Demonstrating the Efficacy …
www.ncbi.nlm.nih.gov/pmc/articles/PMC8088823/
Ivermectin hastens recovery and prevents deterioration in patients with mild to moderate disease treated early after symptoms. 45,49–52,61,62. Ivermec …
Ivermectin for preventing and treating COVID‐19
www.ncbi.nlm.nih.gov/pmc/articles/PMC8406455/
What is ivermectin? Ivermectin is a medicine used to treat parasites such as intestinal parasites in animals and scabies in humans. It is cheap and is …
Use of ivermectin in the treatment of Covid-19: A pilot trial
pubmed.ncbi.nlm.nih.gov/33723507/
Ivermectin is safe in patients with SARS-CoV-2, reducing symptomatology and the SARS-CoV-2 viral load. This antiviral effect appears to depend on the …
Ivermectin: a systematic review from antiviral effects to …
pubmed.ncbi.nlm.nih.gov/32533071/
Ivermectin proposes many potentials effects to treat a range of diseases, with its antimicrobial, antiviral, and anti-cancer properties as a wonder …
Ivermectin: a multifaceted drug of Nobel prize-honoured …
pubmed.ncbi.nlm.nih.gov/34466270/
In 2015, the Nobel Committee for Physiology or Medicine, in its only award for treatments of infectious diseases since six decades prior, honoured the …
Ivermectin, ‘Wonder drug’ from Japan: the human use …
www.ncbi.nlm.nih.gov/pmc/articles/PMC3043740/
Ivermectin proved to be virtually purpose-built to combat Onchocerciasis, which has two main manifestations, dermal damage resulting from …
Use of Ivermectin Is Associated With Lower Mortality in …
pubmed.ncbi.nlm.nih.gov/33065103/
Background: Ivermectin was shown to inhibit severe acute respiratory syndrome coronavirus 2 replication in vitro, which has led to off-label use, but …
Ivermectin for preventing and treating COVID-19
pubmed.ncbi.nlm.nih.gov/34318930/
Ivermectin compared to no treatment for prevention of SARS-CoV-2 infection We found one study. Mortality up to 28 days was the only outcome eligible …
Effects of Ivermectin in Patients With COVID-19: A …
www.ncbi.nlm.nih.gov/pmc/articles/PMC8101859/
Ivermectin has been suggested as a potential COVID-19 treatment. 6 It is a US Food and Drug Administration (FDA)-approved broad-spectrum antiparasitic …
Drug repurposing in COVID-19: A review with past, present …
www.ncbi.nlm.nih.gov/pmc/articles/PMC8387125/
Ivermectin, a broad-spectrum anti-parasitic agent has also been shown to be effective against some viral infections. Recently, this drug was studied …
Ivermectin | C48H74O14 – PubChem
pubchem.ncbi.nlm.nih.gov/compound/ivermectin
Ivermectin is an orally bioavailable macrocyclic lactone derived from Streptomyces avermitilis, with antiparasitic and potential anti-viral …
The Role of Distinct Subsets of Macrophages in the …
www.ncbi.nlm.nih.gov/pmc/articles/PMC8417824/
Ivermectin (IVM) is a semisynthetic macrocyclic lactone that FDA has approved for parasitic disease treatment. IVM interacts with P2X4R and …
Ivermectin, a potential anticancer drug derived from an …
www.ncbi.nlm.nih.gov/pmc/articles/PMC7505114/
Ivermectin is a macrolide antiparasitic drug with a 16-membered ring that is widely used for the treatment of many parasitic diseases such as river …
Ivermectin: a multifaceted drug of Nobel prize-honoured …
www.ncbi.nlm.nih.gov/pmc/articles/PMC8383101/
Introduction. The 2015 Nobel prize for the discovery of ivermectin (IVM) and an antimalarial treatment was the Nobel committee’s first award for …
Is indiscriminate use of medicines the main reason for …
www.ncbi.nlm.nih.gov/pmc/articles/PMC8384350/
Ivermectin, another controversial drug, has gained widespread momentum in India and South America despite WHO recommending against its use. It was …
Absence of Evidence is Not Evidence Against
Adesuyi Ajayi, MD, PhD | Adjunct Prof Clinical Pharmacology & Medicine, Baylor College of Medicine 3/17/21
Lopez-Medina et al, conclude ivermectin is ineffective for COVID 19 based on a “soft end point ” of a 2 level deterioration in an 8 point questionnaire ordinal scale. This is a grossly inadequate premise as other studies including ours (1) that used “hard end point” RT-PCR for SARS-CoV-2 repeated measurements over 14 days showed significant, dose-dependent virucidal effects of ivermectin to reduce days to COVID 19 negativity by Kaplan-Meir statistics as well as by the treatment effect by 2-way repeated measures ANOVA. There are many confounders to symptomatic amelioration, and measures of virucidal activity is therefore imperative to show efficacy of ivermectin. RT-PCR provides this. Further, symptoms such as hiccups, myalgia, headaches, tiredness, and amnesia may persist after SARS-CoV-2 virological clearance, giving rise to chronic COVID-19 or the Long Haulers syndrome” of COVID-19. The 95% confidence intervals of the symptomatic parameters reported all tend to suggest ivermectin superiority over placebo in the study. There were minimal objective laboratory parameters such as SPO2%, D dimer, cytokine indices, or thrombosis parameters measured to support the conclusion of lack of ivermectin efficacy. This a classic example of the dictum that ” Absence of evidence, is not evidence against” as the valid and sensitive end points of anti SARS-CoV-2 efficacy were not employed in the study. Larger international RCTs of sufficient power and hard end points are required to establish the utility of ivermectin in Covid-19 and as a potential broad spectrum antiviral drug against future RNA viruses.
Reference
1. Babalola OE, Bode CO, Ajayi AA et al QJM 2021, 114 doi,org /10.1083/qjmed/hcab035
Ivermectin-Typical AEs in Placebo Group 3/12/21
David Scheim, PhD (MIT) | U.S. Public Health Service, Commissioned Corps, Inactive Reserve
The Lopez-Medina et al. study reported a striking anomaly: adverse effects that are characteristic of ivermectin (IVM), described in the study protocol as “security parameters” for its cumulative high IVM dose, occurred at almost identical rates in its IVM and placebo arms.
Most notable of these AEs were blurred vision and dizziness, both characteristic of higher dose IVM use [1, 2] but of limited incidence in COVID-19 [3-5]. These AEs occurred in percentages of 11.3%, 11.6% for blurred vision and 35.6%, 34.3% for dizziness, respectively, of the IVM and placebo groups. These signs of IVM use in controls occurred against a backdrop of surging over-the-counter (OTC) sales of IVM in the study region (available without prescription) during the study period to a total of 154,919 units, 1.6 times the number of COVID-19 cases [6].
The study was in fact lax in protecting boundaries between its IVM and placebo groups. IVM was substituted for placebo for 38 designated control patients, discovered one month after the fact when “the lead pharmacist observed that a labeling error had occurred” (study paper, p. 3; study protocol, p. 43). Blinding was also compromised, since bitter-tasting IVM was easily distinguishable from the 5% dextrose solution received by the first 64 placebo subjects in the study (supplement 2, eFigure 1). For the other 134 controls, a placebo of unspecified composition was then used, with no report provided of having it tested by taste or otherwise.
These distinctive AEs for IVM in controls, in the context of widespread OTC availability of IVM in the study region during the study period and multiple protocol lapses and deficiencies, seriously compromise the study. Valuable information has nevertheless been gleaned from outcomes for patients in the IVM treatment arm, which had no deaths, generally mild symptoms, and AEs typical for high-dose IVM (replicated in the control group) that were generally mild and transient.
References
1. Kamgno J, Gardon J, Gardon-Wendel N, Demanga N, Duke BO, Boussinesq M. Adverse systemic reactions to treatment of onchocerciasis with ivermectin at normal and high doses given annually or three-monthly. Trans R Soc Trop Med Hyg. 2004;98(8):496-504.
2. Smit MR, Ochomo EO, Aljayyoussi G, Kwambai TK, Abong’o BO, Chen T, et al. Safety and mosquitocidal efficacy of high-dose ivermectin when co-administered with dihydroartemisinin-piperaquine in Kenyan adults with uncomplicated malaria (IVERMAL): a randomised, double-blind, placebo-controlled trial. Lancet Infect Dis. 2018;18(6):615-626.
3. Pardhan S, Vaughan M, Zhang J, Smith L, Chichger H. Sore eyes as the most significant ocular symptom experienced by people with COVID-19: a comparison between pre-COVID-19 and during COVID-19 states. BMJ Open Ophthalmology. 2020;5(1):e000632.
4. Pinzon RT, Wijaya VO, Buana RB, Al Jody A, Nunsio PN. Neurologic Characteristics in Coronavirus Disease 2019 (COVID-19): A Systematic Review and Meta-Analysis. Frontiers in Neurology. 2020;11(565).
5. Chen X, Laurent S, Onur OA, Kleineberg NN, Fink GR, Schweitzer F, et al. A systematic review of neurological symptoms and complications of COVID-19. Journal of Neurology. 2021;268(2):392-402.
6. Scheim DE. Ivermectin sales in Valle del Cauca, Colombia, patterns of AEs, and other background re López-Medina et al. 2021: OSF Preprints; 2021 [Available from: https://doi.org/10.31219/osf.io/6m3ch]. Access date March 11, 2021.
Author Response 3/23/21
Eduardo Lopez-Medina, MD, MSc | Centro de Estudios en Infectología Pediátrica
Our study has received several comments. Most of the criticisms were acknowledged in the manuscript, including the lack of virological assessments and ivermectin plasma levels or the fact that the original primary outcome to detect the ability of ivermectin to prevent clinical deterioration was changed 6 weeks into the trial. We also acknowledged that the placebo used in the first 65 patients differed in taste and smell from ivermectin and explained why this limitation did not compromise the study or its conclusions. Of note, we did report that the manufacturer’s placebo had similar organoleptic properties to ivermectin. Participants were instructed to take ivermectin on an empty stomach, as recommended by the FDA.
It is important to highlight that the primary outcome of this trial was the time to resolution of symptoms. Therefore, even in a relatively young and healthy population, if the true HR of ivermectin vs. placebo was 1.4 (as assumed when calculating the sample size), the study had sufficient power to detect that difference. We have also noted that our study may have been underpowered to detect smaller differences and that larger trials may be needed to understand the effects of ivermectin on other clinically relevant outcomes.
An interesting observation was made concerning the similar proportions of patients in the ivermectin and placebo groups who had side effects that are characteristic of high ivermectin doses (visual disturbances or dizziness), raising concerns that patients in the placebo group were taking ivermectin during the trial. As described in the article, patients were contacted daily for a structured interview that documented the use of any medications outside the study. In addition, we performed an analysis that excluded patients in the placebo group who had visual disturbances or dizziness, in order to restrict the subgroup to patients who were “true placebo.” The HRs for recovery were (>1 indicates benefit of ivermectin):
Ivermectin group vs. patients in the placebo group who
• did not have dizziness: HR: 0.89 (95% CI: 0.71 to 1.13), P value: 0.35
• did not have vision changes: HR: 0.96 (95% CI: 0.77 to 1.19), P value: 0.72
Regarding the question of whether the informed consent form (ICF) used “D11AX22 Molecule” to refer to ivermectin, the answer is yes. This was discussed and approved by the ethics committee and the national regulatory agency. The ICF met all requirements of the International Conference on Harmonization of GCP, including details related to the drug and the possibility to reposition it against COVID-19. The need to use D11AX22 rather than ivermectin in the ICF arose from the extensive use of ivermectin in the city of Cali during the study period, extensive recommendations from some political and medical leaders to use it against COVID-19, and the fact that the initial placebo had a different taste from ivermectin. The only option to maintain the blind and prevent self-medication for participants in the placebo group during the dextrose/saline-placebo period was to use “D11AX22 Molecule” in the ICF.
Finally, concerns have been raised regarding the allocation of patients to the incorrect treatment group between September 29 and October 15, 2020 and whether the same mistake might have been made in other participants. When the labeling error was detected, the non-blinded study personnel double-checked the entire randomization/labeling/assignment process, confirming that the correct allocation was given to patients in the rest of the study. The study blind was not unmasked due to this error
Defining Status of Illness at the Beginning of Treatment
Marianella Herrera-Cuenca, MD, PhD | Universidad Central de Venezuela
Can you clarify how many days on average elapsed between the diagnosis of COVID-19 and the beginning of treatment with ivermectin? It seems as if 2 “bonus” days are not bad at all. Maybe another statistical assessment?
This work is very important in context of the low and middle income countries where vaccines might be delayed, so getting more information on ivermectin usage can be of help if demonstrated as an effective and cost-beneficial alternative.
Ivermectin vs Placebo and Time to Symptom Resolution 4/14/21
Stephen Strum, MD, FACP | Practice of Hematology & Oncology
I have read every available publication, peer-reviewed & not, on ivermectin (IVM) vs COVID-19. It is amazing to see the spectrum of results from ineffective to highly effective. What is troubling is that some papers do not use PCR positivity at diagnosis of COVID-19 and evidence of PCR negativity for SARS-CoV-2 as an end-point. Others use different doses of IVM, and schedules that are either single dose or multiple doses each day time 5 days or more. Most studies do not have pharmacokinetics, an exception being that of Krolewiecki et al. who showed a response related to plasma IVM of 160 ng/ml or higher (1).
What is especially disturbing is a lack of cooperation among investigators in getting to the core issue based on a clean protocol design. We hear talk of the importance of collaboration and given the criticality of COVID-19 and the emergence of variants, the physicians & scientists cannot seem to create a task force that would definitively answer questions as to efficacy. Instead, we perpetuate controversy, as we did with hydroxychloroquine and other agents. How about some “resolution-based” collaboration?
Reference
1. https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3714649
Early Administration? 4/25/21
Silvina Alfieri | Farmacéutica, Oficina de Farmacia Privada
Adult men and non-pregnant or lactating women were eligible if their symptoms started in the previous 7 days and they had mild illness. Can the first days of infection be considered as day 7? Early administration is critical. The dissonance observed between clinical trials and the version reported by patients treated with ivermectin is striking.
In addition to the above standard tests, because of the skew towards moderately higher than expected p-values for chi square tests of baseline comborbidities I also examined the order of dichotomous data. I constructed continuous duplicates and triplicates exhaustvely (i.e. all possible combinations) and compared the frequency of positive duplicates (1,1) negative duplicates (0,0) positive triplicates (1,1,1) and negative triplicates (0,0,0). No irregularities were found across all baseline variables. This is very reassuring. While this can technically be non-experimentally produced using an RNG, in that case expected correlations (e.g. between certain comorbidities) are lost, and I can confirm in this case they were preserved. This is essentially impossible to fake over this volume of data.
The raw data are consistent with the published paper.
In short, the authors provided a complete data set (except for anonymisation), the data set did not show any signs of manipulation or fabrication. My assessment is that the data are genuine and are consistent with the trial being conducted as described in this paperFederal Input Demands Simple Answers to Complex Problems
By Ted Noel, MD 9/6/21
Early twentieth-century satirist H.L. Mencken quipped, “For every complex problem, there is a solution that is simple, neat, and wrong.” The ongoing COVID-19 disaster illustrates the truth of Mencken’s Meta-Law.
We have a virus afflicting us. We have multiple viruses, and alpha, beta, gamma, delta, lambda, and who knows how many other variants of COVID. Yet the answer given by the CDC, Fauci, Biden, and the media is that we have to wear masks, get vaxxed, socially distance, and maybe lock down. Those are simple solutions to a much more complicated problem than those problem-solvers are willing to admit.
There are at least five notable variants: like with hurricanes, they are named for Greek letters.
Those viruses aren’t identical. We now know that the vaccines don’t work equally well on all of them. We know that more variants are coming. Today’s vaccine won’t necessarily give the same immunity for today’s variant and tomorrow’s variant.
We know that some people are naturally immune to COVID without ever knowingly being exposed to either the virus or the vaccinex. We’re not sure why, but one suggestion is that they’ve already been exposed to some other coronavirus. That wouldn’t be a surprise because at least two coronaviruses can cause the common cold. Others have recovered from CoV2-19 and have more robust immunity to more variants than people who only received the current vaccine(s).
We learned very early on that the elderly and infirm were at highest risk. The Italian data showed 2.3 serious comorbidities (other illnesses) plus an age of 82 in the average person who died. It turns out that the average life expectancy for those people without COVID was another seven months.
In younger people, the numbers are quite different. Older people died at about 400 times the rate of young people in the early data. Now we know that the real number is closer to 1,000 times.
When we look at average mortality rates from infection, we find ourselves firmly trapped in Simpson’s Paradox. Worldwide, the average mortality from COVID is 0.27%, in the ballpark with seasonal flu. But that number is meaningless without context. In most of the U.S., mortality under age 70 is at or below 0.1%. English data are similar. In the U.K., delta variant infections have a 2.0% mortality in patients over 50, but the death rate below 50 is statistically indistinguishable from zero. Put bluntly, lumping everyone together is guaranteed to yield a biased answer, while separating distinct groups makes good sense. We have to have different answers for elders and the young.
If someone arrives at the hospital in extremis with CoV2-19/COVID, they are probably is in Stage III of the disease. By then, the virus has stopped being a problem. The body’s “cytokine storm” has taken over.
Source.
If you use antivirals in Stage III, they do very little good because SARS CoV-2 is long gone. That’s why Remdesivir did so poorly in the original COVID trials. It’s also why HCQ doesn’t do well in ICU patients. Its antiviral effects have no virus left to work on. But if you use antivirals in the early stage of the infection, when there’s lots of virus around, they can be very good. There are at least thirteen outpatient treatment protocols that address this issue.
The problem is complex and the simple answer is wrong. The CDC has favored specific therapies for advanced cases at the same time another government agency published multiple studies showing that “large reductions in COVID-19 deaths are possible using ivermectin.”
Fauci and the CDC are pushing universal vaccination, while efficacy of the vaccines is waning. Their answer seems to be simeply “double down” omitting that the death rate from CoV2-19 in the young and healthy approximates zero. The young don’t even spread the bug very well–evenquestion the need for vaccination. And the young are prone to heart inflammation and miscarriages from the shot at a statisically rlevant number, even thought hat number is small. If the youngget sick with the actual virus, they can use the very inexpensive cocktail that India used with nearly universal success.
We cannot leave the issue of complex questions without discussing masks. There are literally dozens of studies on masking. Some do provide adequate confidence that masking by the general public has any benefit in reducing the spread of CoV2-19/COVID if it is the proper mask and how it is worn is correctly u to the bridge of the nose while covering the mouth and chin.
There is one area where masking has proven benefit: COVID units in hospitals. In those portions of the hospital, the presumed high intensity of virus in the air is beaten by high-quality N-95 masks or better, but only when properly fitted, worn, and disposed of. Masks that don’t fit tightly actually increase the risk of infection. And once again, Simpson’s Paradox strikes us squarely in the face. If you have different populations in different circumstances, they will have different responses to interventions. Estrogen receptor–positive breast cancer requires a different treatment from triple-negative breast cancer. The same for a host of other diseases. Every doctor has learned this.
Mask, distance, vaccine! Rinse and repeat. This is a simple answer to a complex question. The list of errors and vacillations is too long to tackle here. Their prescriptions may well be responsible for uncountable deaths and untold misery.
Evaluation of potential COVID-19 recurrence in patients with late repeat positive SARS-CoV-2 testing
• Ithan D. Peltan , et al PLOS 5/2021
• https://doi.org/10.1371/journal.pone.0251214
• Figures
Figures
Abstract
Background
SARS-CoV-2 reinfection and reactivation has mostly been described in case reports. We therefore investigated the epidemiology of recurrent COVID-19 SARS-CoV-2.
Methods
Among patients testing positive for SARS-CoV-2 between March 11 and July 31, 2020 within an integrated healthcare system, we identified patients with a recurrent positive SARS-CoV-2 reverse transcription polymerase chain reaction (RT-PCR) assay ≥60 days after an initial positive test. To assign an overall likelihood of COVID-19 recurrence, we combined quantitative data from initial and recurrent positive RT-PCR cycle thresholds—a value inversely correlated with viral RNA burden— with a clinical recurrence likelihood assigned based on independent, standardized case review by two physicians. “Probable” or “possible” recurrence by clinical assessment was confirmed as the final recurrence likelihood only if a cycle threshold value obtained ≥60 days after initial testing was lower than its preceding cycle threshold or if the patient had an interval negative RT-PCR.
Results
Among 23,176 patients testing positive for SARS-CoV-2, 1,301 (5.6%) had at least one additional SARS-CoV-2 RT-PCRs assay ≥60 days later. Of 122 testing positive, 114 had sufficient data for evaluation. The median interval to the recurrent positive RT-PCR was 85.5 (IQR 74–107) days. After combining clinical and RT-PCR cycle threshold data, four patients (3.5%) met criteria for probable COVID-19 recurrence. All four exhibited symptoms at recurrence and three required a higher level of medical care compared to their initial diagnosis. After including six additional patients (5.3%) with possible recurrence, recurrence incidence was 4.3 (95% CI 2.1–7.9) cases per 10,000 COVID-19 patients.
Conclusions
Only 0.04% of all COVID-19 patients in our health system experienced probable or possible recurrence; 90% of repeat positive SARS-CoV-2 RT-PCRs were not consistent with true recurrence. Our pragmatic approach combining clinical and quantitative RT-PCR data could aid assessment of COVID-19 reinfection or reactivation by clinicians and public health personnel. HRS states almost certainly as a minority opinion, this implies there is no need to immunize those who have been previously infected unless they have some immune compromise.
Interpreting SARS-CoV-2 Test Results
KC Coffey, MD, MPH1; Daniel J. Diekema, MD, MS2; Daniel J. Morgan, MD, MS1
JAMA. Published online September 17, 2021. doi:10.1001/jama.2021.16146
•
Research Letter
Mass SARS-CoV-2 Testing of Asymptomatic Patients Before Preplanned Procedures
Scott C. Roberts, MD, MS; David R. Peaper, MD, PhD; L. Scott Sussman, MD; Richard A. Martinello, MD; Christian M. Pettker, MD
Case
A 53-year-old woman was referred to the gastroenterology clinic for endoscopy because of a submucosal gastric nodule. She had not received a COVID-19 vaccination and lived in Maryland, which had a 7-day cumulative COVID-19 case rate of 70 per 100 000 individuals at the time of her visit. Review of systems was unremarkable except for intermittent abdominal pain. She had no fever, cough, shortness of breath, difficulty breathing, muscle aches, headache, sore throat, anosmia, dysgeusia, or diarrhea. SARS-CoV-2 reverse transcriptase–polymerase chain reaction (RT-PCR) testing prior to the procedure was performed, in accordance with the American Society of Anesthesiologists Statement on Perioperative Testing1 for monitored anesthesia. Results of preoperative testing are shown in Table 1.
Table 1. Patient’s Test Results
View LargeDownload
Based on the results, additional history was obtained. The patient reported testing positive for SARS-CoV-2 30 days prior at another hospital. At that time, she had symptoms of dry cough, low-grade fever, and body aches, which resolved after 14 days. Fourteen days before the current presentation, results of a rapid antigen test performed at a local pharmacy were negative for SARS-CoV-2.
What Would You Do Next?
1. Cancel the endoscopy procedure due to likely reinfection
2. Cancel the endoscopy procedure due to persistent infection
3. Proceed with endoscopy, the patient is clinically recovered and not contagious
4. Repeat the RT-PCR testing now because this is likely a false-positive result
Discussion
Answer
C. Proceed with endoscopy, the patient is clinically recovered and not contagious
Test Characteristics
SARS-CoV-2 RT-PCR is the primary diagnostic test for COVID-19 (Medicare reimbursement, $75). The test amplifies targeted nucleic acid sequences to detect SARS-CoV-2 RNA. RT-PCR testing detects SARS-CoV-2 RNA at low levels, with analytic sensitivity of 98% and specificity of 97%.2 Analytic sensitivity and specificity refer to RT-PCR detection of SARS-CoV-2 RNA in laboratory samples, while clinical sensitivity and specificity refer to identifying patients with and without COVID-19. Clinical sensitivity is approximately 90% and clinical specificity is approximately 95%.3-5 Time from symptom onset, specimen source, and user error all affect clinical sensitivity (Table 2). Sensitivity of RT-PCR to detect patients with SARS-CoV-2 that can be cultured and infect others is 99%; however, specificity is limited by persistent detection of noninfectious viral RNA.4,5,7
Table 2. Factors That Affect the Clinical Sensitivity or Specificity of SARS-CoV-2 Tests
View LargeDownload
The Centers for Disease Control and Prevention advises that immune-competent adults are not infectious more than 10 days after symptom onset.7 RT-PCR testing detects noninfectious viral RNA up to 12 weeks after infection.7 To distinguish infectious from noninfectious virus, cycle thresholds may be used. The cycle threshold is the number of cycles a sample must be amplified in the laboratory before virus can be detected. A low cycle threshold value correlates with higher viral load and contagiousness because fewer cycles are required to detect virus. Cycle threshold values are also affected by collection technique and vary by assay, reducing their reliability.8
Application to This Patient
This patient had persistently positive RT-PCR test results for SARS-CoV-2 less than 90 days from prior infection. Because she remained asymptomatic, retesting provided no useful information. The Centers for Disease Control and Prevention recommend that patients infected within the past 90 days without new COVID-19 symptoms should not be retested.9 Reinfection with SARS-CoV-2 is rare (risk of reinfection, 0.17%).10 However, whether reinfection rates will increase due to Delta or other variants is unknown. There is no evidence of altered test performance with variants.
Patients undergoing asymptomatic screening have a pretest probability of COVID-19 that mirrors local prevalence. Thus, at the time of testing in this patient, the pretest probability of COVID-19 was approximately 0.35% (7-day cumulative average [0.07%] multiplied 5-fold to account for undertesting). Estimating 90% sensitivity and 95% specificity, the test positive predictive value was 24% but declined to approximately 0% given the patient’s recent history of COVID-19.
The ability of preprocedural testing to prevent nosocomial COVID-19 transmission varies with disease prevalence, infection history, the patient’s immunization status, employee vaccination rate, and personal protective equipment availability. Testing programs must consider what procedures have a high risk of transmission (eg, intubation and bronchoscopy aerosolize particles from the respiratory tract and are associated with higher transmission risk) and balance transmission risk reduction with potential harms to patients that result from delayed procedures following a positive test result.
What Are Alternative Diagnostic Testing Approaches?
Alternative tests include point-of-care antigen tests, which are less sensitive than RT-PCR but better predict contagiousness, indicated by correlation with viral culture and higher viral loads.4,6 Using viral culture as criterion standard, small studies of antigen tests have reported sensitivity of approximately 90% and specificity greater than 98%.4-6
Patient Outcome
Consultation from an infectious disease specialist determined that the patient did not pose a transmission risk to medical staff. However, after further review the patient did not require endoscopic evaluation of the submucosal nodule for another 6 months, and the procedure was postponed.
Clinical Bottom Line
• SARS-CoV-2 reverse transcriptase–polymerase chain reaction (RT-PCR) testing is highly sensitive (99%) but less specific (approximately 95%) for contagious COVID-19 and may be positive for ≥90 days after infection
• Viral antigen tests have lower analytic sensitivity than RT-PCR but are more specific for contagious disease
• Preprocedural COVID-19 testing can delay access to care, and testing asymptomatic low-risk patients undergoing an aerosol-generating procedure is not recommended when procedure-based personal protective equipment is available2
Yfefstgrreprdmayon aStod 1:03 PaM ·
‘The Atlantic’ magazine found in a deep-dive study that looked at the data for all people hospitalized recently in America for CoV2-19 discovered that almost half the cases had minimal or no symptoms and therefore were likely being hospitalized for conditions other than CoV2-19:
The authors of the paper out this week took a different tack. Instead of meticulously looking at why a few hundred patients were admitted to a pair of hospitals, they analyzed the electronic records for nearly 50,000 CoV2-19 hospital admissions at the more than 100 VA hospitals across the country. Then they checked to see whether each patient required supplemental oxygen or had a blood oxygen level below 94 percent. (The latter criterion is based on the National Institutes of Health definition of “severe COVID.”) If either of these conditions was met, the authors classified that patient as having moderate to severe disease REQUIRING hospitalization due to CoV2-19; otherwise, the case was considered mild or asymptomatic and the admission to the hospital was UNLIKELY due to CoV2-19.
The study found that from March, 2020, through early January, 2021, — before vaccination was widespread, and before the Delta variant had arrived — the proportion of patients with mild or asymptomatic disease was 36 %. From mid-January, 2021 through the end of June ,2021, however, that number rose to 48 %. In other words, the study suggests that roughly half of all the hospitalized patients showing up on CoV2-19 data dashboards in 2021 may have been admitted for another reason entirely, or had only a mild presentation of disease.
Additionally, it seems that people who are vaccinated but hospitalized with CoV2-19 are even more likely to have negligible to no symptoms.
The CDC’s Kentucky COVID Research
By Ron Pillman 8/13/21
On Aug 6, 2021 the CDC presented a paper titled “Reduced Risk of Reinfection with SARS-CoV-2 After COVID-19 Vaccination — Kentucky (KY), May–June 2021.”
On the surface, the CDC’s paper appears to be straightforward: they did a statistical analysis of people in Kentucky who previously had COVID-19 and later were reinfected, finding that people who were unvaccinated (1:1000) had a greater than two-times risk of being rei-nfected vs those who were vaccinated (1:2500). This led the CDC to conclude “to reduce their likelihood for future infection, all eligible persons should be offered COVID-19 vaccine, even those with previous SARS-CoV-2 infection.”
The CDC’s findings differ from those of other studies
In the case of the CDC’s KY research, both the findings themselves (unvaccinated patients being rei-nfected at a high rate) and the conclusion (that natural immunity doesn’t last and therefore patients with a prior infection should be vaccinated) are contrary to multiple pieces of other research — specifically a June, 2021, publication from the Cleveland Clinic, a May, 2021, publication from the Washington University of St. Louis, an August, 2021, publication from the University of Barcelona, the April, 2021, SIREN study from the UK, and numerous other studies — unlike the CDC, these groups found that natural immunity after a COVID-19 infection was durable, long-lasting, and provided excellent protection against reinfection.
The CDC’s research is an outlier compared to other studies.
The sample size of the CDC research is small.
In the KY research, the sample size was 246 patients in the study group and 492 in the control. As a points of reference, the Cleveland Clinic study included 52,000+ people and the UK SIREN Study had over 25,000 people.
The research by the Cleveland Clinic, et. al. had no conflict of interest — they were pure science studies. But what about the CDC’s KY Research? While the authors declared no conflict of interest, 3 of the 5 authors were scientists at divisions of the CDC. The CDC is also on record many times speaking in favor of the vaccine, therefore the CDC has a strong investigator bias to want to see an outcome favoring vaccines.
The fact is that the CDC funded the research, the CDC helped conduct the research, and the CDC helped author the publication — did the CDC find the result it wanted?
The CDC research relied on the PCR test, however what constitutes a positive case is DIFFERENT for vaccinated vs unvaccinated patients.
On May 1, 2021, the CDC changed the definition of a ‘positive’ case for a PCR test depending on whether the subject is vaccinated. For unvaccinated subjects, the PCR test continued to use the aggressively high cycle threshold of 40+ and if the test came back positive, the unvaccinated patient was considered to have COVID-19, even if they were asymptomatic. However for vaccinated subjects, a positive case was now defined differently — the cycle threshold was lowered (28 or fewer) which meant far less chance for the test to recognize a viral marker. Additionally, if vaccinated subjects were asymptomatic or had only mild symptoms, the tests were now declared to be negative because under the new rules, a vaccinated subject would only be considered COVID-positive, if they were hospitalized or died.
To compare an unvaccinated asymptomatic subject whose only marker for COVID is a highly upcycled PCR test against vaccinated patients who were hospitalized or died is the very definition of comparing apples and oranges.
In the case of the CDC’s KY Research, the sample period to measure for reinfection was May-Jun, 2021, — this is conveniently after the CDC changed the PCR test definition. Given that the CDC set up a system in which unvaccinated subjects have many more opportunities to be tagged as COVID-positive, it is no surprise that their research ‘discovered’ a higher reinfection rate among unvaccinated subjects.
The CDC recently admitted the PCR Test is inaccurate.
In July, 2021, the CDC revealed that it will be phasing out the Real-Time RT-PCR Diagnostic Panel test because they found that the test is unable to differentiate between SARS-CoV-2 and other influenza viruses. The RT-PCR test is one of the tests used in the KY research. As we say in the pharmaceutical industry. If the CDC knew the RT-PCT/NAAT test was inaccurate in July, why publish research in Aug using bad data?
The CDC’s conclusions were based solely on test results and not hospitalization/death rates.
Remember, the CDC decided in May to change the definition of a “positive” case for vaccinated patients. The given reason was to ‘maximize the quality of the data’ by focusing on what matters — hospitalization and death. If hospitalization and death are the only data that matter for a vaccinated patient, and the mere presence of a positive PCR-test for an asymptomatic vaccinated patient doesn’t matter, the same rules should apply for asymptomatic unvaccinated patients?
The CDC’s research shows vaccinated patients experiencing breakthrough infections at a rate much higher than the CDC has admitted elsewhere.
We have been told that COVID infections and reinfections among vaccinated patients are ‘very rare.’ Real world evidence that recently came out of Massachusetts (74% of recent infections were vaccinated patients), the data from the Israel Minister of Health (40% of recent infections were vaccinated patients, only 1% were patients with a prior infection), and the KY research that we’ve been discussing that showed a 20-27% reinfection rate among vaccinated patients.
The authors themselves pointed out numerous limitations:
1. The inability to prove if the reinfection was new or just residual from a prior infection.
2. Vaccination data of uncertain quality — people in the original unvaccinated patient or control group categories may have gotten the vaccine in another state or in between the study periods and therefore if reinfected they should have been counted as a case in the vaccinated group.
3. This was a retrospective study design from a single state during a short two-month period; therefore, “these findings cannot be used to infer causation.” This small analysis should not be used to make large-scale assumptions or public health decisions.
The CDC’s seems to have a research agenda — (the Mannequin trial on face masks, their obscuring ‘unpublished data’ from a peer-rejected paper from India, etc.): why have confidence -in the KY?
The conclusion: the CDC appears to have an a predis[ostion to present a particular point of view.
Study of T cells from COVID-19 convalescents guides vaccine strategies
by The Korea Advanced Institute of Science and Technology (KAIST) 7/5/21
Overview of the SARS-CoV-2-specific immune response kinetics. Memory T cells are maintained after recovery from COVID-19 with the generation of stem cell-like memory T cell. Credit: The Korea Advanced Institute of Science and Technology (KAIST)
A KAIST immunology research team found that most convalescent patients of COVID-19 develop and maintain T cell memory for over 10 months regardless of the severity of their symptoms. In addition, memory T cells proliferate rapidly after encountering their cognate antigen and accomplish their multifunctional roles. This study provides new insights for effective vaccine strategies against COVID-19, considering the self-renewal capacity and multipotency of memory T cells.
COVID-19 is a disease caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection. When patients recover from COVID-19, SARS-CoV-2-specific adaptive immune memory is developed. The adaptive immune system consists of two principal components: B cells that produce antibodies and T cells that eliminate infected cells. The current results suggest that the protective immune function of memory T cells will be implemented upon re-exposure to SARS-CoV-2.
Recently, the role of memory T cells against SARS-CoV-2 has been gaining attention as neutralizing antibodies wane after recovery. Although memory T cells cannot prevent the infection itself, they play a central role in preventing the severe progression of COVID-19. However, the longevity and functional maintenance of SARS-CoV-2-specific memory T cells remain unknown.
Professor Eui-Cheol Shin and his collaborators investigated the characteristics and functions of stem cell-like memory T cells, which are expected to play a crucial role in long-term immunity. Researchers analyzed the generation of stem cell-like memory T cells and multi-cytokine producing polyfunctional memory T cells, using cutting-edge immunological techniques.
This research is significant in that revealing the long-term immunity of COVID-19 convalescent patients provides an indicator regarding the long-term persistence of T cell immunity, one of the main goals of future vaccine development, as well as evaluating the long-term efficacy of currently available COVID-19 vaccines.
The research team is presently conducting a follow-up study to identify the memory T cell formation and functional characteristics of those who received COVID-19 vaccines, and to understand the immunological effect of COVID-19 vaccines by comparing the characteristics of memory T cells from vaccinated individuals with those of COVID-19 convalescent patients.
Ph.D. candidate Jae Hyung Jung and Dr. Min-Seok Rha, a clinical fellow at Yonsei Severance Hospital, who led the study together explained, “Our analysis will enhance the understanding of COVID-19 immunity and establish an index for COVID-19 vaccine-induced memory T cells.”
“This study is the world’s longest longitudinal study on differentiation and functions of memory T cells among COVID-19 convalescent patients. The research on the temporal dynamics of immune responses has laid the groundwork for building a strategy for next-generation vaccine development,” Professor Shin added. This work was supported by the Samsung Science and Technology Foundation and KAIST, and was published in Nature Communications on June 30.
Vaccines charge up natural immunity against SARS-CoV-2
More information: Jae Hyung Jung et al, SARS-CoV-2-specific T cell memory is sustained in COVID-19 convalescent patients for 10 months with successful development of stem cell-like memory T cells, Nature Communications (2021). DOI: 10.1038/s41467-021-24377-1
Thisone has facts and a political tilt:
Research: Natural Covid Infection Creates Immunity: Vaccines ….
Testing “positive” for the Wuhan coronavirus (Covid-19) and later recovering from it, assuming you got sick at all, provides lasting immunity” that seems to be about the equal of vaccination (edited by HRS)
The latest figures presented to the Israeli Health Ministry show that CoV2-19 immunizations do not always provide a lasting protection against disease.
According to the science, those who already had the CoV2-19 infection at some point throughout the past year were immune to Israel’s recent “outbreak,” while those who got the vaccination more often became ill and died from the latest (Delta) “wave” of the virus.
More than 7,700 “cases” of CoV2-19 were detected in Israel during the latest variant surge, and only 72 of them, or less than one percent, occurred in people who had previously contracted CoV2-19. More than 3,000 patients, or about 40 percent of new cases, meanwhile, were detected in people who had gotten injected in compliance with the government’s requirement.
“With a total of 835,792 Israelis known to have recovered from the virus, the 72 instances of reinfection amount to 0.0086% of people who were already infected with COVID,” reported Israel National News.
“By contrast, Israelis who were vaccinated were 6.72 times more likely to get infected after the vaccination than after natural infection, with over 3,000 of the 5,193,499, or 0.0578%, of Israelis who were vaccinated getting infected in the latest wave.”
The news has, as you might expect, greatly divided Israel’s Health Ministry experts have put their full and undivided faith in the immunizations, and others who are more skeptical.
Governments seem to be unable to process a world in which chemical mRNA injections are anything other than the near perfect cure for CoV2-19
“T cells can mount attacks against many SARS-CoV-2 targets-even on new virus variant
MedicalXpress1/28/21
La Jolla Institute for Immunology (LJI) suggests that T cells try to fight SARS-CoV-2 by targeting a broad range of sites on the virus—beyond the key sites on the virus’s spike protein. By attacking the virus from many angles, the body has the tools to recognize different SARS-CoV-2 variants.
Published 1/27/21 in Cell Report Medicine: proteins on SARS-CoV-2 stimulate the strongest responses from the immune system’s “helper” CD4+ T cells and “killer” CD8+ T cells says LJI Professor Alessandro Sette, Dr. Biol. Sci. and LJI Instructor Alba Grifoni, PhD.
Some people have strong immune responses and do well. Others have disjointed immune responses and are more likely to end up in the hospital. By re-scrambling genetic material, it can make T cells that respond to a huge range of targets, or epitopes, on a pathogen. Some T cell responses will be stronger against some epitopes than others. Researchers call the targets that prompt a strong immune cells response “immunodominant.”
They examined T cells from 100 people who had recovered from SARS-CoV-2 infection. Not all parts of the virus induce the same strong immune response in everyone. In fact, T cells can recognize dozens of epitopes on SARS-CoV-2, and these immunodominant sites change from person to person. Each study participant had the ability to recognize about 17 CD8+ T cells epitopes and 19 CD4+ T cell epitopes.
Without a strong CD4+ T cell response, however, people may be slow to mount the kind of neutralizing immune response that quickly wipes out the virus. Luckily, the broad immune response can recognize sites other than the receptor binding domain. By targeting many vulnerable sites on the spike protein, the immune system would still be able to fight infection, even if some sites on the virus change due to mutations.
The immune response is broad enough to compensate for that.
Since the announcement of the fast-spreading UK variant of SARS-CoV-2 (called SARS-CoV-2 VUI 202012/01), the researchers have compared the mutated sites on that virus to the epitopes they found. Sette notes that the mutations described in the UK variant for the spike protein affect only 8% of the epitopes recognized by CD4+ T cells, while 92% of the responses is conserved.
Sette emphasized that the new study is the results of months of long hours and international collaboration between labs at LJI; the U of California, San Diego; and researcher’s at Australia’s Murdoch U.”
Comment on this paper
Distinct antibody and memory B cell responses in SARS-CoV-2 naïve and recovered individuals following mRNA vaccination
Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection
View ORCID ProfileShuo Feng, Daniel J. Phillips, Thomas White, Homesh Sayal, View ORCID ProfileParvinder K. Aley, View ORCID ProfileSagida Bibi, View ORCID ProfileChristina Dold, View ORCID ProfileMichelle Fuskova, View ORCID ProfileSarah C. Gilbert, Ian Hirsch, View ORCID ProfileHolly E. Humphries, View ORCID ProfileBrett Jepson, Elizabeth J. Kelly, Emma Plested, Kathryn Shoemaker, Kelly M. Thomas, Johan Vekemans, Tonya L. Villafana, View ORCID ProfileTeresa Lambe, View ORCID ProfileAndrew J Pollard, View ORCID ProfileMerryn Voysey, the Oxford COVID Vaccine Trial Group
doi: https://doi.org/10.1101/2021.06.21.21258528
This article is a preprint and has not been peer-reviewed [what does this mean?]. It reports new medical research that has yet to be evaluated and so should not be used to guide clinical practice.
Background Although 6 COVID-19 vaccines have been approved by the World Health Organisation as of 16th June 2021, global supply remains limited. An understanding of the immune response associated with protection could facilitate rapid licensure of new vaccines.
Methods Data from a randomised efficacy trial of ChAdOx1 nCoV-19 (AZD1222) vaccine in the UK was analysed to determine the antibody levels associated with protection against SARS-CoV-2. Anti-spike and anti-RBD IgG by multiplex immunoassay, pseudovirus and live neutralising antibody at 28 days after the second dose were measured in infected and non-infected vaccine recipients. Weighted generalised additive models for binary data were applied to symptomatic and asymptomatic SARS-CoV-2 infection data from ChAdOx1 nCoV-19 recipients. Cubic spline smoothed log antibody levels, and weights were applied to account for potential selection bias in sample processing. Models were adjusted for baseline risk of exposure to SARS-CoV-2 infection.
Results Higher levels of all immune markers were correlated with a reduced risk of symptomatic infection. Vaccine efficacy of 80% against primary symptomatic COVID-19 was achieved with an antibody level of 40923 (95% CI: 16748, 125017) and 63383 (95% CI: 16903, not computed (NC)) for anti-spike and anti-RBD, and 185 (95% CI: NC, NC) and 247 (95% CI: 101, NC) for pseudo- and live-neutralisation assays respectively. Antibody responses did not correlate with overall protection against asymptomatic infection.
Conclusions Correlates of protection can be used to bridge to new populations using validated assays. The data can be used to extrapolate efficacy estimates for new vaccines where large efficacy trials cannot be conducted. More work is needed to assess correlates for emerging variants.
R Shinnakasu 10/2021has identified how to make vaccines to ALL coronaviruses by hiding the usual superficial of the RBD/receptor binding domain site that creates the vaccine immunity, allowing immunity to develop the shared core portion of all corona viruses: “ … The receptor-binding domain of the spike protein consists of two parts: a “core” region that is very similar in all coronaviruses, and a more specialized “head” region that mediates binding to ACE2.
Antibodies that recognize the head region of the spike receptor-binding domain can block the entry of SARS-CoV-2 into cells but offer little protection against other coronaviruses, such as the SARS-CoV-1 virus responsible for the severe acute respiratory syndrome outbreak of 2002. Antibodies that recognize the core region of the spike receptor-binding domain, in contrast, can prevent the entry of various coronaviruses into human cells. Unfortunately, however, individuals exposed to the viral spike protein tend to produce lots of antibodies against the head region but few, if any, antibodies that recognize the core region. Ryo Shinnakasu et al, Glycan engineering of the SARS-CoV-2 receptor-binding domain elicits cross-neutralizing antibodies for SARS-related viruses, Journal of Experimental Medicine (2021). DOI: 10.1084/jem.20211003
Rishi R. Goel1,2,†,et al
Science Immunology 15 Apr 2021:
Vol. 6, Issue 58, eabi6950
DOI: 10.1126/sciimmunol.abi6950
Abstract
Novel mRNA vaccines for SARS-CoV-2 have been authorized for emergency use. Despite their efficacy in clinical trials, data on mRNA vaccine-induced immune responses are mostly limited to serological analyses. Here, we interrogated antibody and antigen-specific memory B cells over time in 33 SARS-CoV-2 naïve and 11 SARS-CoV-2 recovered subjects. SARS-CoV-2 naïve individuals required both vaccine doses for optimal increases in antibodies, particularly for neutralizing titers against the B.1.351 variant. Memory B cells specific for full-length spike protein and the spike receptor binding domain (RBD) were also efficiently primed by mRNA vaccination and detectable in all SARS-CoV-2 naive subjects after the second vaccine dose, though the memory B cell response declined slightly with age. In SARS-CoV-2 recovered individuals, antibody and memory B cell responses were significantly boosted after the first vaccine dose; however, there was no increase in circulating antibodies, neutralizing titers, or antigen-specific memory B cells after the second dose. This robust boosting after the first vaccine dose strongly correlated with levels of pre-existing memory B cells in recovered individuals, identifying a key role for memory B cells in mounting recall responses to SARS-CoV-2 antigens. Together, our data demonstrated robust serological and cellular priming by mRNA vaccines and revealed distinct responses based on prior SARS-CoV-2 exposure, whereby COVID-19 recovered subjects may only require a single vaccine dose to achieve peak antibody and memory B cell responses. These findings also highlight the utility of defining cellular responses in addition to serologies and may inform SARS-CoV-2 vaccine distribution in a resource-limited setting.
The humoral immune response to infection or vaccination results in two major outcomes: the production of antibodies by antibody secreting cells (ASCs) that can provide rapid serological immunity, and the generation of long-lived memory B cells capable of mounting recall responses (6, 7). If circulating antibodies fail to confer protection to a future exposure, memory B cells drive the recall response by producing new antibodies through forming new ASCs or re-entering germinal centers for additional rounds of somatic hypermutation (8, 9). In the context of acute SARS-CoV-2 infection, immunological memory in the form of antibodies and memory B cells are durable for over 8 months post-symptom onset (10–14). However, studies on vaccinated individuals have largely focused on measuring binding and/or neutralizing antibodies as primary endpoints (15–17), and the induction of memory B cells by mRNA vaccines remains poorly understood. Although antibodies are a central component of vaccine efficacy, memory B cells may be important for long-term protection, responses to subsequent infection, and the ability to respond to emerging variant strains (18). Furthermore, it is unclear how memory B cell responses relate to serological responses for novel SARS-CoV-2 mRNA vaccines, and how memory B cell responses differ after vaccination in subjects who previously experienced SARS-CoV-2 infection compared to those who are SARS-CoV-2 naïve.
A related question is whether individuals who experienced prior SARS-CoV-2 infection require a second dose of mRNA vaccine. As these individuals have already generated a primary immune response to SARS-CoV-2 during their natural infection, it is possible that a single dose of vaccine could be sufficient to boost antibody and memory B cell responses. This question is particularly relevant in settings of limited vaccine supply and challenging vaccine deployment (19). Indeed, several recent studies have indicated that antibody responses can be robustly induced in SARS-CoV-2 experienced individuals, consistent with an anamnestic response (20–23). Although one study suggests that memory B cells might also be boosted after a single vaccine dose (24), it remains unclear how memory B cell responses are affected by the second dose of mRNA vaccine in SARS-CoV-2 naïve versus recovered individuals. These key gaps in our understanding require longitudinal analysis of antibodies together with memory B cell responses after the first and second dose of mRNA vaccine in SARS-CoV-2 naïve and experienced subjects.
Here, we established a longitudinal cohort of SARS-CoV-2 naïve and SARS-CoV-2 recovered individuals who received SARS-CoV-2 mRNA vaccines. From these longitudinal samples, we assessed both circulating antibodies and antigen-specific memory B cells over the course of first and second immunization. We also compared vaccine responses with demographic and clinical metadata, including age and side effects. These data offer new insights into the B cell response to SARS-CoV-2 mRNA vaccines.
RESULTS
For this study, we recruited 44 healthy individuals (i.e., no self-reported chronic health conditions) who received SARS-CoV-2 mRNA vaccines (Pfizer BNT162b2 or Moderna mRNA-1273) at the University of Pennsylvania Health System. Full cohort information is described in figure S1. Of this cohort, 11 individuals had a prior SARS-CoV-2 infection, ranging from 65 to 275 days prior to vaccination. Peripheral blood samples were collected for immunological analysis at 4 key timepoints (Fig. 1A): pre-vaccine baseline (timepoint 1), 2 weeks following the first dose (timepoint 2), the day of second dose (timepoint 3), and 1 week following the second dose (timepoint 4). This study design allowed us to investigate the kinetics of immune responses following both primary and secondary immunizations.
Fig. 1
Antibody responses following mRNA vaccination in SARS-CoV-2 naïve and recovered individuals. A) UPenn Immune Health COVID vaccine study design. B) Concentration of anti-spike and anti-RBD IgG antibodies in vaccinated individuals over time. C) Focus reduction neutralization titer 50% (FRNT50) of vaccine-induced sera against pseudotyped virus expressing SARS-CoV-2 D614G (wild-type) or B.1.351 (South African) variant spike protein. D) Paired analysis of neutralization titers against D614G and B.1.351 in vaccine-induced sera at baseline (timepoint 1), pre-boost (timepoint 2), and post-boost (timepoint 4). E) Bivariate analysis of total anti-spike and anti-RBD binding antibodies with pseudovirus neutralization titers against D614G and B.1.351. Associations between total antibody levels and neutralizing ability were calculated using Spearman rank correlation and are shown with linear trend lines. Dotted lines indicate the limit of detection (LOD) for the assay. Statistics were calculated using unpaired Wilcoxon test (comparisons between timepoints and comparisons between naïve and recovered) or paired Wilcoxon test (comparisons between D614G and B.1.351) with Holm correction for multiple comparisons. Blue and red values indicate statistical comparisons within naïve or recovered groups. Black values indicate statistical comparisons between naïve or recovered groups.
Antibody responses to SARS-CoV-2 mRNA vaccination
We first measured circulating antibody responses in longitudinal serum samples by ELISA. At baseline, SARS-CoV-2 naïve individuals had undetectable levels of IgG antibodies specific for either full-length spike protein or the spike receptor binding domain (RBD) (Fig. 1B). Primary vaccination induced a significant increase in SARS-CoV-2-specific antibodies, that was further enhanced by the booster dose (Fig. 1B). In contrast, all SARS-CoV-2 recovered individuals had detectable levels of anti-spike and anti-RBD IgG at baseline, and these antibody responses were significantly increased after the first dose of vaccine (Fig. 1B). However, in SARS-CoV-2 recovered subjects, there was no additional increase in antibody levels following the second vaccine dose (Fig. 1B). Notably, the levels of anti-RBD IgG were similar in the SARS-CoV-2 naïve and SARS-CoV-2 recovered individuals at 1 week post-boost (timepoint 4) (Fig. 1B).
In addition to total spike- and RBD-binding antibody, we further assessed antibody function using a pseudovirus neutralization assay. Specifically, we tested the ability of vaccine-induced sera to neutralize pseudotyped virus expressing either the D614G (the initial dominant strain at the time of the study) spike protein or the B.1.351 variant (commonly referred to as the South African variant) spike protein. SARS-CoV-2 naïve individuals had a moderate response to primary immunization with ~50% of participants developing detectable levels of neutralizing antibodies against D614G two weeks post-primary (Fig. 1C-D). In contrast, primary immunization was largely ineffective to induce functional antibodies against the B.1.351 variant with only 4/25 individuals developing neutralizing titers above limit of detection over the same time frame (Fig. 1C-D). Neutralizing titers were significantly increased after the second dose in SARS-CoV-2 naïve individuals, with all participants achieving neutralization against D614G and 26/27 achieving detectable neutralization against B.1.351 at 7 days post-boost (Fig. 1C-D). Consistent with anti-spike and anti-RBD antibody levels, SARS-CoV-2 experienced individuals had a robust increase in neutralizing antibodies following primary immunization, with no further increase in neutralization titers against D614G and B.1.351 after the second dose (Fig. 1C). Interestingly, the first dose of vaccine also appeared to resolve baseline differences in neutralization between D614G and B.1.351 in this group (Fig. 1D).
Based on these data, we quantified the relationship between total antibody levels and neutralization ability in SARS-CoV-2 naïve individuals to assess the relative quality of antibodies induced by the first and second dose of mRNA vaccine. Before the second dose, anti-spike antibodies were only moderately correlated with neutralizing titers against D614G, with further dropoff for the B.1.351 variant (Fig. 1E). Pre-boost anti-RBD antibodies were more predictive of neutralization titers against D614G and B.1.351 (Fig. 1E) than anti-spike antibodies. Both anti-spike and anti-RBD antibodies correlated more strongly with neutralizing titers against D614G and B.1.351 after the second dose (Fig. 1E), indicating a marked improvement in the quality of the antibody response. Together, these data supported the importance of a 2 dose-regimen for effective antibody responses, especially against the B.1.351 variant, in SARS-CoV-2 naïve individuals. Conversely, a single dose of vaccine was able to achieve highly effective antibody responses in SARS-CoV-2 recovered individuals with no further improvement post-boost.
Memory B Cell responses to SARS-CoV-2 mRNA vaccination
We next asked how mRNA vaccination impacted the responses of memory B cells specific for SARS-CoV-2. To address this question, we developed a flow cytometric assay using a combination of fluorescently labeled antigens as probes to track the induction of virus-specific memory B cells in longitudinal PBMC samples (figure S2A) (11, 13, 25). Analysis of bulk B cell populations revealed no change in the frequency of naïve, non-naïve, or memory B cells across the timecourse of vaccination, or between SARS-CoV-2 naïve and recovered individuals (figure S2B), highlighting the stability of the overall B cell compartment.
Despite a stable frequency of total memory B cells, there were marked changes in SARS-CoV-2 antigen-specific B cell populations in response to vaccination. Consistent with the antibody data, SARS-CoV-2 naïve individuals had minimal spike-specific memory B cells at baseline, whereas SARS-CoV-2 recovered individuals had a significant population of spike-specific memory B cells ranging from ~0.15-0.8% of total memory B cells (Fig. 2A-B). Memory B cells targeting the spike RBD followed a similar trend and the frequency of these antigen-specific memory B cells was comparable to a separate cohort of non-vaccinated SARS-CoV-2 recovered donors (Fig. 2A-B). After primary immunization, SARS-CoV-2 naïve individuals had a significant increase in spike-specific and RBD-specific memory B cells over baseline (Fig. 2B). These memory B cells were also significantly boosted after adminstration of the second vaccine dose, approaching the levels of memory B cells observed in non-vaccinated SARS-CoV-2 recovered donors (Fig. 2B). In contrast, SARS-CoV-2 recovered individuals had a robust expansion of spike- and RBD-specific memory B cells following primary immunization, but had no additional boosting after the second vaccine dose (Fig. 2B). As a control we also examined the frequency of influenza hemagglutinin (HA)-specific memory B cells in both SARS-CoV-2 naïve and recovered individuals following SARS-CoV-2 vaccination. The frequency of these antigen-unrelated memory B cells remained stable throughout the mRNA vaccination timecourse (Fig. 2B), confirming the specificity of this memory B cell assay. Together, these results demonstrated robust induction of SARS-CoV-2-specific memory B cells by two doses of mRNA vaccine in SARS-CoV-2 naïve subjects. In contrast, a single dose of mRNA vaccine amplified pre-existing antigen-specific memory B cells in SARS-CoV-2 recovered subjects, with no additional quantitative benefit after the second vaccine dose.
Fig. 2
Antigen-specific memory B cell responses following mRNA vaccination in SARS-CoV-2 naïve and recovered individuals. A) Gating strategy and representative plots for flow cytometric analysis of SARS-CoV-2-specific B cells. B) Frequency of spike+, spike+/RBD+, and HA+ memory B cells over time in vaccinated individuals. Data are represented as frequency of antigen-specific cells in the total memory B cell compartment. C) Frequency of IgG and IgM isotypes over time in the antigen-specific memory B cell compartments. D) Frequency of RBD+ memory B cells over time in vaccinated individuals, as a percentage of spike+ memory B cells. E) Somatic hypermutation (SHM) status of spike+ memory B cell clones over time in SARS-CoV-2 recovered individuals. Data are represented as percent of VH-gene nucleotides that are mutated. F) SHM of productive spike-binding clones sampled at timepoint 1 which were also found in at least one other timepoint. Clones with fewer than 10 copies in each patient were excluded. G) Clonal evolution of spike-binding memory B cell lineages that were present prior to vaccination in a recovered individual. For representative lineages, numbers refer to mutations compared to the preceding vertical node. Colors indicate timepoint, black dots indicate inferred nodes, and size is proportional to sequence copy number; GL = germline sequence. All panels: Dotted lines indicate the mean at baseline. RD = non-vaccinated, SARS-CoV-2 recovered donors. Statistics were calculated using unpaired Wilcoxon test (comparisons between timepoints and comparisons between naïve and recovered) with Holm correction for multiple comparisons. Blue and red values indicate statistical comparisons within naïve or recovered groups. Black values indicate statistical comparisons between naïve or recovered groups.
We further analyzed the phenotype of SARS-CoV-2 specific memory B cells. On day 15 after primary immunization, ~25-30% of spike-specific memory B cells were IgG+ and ~40-50% were IgM+ in SARS-CoV-2 naïve individuals (Fig. 2C). The frequency of IgG+ memory B cells increased to >50% following the second dose of vaccine in these subjects (Fig. 2C), consistent with a qualitative improvement in memory B cells after the boost. Conversely, in SARS-CoV-2 recovered individuals, ~60-70% of spike-specific memory B cells detected prior to vaccination were IgG+ (Fig. 2C). Although the frequency of IgG+ memory B cells increased slightly to ~75% following the first dose of vaccine, further increases were not observed after the second immunization (Fig. 2C-D). A similar pattern of IgG frequency was observed for RBD-specific memory B cells (Fig. 2C). In addition, the fraction of spike-specific memory B cells that recognized RBD remained stable over time in SARS-CoV-2 recovered individuals. In SARS-CoV-2 naïve subjects, the fraction of the overall spike-specific memory B cell response that was focused on RBD increased over time, becoming equivalent to that observed in SARS-CoV-2 recovered individuals after the second vaccine dose (Fig. 2D). Overall, these data indicated a qualitative benefit to the virus-specific memory B cell response following both doses of vaccine in SARS-CoV-2 naïve individuals, and qualitative improvement following the first but not the second vaccine dose in SARS-CoV-2 recovered subjects.
Finally, we sorted spike+ memory B cells from 5 recovered donors at baseline (timepoint 1), post-primary (timepoint 2), and post-boost (timepoint 4) for B cell receptor (BCR) sequencing to further evaluate potential changes in the memory B cell response induced by vaccination. Somatic hypermutation (SHM) is a process of DNA point hypermutation that occurs in immunoglobulin variable gene sequences and usually accompanies T cell-dependent B cell responses within germinal centers (26). Accordingly, SHM is a frequently used marker for the evaluation of immune memory (27). Here, SHM was calculated as the average percentage of mutated VH-gene nucleotides in each clone, counting each clone only once. Full sequencing information, including the number of clones identified for each sample, is listed in table S3. Mutational analysis of total spike-binding memory clones revealed a modest shift toward higher SHM at the post-primary and post-boost timepoint in some individuals (Fig. 2E); however, there was no clear pattern across the 5 individuals measured. To determine if SHM changed within pre-existing spike-binding clones, we next looked for high-copy spike-binding clones that were shared between the baseline timepoint and at least one other timepoint. These clones, which were present before the first vaccine dose, presumably arose during the initial infection with SARS-CoV-2. Subject 29 was not included in this analysis because there was only one clone that met the copy number cutoff. SHM levels in the overlapping clones did not increase after vaccination (Fig. 2F). The stability of SHM could also be seen within lineage trees for subject 20, who had the largest number of clones sampled. Specifically, the nodes (sequence variants) within lineages exhibited mixing between the timepoints, and where they were separate, they were not consistently found at higher frequencies in parts of the trees with higher levels of SHM (Fig. 2G, figure S3). These data suggested that pre-existing spike-specific memory clones in SARS-CoV-2 recovered individuals did not increase their level of SHM in response to either dose of vaccine.
Demographic and clinical factors associate with B cell responses to SARS-CoV-2 mRNA vaccination
In addition to prior SARS-CoV-2 exposure, we also investigated associations between other demographic and clinical metadata with vaccine-induced B cell responses. Several previous studies have reported a negative association between age and vaccine-induced antibody titers after a single dose of mRNA vaccines (28, 29). We therefore investigated potential relationships between sex or age and B cell responses after one or two doses of vaccine. In our cohort of SARS-CoV-2 naïve vaccinees, there were no associations between sex and antibody or memory B cell responses (Fig. 3A, 3B). While there was no association between age and anti-spike IgG after the first immunization (i.e., pre-boost), there was a trend toward a negative relationship between age and pre-boost RBD-specific IgG (Fig. 3C). Antibody for both spike and RBD had a similarly negative, but statistically insignificant, correlation with age after the second vaccine dose (Fig. 3C). However, there was a clear negative correlation between the post-boost frequency of antigen-specific memory B cells and age (Fig. 3D). Although this relationship represented weaker induction of memory B cells with older age, all age groups still displayed an increase in the frequency of SARS-CoV-2 specific memory B cells compared to pre-vaccine baseline (Fig. 3C-D). There was also no change in the frequency of total memory B cells by sex or age, indicating the antigen-specific nature of this effect (figure S4). Although our cohort is not extensively enriched in those over 50 years old and does not directly address elderly vaccinees, these data pointed to potentially relevant age-related changes in immune response to vaccination.
Fig. 3
Association of age and side-effects with B cell responses following mRNA vaccination. A, C) Concentration of anti-spike and anti-RBD IgG antibodies over time compared with sex (A) and age (C) in SARS-CoV-2 naïve individuals. Dotted lines indicate the limit of detection for the assay. B, D) Frequency of spike+ and spike+/RBD+ memory B cells over time compared with sex (B) and age (D) in SARS-CoV-2 naïve individuals. Dotted lines indicate the mean frequency of cells at baseline. Pre-boost indicates samples collected at timepoint 2 (~15 days post-primary vaccination). Post-boost indicates samples collected at timepoint 4 (~7 days post-secondary vaccination). Statistics for sex were calculated using Wilcoxon test. Associations with age were calculated using Spearman rank correlation and are shown with linear trend lines. E) Concentration of anti-spike and anti-RBD IgG antibodies and frequency of spike+ and spike+/RBD+ memory B cells at the post-boost timepoint compared with self-reported side effects after the second dose. Reactogenicity was categorized into either no/local symptoms or systemic symptoms. F) Multivariable linear regression between antibody or memory B cell responses and side effects, controlling for sex and age. Data are represented as estimated regression coefficients with a 95% confidence interval.
An additional question is whether vaccine-induced side effects have any relationship to immune responses (20). We addressed this question by comparing vaccine-induced antibody and memory B cell responses in subjects with or without self-reported systemic side effects (i.e., fever, chills, headache, fatigue, myalgia; figure S1C). In SARS-CoV-2 naïve vaccinees with systemic side-effects following the second dose, there was a trend toward an increase in antibody responses at the post-boost timepoint (Fig. 3E). Such a trend was not observed for the memory B cell response (Fig. 3E). We further investigated the potential association between reactogenicity and increased antibody response using a multivariate regression to control for the effects of sex and age. This multivariate analysis similarly revealed a positive association of systemic side effects with anti-spike and anti-RBD antibody levels 7 days after the booster immunization (Fig. 3F). Although these data only represent a statistical trend (p=0.051), they do provoke questions about potential relationships between early vaccine-induced inflammation and the induction of antibody responses that should be addressed in future studies.
Relationships between antibody and memory B cell responses to SARS-CoV-2 mRNA vaccination
Finally, we investigated the potential relationships between antibody and memory B cell responses. To address this question, we first performed hierarchical clustering of vaccine-induced B cell responses in SARS-CoV-2 naïve subjects. As expected, post-boost (timepoint 4) samples clustered away from the earlier timepoints, with some sub-grouping of patients based on the relative magnitude of antibody and memory B cell responses (Fig. 4A). Hierarchical clustering of the different readouts of antigen-specific humoral immunity also revealed that antibodies and memory B cells clustered separately (Fig. 4A). We next performed a principal component analysis (PCA) of post-boost B cell responses. Antibody and memory B cell measurements had distinct contributions to the first 2 principal components, with total binding antibodies and neutralizing titers primarily contributing to dimension 1 (Dim1) and memory cells primarily contributing to dimension 2 (Dim2) (Fig. 4B). Based on these data, we further examined the relationship between circulating antibody responses and corresponding memory B cell responses after two doses of vaccine in a bivariate analysis. Despite strong induction of both spike- and RBD-specific antibody and memory B cells in these subjects, there was no association between the levels of post-boost antibodies and B cell memory (Fig. 4C), indicating that short-term serological responses and memory B cell responses may be distinct immunological features of response to mRNA vaccination. Similarly, pre-vaccine baseline antibody levels did not correlate with baseline memory B cell frequencies in SARS-CoV-2 recovered individuals (figure S5A). We next asked which measure of humoral immunity predicted antibody recall responses post-vaccination. Interestingly, the baseline levels of SARS-CoV-2-specific antibody correlated with the level of anti-spike, but not anti-RBD antibody achieved after primary vaccine in SARS-CoV-2 recovered donors (figure S5B). However, the baseline frequency of antigen-specific memory B cells strongly correlated with post-primary vaccination antibody levels for both spike and RBD (Fig. 4D), consistent with the notion that these pre-vaccination memory B cells are major contributors to the SARS-CoV-2 antibody recall response. These data highlight the importance of measuring antigen-specific memory B cells in addition to serologic antibody evaluation as an immunological correlate of vaccine-induced immunity.
Fig. 4
Antigen-specific memory B cells were a distinct measure of vaccine efficacy and correlated to antibody recall responses. A) Heatmap and hierarchical clustering of vaccine-induced antibody and memory B cell responses. B) Principal component analysis and biplot of vaccine-induced antibody and memory B cell responses. C) Association of post-boost (timepoint 4) antibody levels with post-boost (timepoint 4) antigen-specific memory B cell frequencies in SARS-CoV-2 naïve individuals. D) Association of baseline (timepoint 1) antigen-specific memory B cell frequencies with post-primary vaccination (timepoint 2) antibody levels in SARS-CoV-2 recovered individuals. Illustrations in (C) and (D) represent the corresponding immune relationship. Associations between immunological parameters were calculated using Spearman rank correlation and are shown with non-parametric trend lines (Theil-Sen estimator).
Overall, we tracked antibody and antigen-specific memory B cells over time following SARS-CoV-2 mRNA vaccination and documented robust priming of antibody as well as memory B cell responses (Fig. 5A). Our analysis revealed key differences in vaccine-induced immune response between SARS-CoV-2 naïve and recovered subjects after the first versus second dose of vaccine. (Fig. 5B). SARS-CoV-2 naive individuals required two doses of vaccine to achieve optimal priming of antibodies, including neutralizing antibodies to the B.1.351 strain and memory B cells. In contrast, SARS-CoV-2 recovered subjects may only require a single vaccine dose to achieve peak antibody and memory B cell responses. We also revealed age-related differences in vaccine-induction of immune responses (Fig. 5C) and highlighted the importance of memory B cells in mounting recall antibodies in SARS-CoV-2 recovered subjects (Fig. 5D).
Fig. 5
Study summary and key findings). A) Cohort design and objectives. Longitudinal samples were collected from SARS-CoV-2 naïve and recovered individuals and measured for both antibodies and memory B cells. B) Distinct patterns of antibody and memory B cell responses to mRNA vaccination in SARS-CoV-2 naïve and recovered individuals. C) Age-associated differences in antibody and memory B cell responses to mRNA vaccination. D) Baseline memory B cells in SARS-CoV-2 recovered individuals contribute to recall responses following mRNA vaccination.
DISCUSSION
Here, we demonstrated that mRNA vaccines to SARS-CoV-2 induced robust antibody and memory B cell responses to full-length spike and the RBD. These results are encouraging for both short- and long-term vaccine efficacy and add to our understanding of SARS-CoV-2 mRNA vaccine-induced immune responses in several ways. First, our serological data are consistent with several other recent studies (20, 21, 23, 24, 28, 29) indicating robust boosting of antibody responses in SARS-CoV-2 recovered subjects after the first vaccine dose, but little benefit to antibody levels after the second vaccine dose. This finding was also reflected in the observation that neutralizing titers against both D614G and the B.1.351 South African variant reached a peak after the first dose in recovered subjects. Moreover, we found a similar effect for virus-specific memory B cells, identifying a quantitative and qualitative plateau in vaccine-induced memory B cells in COVID-19 recovered subjects after the first dose of vaccine with little additional change to the memory B cell response following booster vaccination. These data suggest that only a single vaccine dose in individuals confirmed to have previously been infected with SARS-CoV-2 may be enough to induce antibody and memory B cell responses.
The data presented document key differences in immune responses associated with vaccine efficacy in SARS-CoV-2 naïve versus SARS-CoV-2 recovered individuals. However, with a study of this size designed for deep immunological analysis, it was not possible to directly address protection or true vaccine efficacy. Accordingly, larger-scale clinical studies would be necessary to fully examine the question of a one or two dose regimen in SARS-CoV-2 recovered individuals. Our cohort also consisted of individuals who were not hospitalized during their SARS-CoV-2 infections, and it may be necessary to address this question of one versus two doses of vaccine in individuals who experienced more severe COVID-19. Moreover, there may be practical challenges to identifying SARS-CoV-2 recovered individuals based on self-reported infection or laboratory confirmed tests such as RT-PCR or serology. Despite these limitations, the robust boosting of both antibody and memory B cells in these subjects after one dose may have implications for vaccine distribution in settings where supply is limited.
An additional question is whether the second vacccine dose in recovered individuals has other immunological effects not reflected in overall antibody titers or memory B cell frequency and phenotype. Given the relatively short timeframe of this study, future studies will be necessary to evalute durability of immune responses in these subjects and investigate potential differences in long-term immunological memory. Our data indicates that pre-formed spike-binding memory B cell clones that were resampled at multiple time points did not have obvious increases in SHM, suggesting that the B cell clones boosted by mRNA vaccination in SARS-CoV-2 recovered individuals have stable SHM profiles. However, these analyses were only performed on a small number of individuals and samples were limited to only the first few weeks following vaccination. Thus, it will be important to determine if these clones evolve and undergo further SHM over time as occurs after natural SARS-CoV-2 infection (9, 30, 31). Even small changes in SHM may be biologically relevant, as somatically mutated clones can exhibit higher degrees of cross-protection against different mutant strains of the virus (30). It is also possible that other post-germinal center clones emerge later in the memory phase. Lastly, it is possible that booster vaccination has some beneficial effects on virus-specific T cell responses in SARS-CoV-2 recovered individuals. Given the capacity of mRNA vaccines to induce CD4+ T cell responses (32), this topic merits further investigation.
In contrast to SARS-CoV-2 recovered subjects, SARS-CoV-2 naïve individuals demonstrated considerable benefit to antibody and memory B cell responses from the second dose of mRNA vaccine. It is possible that some of this benefit would occur over time in the absence of a booster vaccination; however, the spike- and RBD-specific antibody levels appeared to plateau between the first and second doses of vaccine before increasing again following booster vaccination. Additionally, only half of SARS-CoV-2 naïve individuals had neutralizing antibodies to wild type virus, and only 2/25 had neutralizing antibodies to the B.1.351 variant after the first dose of vaccine, whereas nearly all subjects achieved neutralizing antibodies following the boost. Moreover, the frequency of memory B cells that were IgG+ and the fraction that was focused on RBD both increased after booster vaccination, indicating an improvement in the quality of the memory B cell response. Together, these data are consistent with the need for a two dose mRNA vaccine schedule in SARS-CoV-2 naïve individuals to achieve optimal levels of humoral immunity, including neutralizing antibodies against the B.1.351 variant.
We also observed a negative association of age with induction of B cell memory. Others have reported a negative association between age and serum antibody titers after a single mRNA vaccine dose (28, 29). We found a similar trend for antibodies following two doses of mRNA vaccination, but this did not reach statistical significance for our cohort. However, the magnitude of the memory B cell response following the second dose was lower with increased age, confirming age as a key variable in mRNA vaccine-induced immunity. It remains unclear if the age-associated effect on memory B cell induction represents a true difference in the magnitude of response or a difference in kinetics that will resolve at later timepoints. It is also challenging to define an exact threshold for how much immunological memory is sufficient to provide long-term protection. Although all subjects, regardless of age, had significant humoral and memory B cell responses to vaccination, these data highlight a need to further understand the age-related changes in responses to mRNA vaccination (33). In examining correlates of vaccine-induced immune responses, we also uncovered a trend suggesting that vaccine-induced side effects may be related to post-vaccination serum antibodies, but not memory B cells. Although more data are needed, it is possible that systemic inflammation early after vaccination could contribute to an initial induction of antibody with less of an impact on the development of memory B cells. Larger cohorts and more quantitative measures of vaccine-induced side effects may further clarify these relationships.
Finally, these analyses highlight the importance of interrogating vaccine-induced memory B cell responses alongside serological analyses. Specifically, we found no relationship between post-vaccination serum antibody levels and memory B cells in SARS-CoV-2 naïve subjects, indicating that antibody and memory B cell induction may be independent features of the immune response to mRNA vaccination. Previous work has found that antibodies and memory B cells correlate for some vaccines or antigens, but do not correlate for many others (34). Current research on SARS-CoV-2 vaccines has largely focused on measuring circulating antibodies without measuring memory B cells, which are important for durability of immune memory and potential recall responses to infection or future booster . Indeed, pre-existing memory B cells in SARS-CoV-2 recovered subjects correlated strongly with post-vaccination antibody levels in our cohort, underscoring the immunological connection between memory B cells and antibody recall responses (35). Taken together, our findings highlight the importance of evaluating memory B cells in addition to serologies to more completely characterize humoral immunity. Although high circulating titers of neutralizing antibodies are common surrogates of protective immunity, there are many scenarios where circulating antibodies may not achieve sterilizing immunity and additional immune responses from memory cells will be necessary (36). For example, high dose viral innoculums may require rapid generation of additional antibody from memory B cells. Moreover, if circulating antibodies wane over time, our data suggest that durable memory B cells are likely to provide a rapid source of protective antibody upon SARS-CoV-2 re-exposure. Lastly, infection with variant strains that partially escape neutralization by existing circulating antibodies (37–39) might require strong memory B cell populations that can re-seed germinal centers and diversify to respond to novel spike antigens (40).
In summary, our analysis of antibodies and cellular memory reveals distinct responses to SARS-CoV-2 mRNA vaccines based on prior history of infection. The addition of memory B cells in this analysis, both in terms of frequency and phenotype, provides complemenary data that strengthens current serology-based evidence (20, 21, 23, 24, 28, 29) for a single-dose vaccine schedule in COVID-19 recovered individuals. We also find associations of vacccine-induced immune responses with age and side effects, which may have relevance for future booster vaccines and public health campaigns. Thus, our study provides insight into the underlying immunobiology of mRNA vaccines in humans and may have implications for vaccination strategies in the future.
The long-term side effects of the corona virus vaccines are unknown. While people do and should have the right to choose, that choice =here isn the United States should also respect the right to choose NOT to take the vaccine when the risk of death is 0.5% (1 in 500 in all ages) overall and less in the young and healthy. That is so regardless of the almost certainly unsettled data regarding the benefit versus risk of corona virus vaccination in those who are perfectly healthy, men or women, under the age of 60. Moreover, there is documented effective treatment for those who do become ill, with a clinical 50% improvement in 1 day further reducing the need for vaccination.
And : Children’s health-related negative behaviors & the pandemic: see bottom
—
H. Robert Silverstein, MD, FACC
Medical Director, Preventive Medicine Center
1000 Asylum Avenue #2109
Hartford, CT 06105
(860) 549-3444
http://www.thepmc.org
September 15, 2021
Link between menstrual changes after COVID-19 vaccination is plausible and should be investigated
by British Medical Journal
Credit: Unsplash/CC0 Public Domain
Some women say their periods change after getting a COVID-19 vaccination. In The BMJ today, Dr. Victoria Male, a reproductive specialist at Imperial College London, says a link between menstrual changes after COVID-19 vaccination is plausible and should be investigated.
Changes to periods or unexpected vaginal bleeding are not listed as common side effects of COVID-19 vaccination, she writes. Yet more than 30,000 such reports had been made to the UK Medicines and Healthcare Products Regulatory Agency (MHRA) surveillance scheme for adverse drug reactions by 2 September.
However, most people find that their period returns to normal the following cycle and, importantly, there is no evidence that COVID-19 vaccination adversely affects fertility.
The MHRA states that its surveillance data does not support a link between changes to menstrual periods and COVID-19 vaccines, since the number of reports is low in relation to both the number of people vaccinated and the prevalence of menstrual disorders generally.
However, the way in which data is collected makes firm conclusions difficult, says Male.
She argues that approaches better equipped to compare rates of menstrual changes in vaccinated versus unvaccinated populations are needed, and points out that the US National Institutes of Health (NIH) has made $1.67 million available to encourage this important research.
Reports of menstrual changes after COVID-19 vaccination have been made for both mRNA and adenovirus-vectored vaccines, she adds, suggesting that, if there is a connection, it is likely to be a result of the immune response to vaccination, rather than to a specific vaccine component.
Indeed, the menstrual cycle may be affected by the body’s immune response to the virus itself, with one study showing menstrual disruption in around a quarter of women infected with SARS-CoV2.
If a link between vaccination and menstrual changes is confirmed, this will allow individuals seeking vaccination to plan in advance for potentially altered cycles, she explains.
In the meantime, she suggests clinicians encourage their patients to report any changes to periods or unexpected vaginal bleeding after vaccination to the MHRA’s scheme. And anyone reporting a change in periods persisting over a number of cycles, or new vaginal bleeding after the menopause, should be managed according to the usual clinical guidelines for these conditions.
“One important lesson is that the effects of medical interventions on menstruation should not be an afterthought in future research,” she concludes.
Can COVID-19 vaccines affect my period?
More information: Editorial: Menstrual changes following COVID-19 vaccination, The BMJ, DOI: 10.1136/bmj.n2211
Provided by British Medical Journal
Children’s health-related behaviors negatively impacted by COVID-19 pandemic
Researchers from the Department of Exercise Science have illuminated additional ways that the COVID-19 pandemic has impacted children’s health. A recent study by members of the Arnold Childhood Obesity Initiative research group has already revealed accelerated increases in children’s BMI and weight gain since the pandemic began. Now the team has published findings from a study on the impacts of the pandemic on children’s health-related behaviors (e.g., physical activity, diet, screen time, sedentary behavior, sleep).
Detection of SARS-CoV-2-Specific Memory B Cells
Antigen-specific B cells were detected using biotinylated proteins in combination with different streptavidin (SA)-fluorophore conjugates. Biotinylated proteins were multimerized with fluorescently labeled SA for 1 hour at 4C. Full-length spike protein (R&D Systems) was mixed with SA-BV421 (Biolegend) at a 10:1 mass ratio (e.g., 200ng spike with 20ng SA; ~4:1 molar ratio). Spike RBD (R&D Systems) was mixed with SA-APC (Biolegend) at a 2:1 mass ratio (e.g., 25ng RBD with 12.5ng SA; ~4:1 molar ratio). Biotinylated influenza HA pools were mixed with SA-PE (Biolegend) at a 6.25:1 mass ratio (e.g., 100ng HA pool with 16ng SA; ~6:1 molar ratio). Individual influenza HA antigens corresponding with the 2019 trivalent vaccine (A/Brisbane/02/2018/H1N1, B/Colorado/06/2017; Immune Technology) were biotinylated using an EZ-Link Micro NHS-PEG4 Biotinylation Kit (Thermo Fisher) according to the manufacturer’s instructions. Excess biotin was subsequently removed using Zebra Spin Desalting Columns 7K MWCO (Thermo Fisher) and protein was quantified with a Pierce BCA Assay (Thermo Fisher). SA-BV711 (BD Bioscience) was used as a decoy probe without biotinylated protein to gate out cells that non-specifically bind streptavidin. All experimental steps were performed in a 50/50 mixture of PBS + 2% FBS and Brilliant Buffer (BD Bioscience). Antigen probes for spike, RBD, and HA were prepared individually and mixed together after multimerization with 5uM free D-biotin (Avidity LLC) to minimize potential cross-reactivity between probes. For staining, 5×106 cryopreserved PBMC samples were prepared in a 96-well U-bottom plate. Cells were first stained with Fc block (Biolegend, 1:200) and Ghost 510 Viability Dye (Tonbo Biosciences, 1:600) for 15 min at 4C. Cells were then washed and stained with 50uL antigen probe master mix containing 200ng spike-BV421, 25ng RBD-APC, 100ng HA-PE, and 20ng SA-BV711 decoy for 1 hour at 4C. Following incubation with antigen probe, cells were washed again and stained with anti-CD3 (BD Bioscience, 1:200), anti-CD19 (Biolegend, 1:100), anti-CD20 (BD Bioscience, 1:500), anti-CD27 (BD Bioscience, 1:200), anti-CD38 (BD Bioscience, 1:200), anti-CD71 (BD Bioscience, 1:50), anti-IgD (BD Bioscience, 1:50), anti-IgM (Biolegend, 1:200), and anti-IgG (Biolegend, 1:400). After surface stain, cells were washed and fixed in 1% PFA overnight at 4C. For sorting, cells were stained with spike and HA probes followed by Fc block and Ghost 510 Viability Dye as described above. Cells were then stained for surface markers with anti-CD4 (Invitrogen, 1:333.3), anti-CD8 (Biolegend, 1:66.7), anti-CD14 (Biolegend, 1:200), anti-CD19 (BD Bioscience, 1:100), anti-CD27 (Biolegend, 1:66.7), and anti-CD38 (1:200). After surface stain, cells were washed and resuspended in PBS + 2% FBS for acquisition. All antibodies and recombinant proteins are listed in table S1 and table S2.
Flow Cytometry and Cell Sorting
Samples were acquired on a BD Symphony A5 instrument. Standardized SPHERO rainbow beads (Spherotech) were used to track and adjust photomultiplier tubes over time. UltraComp eBeads (Thermo Fisher) were used for compensation. Up to 5×106 cells were acquired per sample. Data were analyzed using FlowJo v10 (BD Bioscience). Antigen-specific gates were set based on healthy donors stained without antigen probes (similar to an FMO control) and were kept the same for all experimental runs. All timepoints for individual subjects were run in the same experiment to minimize batch effects. The full gating strategy is shown in figure S2. Cell sorting was performed on a BD FACSAria II instrument in low pressure mode, using a 70 μm nozzle. SARS-CoV-2-specific memory B cells were similarly identified as live, CD14-, CD19+, CD27+ CD38lo/int, HA- Spike+. Cells were sorted into 1.5 DNA LoBind Eppendorf tubes containing 300 μl of cell lysis buffer (Qiagen) and stored at room temperature until nucleic acid extraction.
B Cell Receptor (BCR) Sequencing
DNA was extracted from sorted cells using Gentra Puregene Cell kit (Qiagen, Cat.# 158767). Immunoglobulin heavy-chain family-specific PCRs were performed on genomic DNA samples using primers in FR1 and JH as described previously (46, 47). Two biological replicates were run on all samples. Sequencing was performed in the Human Immunology Core Facility at the University of Pennsylvania using the Illumina 2 × 300-bp paired-end kit (Illumina MiSeq Reagent Kit v3, 600-cycle, Illumina MS-102-3003).
B Cell Receptor (BCR) Sequence Analysis
Raw reads from the Illumina MiSeq were quality controlled with pRESTO v0.6.0 (48) as described in (49). Sequences passing the quality control procedure were imported into IgBLAST v1.17.0 (50) for gene identification and alignment. The primer binding region (IMGT nucleotide positions 1-80) was replaced with Ns and sequences beginning after IMGT position 90 were removed to avoid incorrect V-gene calls and skewed SHM analysis. The remaining sequences were imported into ImmuneDB v0.29.10 (51) for clonal inference, lineage construction, and downstream analyses. Sequences sharing the same VH-gene, JH-gene, CDR3 length, and 85% amino-acid homology in the CDR3 were aggregated into clones. After sequences were collapsed into clones, non-productive sequences and clones with 1 copy number sequences were excluded from all downstream analysis.
Lineages were constructed within ImmuneDB as described in (51). Within each lineage, sequences with fewer than ten copies across all samples in a donor were excluded to reduce the effect of sequencing error and improve fidelity. The resulting lineage structures were visualized with ete3 (52). Each node represents a unique sequence and the size of each node is proportional to the total copy number of the sequence. Nodes are depicted as pie-charts where each wedge indicates the proportion of copies at each timepoint and inferred nodes are shown in black. The number next to each node represents the number of nucleotide mutations as compared to the preceding vertical node.
immunology.sciencemag.org/cgi/content/full/6/58/eabi6950/
Is The Pandemic Over?
By Thomas T. Siler, MD 3/16/21
There is scientific data showing that we may be closer to herd immunity and the end of the pandemic than the media and government let on. SARS-COV-2 is in the family of coronaviruses and shares common characteristics with other members of the same family. Four coronaviruses commonly circulate in our population and cause symptoms of the common cold. People have some cross-immunity to the “new” SARS-COV-2 virus from previous infections with other viruses in the Coronavirus family.
T cells and antibodies can both be measured to study immunity. Studies of T cell function in 2020 showed that patients who had not been exposed to SARS-COV-2 in several countries had evidence of cross-reactivity from known coronaviruses and SARS-COV-2. The range of cross-reactivity ranged from 18% in Sweden to 51% in Singapore. Interestingly, the countries with a higher level of T cell cross-reactivity to SARS-COV-2 had a lower death rate during the pandemic. A study of blood bank samples from 2015 to 2018 in the U.S. showed 50% of the samples had cross-reactive T cells to SARS-COV-2 from prior coronavirus infections.
Levels of pre-existing immunity may explain why some people don’t get infected and why others have a milder case of COVID-19. Because children have a higher chance of catching the “cold” viruses, this may also partly explain why children are not affected very much by SARS-COV-2. This also happened in the 2009 H1N1 Swine flu pandemic: 30% of people over 60 years old had prior immunity to Swine flu from earlier immunity to other influenza infections. This fact lessened the severity of that pandemic.
In addition to pre-existing immunity, persons who have had COVID-19 are generally thought to be immune. Factoring in a correct case count can show we are closer to herd immunity than we thought. Many actual cases of COVID-19 infection have not been counted because patients may have mild infections, may not get tested, or may not have access to testing. In August, the World Health Organization estimated that 10% of the world’s population had contracted COVID-19 infection (760 million). At that time the reported case count was 35 million (20 times lower).
In the United States, the CDC estimated in late November that the total cases could approach 100 million. The actual case count in the United States at the end of December was 20 million (as much a 5 times lower). If most populations have 20-50% pre-existing immunity from prior Coronavirus infections and the actual numbers of COVID-19 infections are much higher (3.5 to 20 times higher), then we could be approaching herd immunity (which is guessimated at 70%) now, even with our current low level of vaccination. If you make those calculations for the United States, then 45 to 90% of the American population could be immune now.
Dr. Mike Yeadon, a former Pfizer scientist with 30 years of experience in immunology, says the pandemic effectively ended, even before we began to vaccinate people. Dr. Marty Makary wrote in a recent article in the WSJ that he feels herd immunity could come by April, 2021, and also be in effect before we have vaccinated “everyone.” Both these scientists came to this conclusion by saying that more people have already been infected (up to 150 million for the U.S.) already and there was pre-existing immunity at some level for a portion of the population. This seems to be what is happening in our experience of COVID-19 tracking. Since January 8th there has been a significant drop in cases in the U.S. Hospitalizations and deaths are also going down in almost every locale.
This cannot be explained by changes in behavior (masking, etc.) and it is too early for the low level of vaccinations to explain this fall in infections. This drop happened despite more travel over Christmas, 2020, and the holidays. We must still protect the elderly and those with pre-existing conditions that could make patients susceptible to more severe infections. These persons should consider taking the vaccines. In my opinion, persons with a low risk of serious infection (healthy persons under 70) can wait on vaccination. Variants of the virus are not likely to change this analysis. This view should also affect the rationale for lockdowns and closing businesses, allowing states to open up sooner. Florida, South Dakota, and Georgia have lessened restrictions on the public and businesses months ago and are doing well. Texas, Mississippi, Arizona, and Connecticut have just lessened their COVID-19 mandates and opened-up more completely.
If it continues to appear that herd immunity is being reached and cases, hospitalizations, and deaths continue to go down, we should lobby all our states to lessen restrictions by the end of spring. The pandemic can be managed with a more targeted approach and the healthy can go on with their lives with less restriction. The pandemic, indeed, may finally be coming to a close.
Marty Makary, MD, professor at the Johns Hopkins School of Medicine and Bloomberg School of Public Health 5/4/21 CONDENSED/EDITED
As the COVID pandemic wanes, Americans perception of the risks by the media and some experts is incorrect. Fear by repeating speculation that variants will evade vaccines is unnecessary. Here are the facts: About 57 percent of adults are vaccinated and approximately half of unvaccinated people have natural immunity from prior infection. That’s why US cases have been plummeting, down 31 percent over the past 18 days.
During the mildest flu season in the last eight years, there were 24 million cases, according to the Centers for Disease Control, and approximately 447,000 daily cases during its peak week. By comparison, we’re averaging 49,641 daily COVID cases. That same mild flu season resulted in 280,000 hospitalizations. By comparison, current CoV2-19 hospitalizations as of May 1 are 34,905.
Downplaying the risk of Co V2-19 among susceptible people is wrong. But for the millions of Americans who are immune and live where the cases are low, the public-health threat is now vastly decreased and below seasonal-flu levels. Given the harm of social isolation, 0verdose, daage tosocial life and business, we need to abandon the goal of absolute risk elimination at all cost.
The case-fatality risk of CoV2-19 is also plummeting. That’s because a remarkable 83 percent of US seniors are now vaccinated and the virus has moved to younger, healthier people. For Americans under age 55, the case-fatality rate last month was approximately 6.5 per 10,000. Since we are now capturing as many as 1 in 4.5 infections with testing, the real-world case-fatality rate is 1 to 1.4 per 10,000, similar to influenza (1.3 per 10,000 in people under 50). And vaccines will likely provide at least some immunity against variants. With far fewer susceptible people and a younger cohort, we’re dealing with a different risk level than even just a few months ago.
Despite this good news, Americans are being told variants and hesitancy will prevent “herd immunity.” Yet noticeably absent from their calculations is the contribution of natural immunity from prior infection or exposure. Dr. Anthony Fauci and Dr. Rochelle Walensky don’t talk about the percent of Americans they estimate have natural immunity. That omission creates a perception that the race to 70 to 85 percent immunity is more desperate, resulting in a prolonged timeline, talk of vaccine mandates and an imperative that young children must be immunized. Some experts do highlight the massive contribution of natural immunity. Dr. Scott Gottlieb, former head of the Food and Drug Administration, and Dr. Monica Gandhi at the University of California are among the many who routinely point to the broad prevalence of natural immunity.
In a recent Danish study of natural immunity, only 0.6 % of those infected ever subsequently tested positive, including asymptomatic positives. A large California study found 38 percent of the state’s population and 45 percent of Los Angeles residents had COVID-19 antibodies in February, 2021. Given the many infections since then and the immunity conferred by activated T cells even when antibodies are not detected, upward of half the population of California likely has natural immunity. And cases in California have dropped dramatically.On a clinical level, we simply have not seen significant reinfections at any concerning rate. Any expert who talks about the path to herd immunity as a simple tally of vaccinations alone – which unfortunately is many – is ignoring data and real-world clinical experience.
In February, 2021, I projected that “based on the current trajectory” we would see significant population immunity take hold in April, 2021, from the combination of vaccinated and natural immunity. While most states are now witnessing the strong suppression of the epidemic today, other states will get there in May, 2021. But herd immunity is not a finish line. In that article, I maintained that “coronavirus will be here for decades to come.” The question is, at what point is it no longer a major public-health threat? For most states, it’s now. Given the now low, manageable and declining CoV2-19 threat, let’s stop depriving people of their livelihoods and move toward normalcy.
New vaccine blocks COVID-19 and variants, plus other coronaviruses
by S Avery, Duke U School of Nursing 5/10/21
A potential new vaccine developed by members of the Duke Human Vaccine Institute has proven effective in protecting monkeys and mice from a variety of coronavirus infections—including SARS-CoV-2 as well as the original SARS-CoV-1 and related bat coronaviruses that could potentially cause the next pandemic.
The new vaccine, called a pan-coronavirus vaccine, triggers neutralizing antibodies via a nanoparticle. The nanoparticle is composed of the coronavirus part that allows it to bind to the body’s cell receptors, and is formulated with a chemical booster called an adjuvant. Success in primates is highly relevant to humans.
Senior author Barton F. Haynes, M.D., director of the Duke Human Vaccine Institute (DHVI).
“This approach not only provided protection against SARS-CoV-2, but the antibodies induced by the vaccine also neutralized variants of concern that originated in the United Kingdom, South Africa and Brazil,” Haynes said. The induced antibodies reacted with quite a large panel of coronaviruses.
The Achilles heel for the coronaviruses is their receptor-binding domain, located on the spike that links the viruses to receptors in human cells. The team then designed a nanoparticle displaying this vulnerable spot.
In tests of its effect on monkeys, the nanoparticle/alum adjuvant vaccine blocked COVID-19 infection by 100%. The new vaccine also elicited significantly higher neutralizing levels in the animals than current vaccine platforms or natural infection in humans.
“Basically what we’ve done is take multiple copies of a small part of the coronavirus to make the body’s immune system respond to it in a heightened way,” Saunders said. More information: Kevin O. Saunders et al. Neutralizing antibody vaccine for pandemic and pre-emergent coronaviruses, Nature (2021). DOI: 10.1038/s41586-021-03594-0
T cells recognize recent SARS-CoV-2 variants
by NIH/National Institute of Allergy and Infectious Diseases 3/30/21
When variants of SARS-CoV-2 (the virus that causes COVID-19) emerged in late 2020, concern arose that they might elude protective immune responses generated by prior infection or vaccination, potentially making reinfection more likely or vaccination less effective. To investigate this possibility, researchers from the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, and colleagues analyzed blood cell samples from 30 people who had contracted and recovered from COVID-19 prior to the emergence of virus variants. They found that one key player in the immune response to SARS-CoV-2—the CD8+ T cell—remained active against the virus.
The research team was led by NIAID’s Andrew Redd, Ph.D., and included scientists from Johns Hopkins University School of Medicine, Johns Hopkins Bloomberg School of Public Health and the immunomics-focused company, ImmunoScape.
The investigators asked whether CD8+ T cells in the blood of recovered COVID-19 patients, infected with the initial virus, could still recognize three SARS-CoV-2 variants: B.1.1.7, which was first detected in the United Kingdom; B.1.351, originally found in the Republic of South Africa; and B.1.1.248, first seen in Brazil. To stay healthy cells must recognize parts of the virus protein presented on the surface of infected cells and killing those cells.
In their study of recovered COVID-19 patients, the researchers determined that SARS-CoV-2-specific CD8+ T-cell responses remained largely intact and could recognize virtually all mutations in the variants studied. While larger studies are needed, the researchers note that their findings suggest that the T cell response in convalescent individuals, and most likely in vaccinees, are largely not affected by the mutations found in these three variants, and should offer protection against emerging variants.
Optimal immunity to SARS-Cov-2 likely requires strong multivalent T-cell responses in addition to neutralizing antibodies and other responses to protect against current SARS-CoV-2 strains and emerging variants, the authors indicate.
Moderna and Pfizer-BioNTech vaccines prime T cells to fight SARS-CoV-2 variants
by La Jolla Institute for Immunology 7/1/21
T cells from people who have recovered from COVID-19 or received the Moderna or Pfizer-BioNTech vaccines are still able to recognize several concerning SARS-CoV-2 variants. Credit: La Jolla Institute for Immunology
Researchers at La Jolla Institute for Immunology (LJI) have found that T cells from people who have recovered from COVID-19 or received the Moderna or Pfizer-BioNTech vaccines are still able to recognize several concerning SARS-CoV-2 variants.
Their new study, published online on July 1, 2021 in Cell Reports Medicine, shows that both CD4+ “helper” T cells and CD8+ “killer” T cells can still recognize mutated forms of the virus. This reactivity is key to the body’s complex immune response to the virus, which allows the body to kill infected cells and stop severe infections.
“This study suggests that the impact of mutations found in the variants of concern is limited,” says LJI Professor Alessandro Sette, Dr.Bio.Sci., study senior author and member of the LJI Center for Infectious Disease and Vaccine Research. “We can presume that T cells would still be available as a line of defense against viral infection.”
“These variants are still a concern, but our study shows that even if there is a decrease in antibodies, as other studies have shown, the T cells remain largely unaffected,” says LJI Instructor Alba Grifoni, Ph.D. “The vaccines still work.”
The Johnson & Johnson/Janssen COVID-19 vaccine was not part of this study because it was not available at the time the study was launched.
LJI findings guide COVID-19 vaccine efforts
This study was previously published online as a pre-print in March 2021. The findings were then highlighted by National Institutes of Allergy and Infectious Diseases Director Anthony Fauci, M.D., in a March 31 White House press briefing.
“We’re learning more and more that these CD4+ and CD8+ T cells are very important in that they cross-react against certain viral variants,” Fauci said.
In the time since the paper was first published, the researchers have nearly tripled their patient cohort and performed an additional method of stringent statistical analysis. “We’ve also added an unexposed donor cohort,” says study first author Alison Tarke, a Ph.D. student of the University of Genoa, guested in the Sette Lab at LJI.
For the new study, the researchers analyzed T cells from three different groups: People who had recovered from COVID-19, people who had received either the Moderna or Pfizer-BioNTech vaccines, and people never exposed to SARS-CoV-2 (from samples taken before the pandemic).
Both the recovered subjects and the vaccinated subjects were likely to have T cells that recognized the “ancestral lineage” of SARS-CoV-2. This was the original strain that emerged at the beginning of the pandemic; however, the virus has mutated since December 2019, and several variants have been identified as VOCs.
The question was whether people with T cells trained to recognize the ancestral strain would also recognize the new variants. The researchers tested T cell responses from the donor groups against four prominent VOCs: Alpha (B.1.1.7), Beta (B.1.351), Gamma (P.1) and Epsilon (B.1.427/B.1.429).
The researchers found that both vaccinated individuals and the recovered patients had cross-reactive T cells that could target these variants. That is good news for anyone who has received one of the two mRNA vaccines and for anyone who has recovered from any variant of the virus.
Similar results reporting largely unaffected T cell reactivity has now been presented through independent studies published in Nature and Science Immunology.-2.
“COVID vaccines do a fantastic job of making antibodies that stop SARS-CoV-2 infections, but some of the vaccines do less well at stopping infections from variants,” Crotty says. “You can think of T cells as a backup system: If the virus gets past the antibodies—if you have vaccine T cells the T cells can probably still stop the variant coronavirus infection before you get pneumonia.”
More information: Cell Reports Medicine (2021). DOI: 10.1016/j.xcrm.2021.100355 , www.cell.com/cell-reports-medi … 2666-3791(21)00204-4
More information: Andrew D Redd et al, CD8+ T cell responses in COVID-19 convalescent individuals target conserved epitopes from multiple prominent SARS-CoV-2 circulating variants, Open Forum Infectious Diseases (2021). DOI: 10.1093/ofid/ofab143
What Are the Factors That Improve COVID Vaccine Antibody Response?
Miriam E. Tucker; CME Author: Charles P. Vega, MD 4/2/2021 Medscape
The emergency use authorizations for both the Pfizer-BioNTech[1] and Moderna[2] vaccines cited good vaccine efficacy in a single dose (82% and 80.2%, respectively). The capacity to mount humoral immune responses to COVID-19 vaccinations may be reduced among people who are heavier, older, and male, new findings suggest. The data pertain specifically to the mRNA vaccine, BNT162b2, developed by Pfizer Inc. and BioNTech SE. The study was conducted by Italian researchers and was published February 26 as a preprint.[3]
The study involved 248 healthcare workers who each received 2 doses of the vaccine. Of the participants, 99.5% developed a humoral immune response after the second dose. Those responses varied by body mass index (BMI), age, and sex.
“The findings imply that female, lean and young people have an increased capacity to mount humoral immune responses compared to male, overweight and older populations,” said Raul Pellini, professor at the IRCCS Regina Elena National Cancer Institute, Rome, Italy, and colleagues.
Results Contrast With Pfizer Trials of Vaccine
The current Italian study showed somewhat lower levels of antibodies in people with obesity (beginning at a BMI OF 23 = 5’6’ & 150 lbs) compared with people who did not have obesity, the phase 3 trial found no difference in symptomatic infection rates. “These results indicate that even with a slightly lower level of antibody induced in obese people, that level was sufficient to protect against symptomatic infection,” Iwasaki told Medscape Medical News. After the second dose, 99.5% of participants developed a humoral immune response; one person did not respond. None tested positive for SARS-CoV-2.
Titers of SARS-CoV-2 binding antibodies were greater in younger than in older participants. There were statistically significant differences between persons aged ≤ 37 years (453.5 AU/mL) in comparison with persons aged 47 to 56 years (239.8 AU/mL; P = .005), persons aged ≤ 37 years vs persons aged > 56 years (453.5 vs 182.4 AU/mL; P < .0001), and persons aged 37 to 47 years vs persons aged > 56 years (330.9 vs 182.4 AU/mL; P = .01). Antibody response was significantly greater for women than for men (338.5 vs 212.6 AU/mL; P = .001).
Humoral responses were greater in persons of normal-weight BMI (18.5 to 24.9 kg/m2; 325.8 AU/mL) and persons of underweight BMI (<18.5 kg/m2; 455.4 AU/mL) compared with persons with pre-obesity, defined as BMI of 25 to 29.9 kg/m2 (222.4 AU/mL), and persons with obesity (BMI ≥ 30 kg/m2; 167 AU/mL; P < .0001). This association remained after adjustment for age (P = .003).
“Our data stresses the importance of close vaccination monitoring of obese people, considering the growing list of countries with obesity problems,” the researchers noted. Hypertension was also associated with lower antibody titers (P = .006), but that lost statistical significance after matching for age (P = .22). “We strongly believe that our results are extremely encouraging and useful for the scientific community,” Pellini and colleagues concluded.
The study population comprised healthcare workers at one hospital in Italy who were presenting for the Pfizer-BioNTech COVID-19 vaccine. All participants were between ages 18 and 75 years, and individuals with evidence of current or previous SARS-CoV-2 infection or a history of possible immunosuppression were excluded.
• Investigators collected blood and a nasopharyngeal samples before the first vaccine dose and 7 days after the booster dose. Researchers collected sera from adults with known COVID-19 to compare immunoglobulin G (IgG) antibody levels against S1/S2 antigens of SARS-CoV-2 with those of vaccinated participants.
• None of the participants had a positive polymerase chain reaction test for SARS-CoV-2 before or after vaccination.
• The antibody geometric mean concentrations after vaccination and among the cohort who provided convalescent sera after COVID-19 infection were 285.9 AU/mL and 39.4 AU/mL, respectively (P < .0001).
• 99.5% of vaccine recipients were considered to have responded after the vaccine. Only one participant did not.
• There was a fairly linear decline in immune response from participants aged < 37 years to groups aged 37 to 47 years, 47 to 56 years, and more than 56 years.
• The other major variable associated with a reduced immune response to the vaccine was overweight or obesity vs underweight or normal weight.
• The presence of hypertension failed to affect vaccine response in adjusted analysis.
The COVID-19 mRNA vaccines appear to have efficacy around 80% against COVID-19 after a single dose.
• In the current study by Pellini and colleagues, older age, male sex, and overweight/obesity were associated with a reduced IgG response after application of an mRNA vaccine against COVID-19.
“How Common Is SARS-CoV-2 Infection After Vaccination?”. 5 positive cases in 10,000 fourteen days after complete vaccination. NONE were moderately or seriously ill.
Click to view: How Common Is SARS-CoV-2 Infection After Vaccination?
by Francesca Torriani, MD UC San Diego Health, San Diego, CA
Thousands of vaccinated people in U.S. infected with COVID-19
COVID-19 infections have occurred in about 5,800 (0.00001%) people in the United States who have been vaccinated against the virus, the U.S. Centers for Disease Control and Prevention says.
But “vaccine breakthrough” infections were expected among the estimated 179 million people in the United States who are fully vaccinated against the new coronavirus, officials added. In clinical trials, none of the vaccines were 100 percent effective in preventing infections. The agency told CNN that 7 percent of people with these vaccine breakthrough infections had to be hospitalized and 74 died.
“Vaccine breakthrough infections were reported among all people of all ages eligible for vaccination. However, a little over 40 percent of the infections were in people 60 or more years of age,” the CDC said. Of the breakthrough infections, 65 percent occurred in women and 29 percent were asymptomatic, CNN reported. The agency said it will try to identify vaccinated people most at risk for breakthrough infections.
The new data are the first to indicate how effective COVID-19 vaccines are in real life and the first to show that they do not completely protect against severe illness and death, CNN reported. Breakthrough infections are not unexpected, and more will occur as the number of people who are vaccinated increases.
Breakthroughs – According to the CDC, as of the end of April 2021, 10,262 people contracted Covid-19 despite being vaccinated (~1% of fully vaccinated people). Who were these people and what happened to them? ✓ Age 58 on average, more females than males. ✓ 27% were asymptomatic. ✓ 10% (1,262) were hospitalized (~0.1% of vaccinees), and nearly a third of these were asymptomatic or hospitalized for a reason other than Covid-19. ✓ 2% (160) died (0.02% of vaccinees or 1 in ~630,000 vaccinees). The median age of those people was 82, with 18% either asymptomatic or dying from a cause unrelated to Covid-19. The bottom line: A vaccinated person’s chance of dying from Covid-19 is about 1 in a million.
” …The risk of dying from COVID doubles roughly every seven years older a patient is. The 35-year difference between a 35-year-old and a 70-year-old means the risk of death between the two patients has doubled five times—equivalently it has increased by a factor of 32. An unvaccinated 70-year-old might be 32 times more likely to die of COVID than an unvaccinated 35-year-old. This dramatic variation of the risk profile with age means that even excellent vaccines don’t reduce the risk of death for older people to below the risk for some younger demographics.
PHE data suggests that being double vaccinated reduces the risk of being hospitalized with the now-dominant delta variant by around 96%. Even conservatively assuming the vaccines are no more effective at preventing death than hospitalization (actually they are likely to be more effective at preventing death) this means the risk of death for double vaccinated people has been cut to less than one-twentieth of the value for unvaccinated people with the same underlying risk profile.
However, the 20-fold decrease in risk afforded by the vaccine isn’t enough to offset the 32-fold increase in underlying risk of death of an 70-year-old over a 35-year-old …”
Duration of Culturable SARS-CoV2-19 in Hospitalized Patients with Covid 19 NEJM 2021;384: 671 M-C Kim. “Viral clearance by culture was 7 days, 34 days by real-time PCR. The latest positive viral culture was 12 days after symptom onset in 1 patient. Viable virus was identified until 3 days after the resolution of fever in 1 patient, viral culture was positive only in samples with a cycle threshold of 28.4 or less.
A 12/14/20 New York Times article with great drawings how the virus infects and how the Pfizer & Moderna mRNA vaccines work:
https://www.nytimes.com/interactive/2020/health/pfizer-biontech-covid-19-vaccine.html
Real-World Data Demonstrate Effectiveness of Pfizer-BioNTech COVID-19 Vaccine
Brian Park, PharmD 3/15/21
“2 weeks after the second dose, the vaccine was at least 97% effective against symptomatic COVID-19 cases, hospitalizations, severe and critical hospitalizations, and deaths”
Patients With Detectable SARS-CoV-2 IgG Antibody Are Noninfectious
British Dental Journal 11/28/20
• After infection, SARS-CoV-2 viral infectivity lasts for 8 days in non-immunocompromised patients. Polymerase chain reaction (PCR) swab tests can remain positive for 7 weeks post infection, but that represents nonviable remnants of virus. Neutralizing SARS-CoV-2 antibodies can be detected 11 days after infection, and persist for variable periods, typically months.
• The best blood and finger-prick SARS-CoV-2 antibody tests have a 95% specificity and 99% sensitivity for IgG and take roughly 10 minutes, whereas PCR swab tests for antigen are only about 70% sensitive. While antibody levels do fall over time, once IgG has been detected, the patient can safely be regarded as noninfectious (99% certainty) and remain immune for at least months.
Contra Costa county California CoV2-19 death rates:
Here are the COVID deaths in Contra Costa County by age groups. 5.6% of the Contra Costa’s total population tested positive for COVID.
• Over 90 years of age, if you tested positive for COVID, the death rate is an astonishing 27.1%.
• 81 to 90 years of age, if you tested positive for COVID, the death rate is a horrendous 16.0%.
• 71 to 80 years of age, if you tested positive for COVID, the death rate is a terrible 7.0%.
• 61 to 70 years of age, if you tested positive for COVID, the death rate is an unacceptable 2.5%.
• 51 to 60 years of age, if you tested positive for COVID, the death rate is still an unacceptable 0.72%.
• 41 to 50 years of age, if you tested positive for COVID, the death rate is 0.20%.
• 31 to 40 years of age, if you tested positive for COVID, the death rate is 0.076%.
• 19 to 30 years of age, if you tested positive for COVID, the death rate is 0.0069%.
• 13 to 18 years of age, if you tested positive for COVID. the death rate is 0.0231%.
• 5 to 12 years of age, if you tested positive for COVID the death rate is 0.0%.
• Newborn to 4 years of age, if you tested positive for COVID the death rate is 0.0%.
10/29/20 Covid-19 is at least three rimes as lethal as the current influenza.
Cases are rising and testing is increasing: now MUCH reduced
Across the United States, Covid-19 hospitalizations were up 45% in October, 2020, in large part with less sick people, and these patients are discharged much sooner due to improved treatment. Covid deaths ha risen slightly: see the chart below.
Link for the lastest case counts, map and trends from the New York Times:
https://www.nytimes.com/interactive/2021/us/covid-cases.html
New York Times
7-day average
New cases
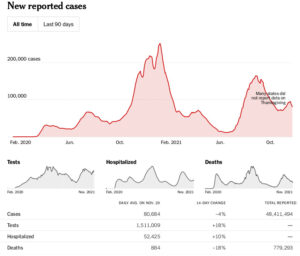
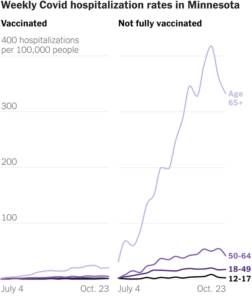
Hospitalization data from the U.S. Department of Health and Human Services; 14-day change trends use 7-day averages.
CoV2-19 vaccines in development designed to prevent clinical infection, disease severity, or both show the induction of an anamnestic immune response to the spike protein with a second dose7 and can generate high levels of neutralizing antibodies comparable with or greater than those seen in sera samples from patients.
Corresponding Author: David S. Stephens, MD, Emory University School of Medicine, Robert W. Woodruff Health Sciences Center, Emory University, 1440 Clifton Rd NE, #420, Atlanta, GA 30322 ([email protected]).
Duke & Washington & Jefferson Universities STOPS the spread of CoV2-19
Duke University’s aggressive COVID testing and surveillance minimized infections
by Duke University Medical Center 11/18/20
An aggressive COVID-19 surveillance and testing effort at Duke University was highly effective in minimizing the spread of the disease among students on campus, according to a case study appearing Tuesday in the CDC’s Morbidity and Mortality Weekly Report. The successful Duke campaign was launched before the start of the semester. Ahead of arriving on campus, all enrolled students were required to self-quarantine for 14 days, sign a code of conduct pledge to obey mask-wearing and social distancing guidelines and have a COVID test.
Once classes started, the university conducted regular surveillance testing using pooled samples to conserve resources, daily symptom self-monitoring, contact tracing with quarantine, and regular testing for those who were symptomatic or had been exposed to someone with COVID-19. The result: The average per-capita infection prevalence among students was lower than in the surrounding community, and large outbreaks seen on other campuses were avoided. Overall, combined testing approaches identified 84 cases among students, with 51% occurring among asymptomatic people. “Our experience at Duke shows that combined risk reduction strategies and surveillance testing can significantly lower transmissions on college campuses and beyond,” said lead author Thomas Denny, professor of medicine at Duke University School of Medicine and chief operating office at the Duke Human Vaccine Institute.
Denny said the Duke experience relied on a combination of strategies. In addition to the testing and quarantining before students arrived on campus, the measures included:
Creating a smartphone app for daily symptom self-monitoring and reporting; having students living on campus conduct twice-weekly tests themselves, using kits with prelabeled tubs, swabs and specimen bags; off-campus students tested at least once a week; strategically locating sites across campus to collect testing samples from students;
Batching samples in a process called pooled testing, with five samples grouped and analyzed for the presence of the virus. Batches that registered positives were then broken into individual samples and tested separately to identify the source of the positive. The Duke Human Vaccine Institute processed 80,000 samples from August-October.
“By late summer there were still things we didn’t fully understand about SARS-CoV-2 transmission, so there was some uncertainty going into the fall semester,” said Steve Haase, associate professor in Duke’s departments of Biology and Medicine. “Over the course of the semester we’ve learned many things, including that it is possible to limit the spread of the virus and create a safer environment for our students to have that invaluable on-campus learning experience.”
“Thanks to the collaboration of literally hundreds of dedicated individuals, along with the high level of engagement by our students, we have had a very positive fall,” said co-author Kyle Cavanaugh, vice president of Administration at Duke University. “Our dynamic surveillance testing strategy has served as a key component of our experience that has also included very high compliance with masking, social distancing and other key public health behaviors.”
Journal information: Morbidity and Mortality Weekly Report
Small behavioral changes determine success or complete failure of epidemic control
by Institute of Science and Technology Austria
Fluid and turbulence physicist Björn Hof and his team applied the statistical methods to epidemic spreading and discovered surprising features of the infection curves.
What does fluid physics have to do with the spreading of the coronavirus? The newest findings about the epidemic come from Physics Professor Björn Hof and his research group at the Institute of Science and Technology Austria (IST Austria).
Turbulence is described by statistical models that are equally used to describe forest fires and epidemics. Programming an epidemic model was a straightforward exercise for Burak Budanur, the group’s theorist and computational expert.
The epidemic curve does not flatten, it collapses
Standard epidemic models suggest that the level of mitigation has a continuous effect on the height of the epidemic peak. “The expectation is that the curve flattens in proportion to the level of social distancing,” says Davide Scarselli, main author. When first simulated epidemics taking limits in testing and contact tracing into account, the picture was a very different. The maximum number of infected people initially decreased as expected, but then suddenly collapsed to almost zero as the mitigation level reached a certain threshold. In one limit, approximately half of the people got infected during the epidemic. In the other one, only 3% caught the disease. It was impossible to obtain a result in between these two outcomes: Either there is an outbreak of considerable size, or there is almost none whatsoever.
Failure yields faster than exponential growth
Testing of known contacts (not testing per se) is one of the most powerful ways to slow down an epidemic. However, the number of cases that can be traced every day is limited and so is the number of tests that can be administered. The researchers found that exceeding these limits at one point during the epidemic has far-reaching consequences. “If this happens,” says Timme, “the disease begins to spread faster in the unchecked areas and this unavoidably causes a super-exponential increase in infections.” Already, exponential growth is immense. It means doubling infections every few days. Super-exponential signifies that even the rate of doubling becomes even faster.
As long as this acceleration can be avoided, epidemic curves collapse to a comparably low case level. Interestingly, it matters relatively little whether contact tracing is protected by a small or a large safety margin. The numbers remain comparatively low. If, on the other hand, the limit is only surpassed by a single case, the super-exponential growth causes the total case numbers to jump tenfold.
Marginal differences and disproportionate effects
“Like most nations, Austria didn´t react early against the second wave. Once not all contacts could be traced anymore last September, it wasn’t difficult to predict that case numbers would soon surge at a faster-than-exponential rate,” says Scarselli. Over the last year, it has become apparent that an early and decisive response is essential when facing exponential growth, though the team’s study shows that test limits make timing even more crucial.
The difference between success and failure of a lockdown is marginal, or as Budanur puts it: “A policy that would have worked yesterday will not only take much longer to take effect, but it may fail entirely if it is implemented a single day too late.” Hof adds: “Most European countries only reacted when health capacity limits became threatened. Actually, policy makers should have paid attention to their contact tracing teams and locked down before this protective shield fell apart.”
More recently, the team has looked into optimal strategies in which lockdowns are used as a preventive tool rather than an emergency brake. A manuscript that outlines the optimal strategy, which minimizes both the number of infected people and the required lockdown time, is currently in progress. The study is published in Nature Communications.
More information: Davide Scarselli et al. Discontinuous epidemic transition due to limited testing, Nature Communications (2021). DOI: 10.1038/s41467-021-22725
“Study: Colleges can prevent 96% of COVID-19 infections with common measures
by Case Western Reserve University 1/13/21
The combined effectiveness of three COVID-prevention strategies on college campuses—mask-wearing, social distancing, and routine testing—are as effective in preventing coronavirus infections as the Pfizer and Moderna vaccines approved by the U.S. Food and Drug Administration (FDA), according to a new study co-authored by a Case Western Reserve University researcher.
The research, published in Annals of Internal Medicine, has immediate significance as college semesters are poised to start again—and as the distribution of approved vaccines lags behind goals.
The study found that a combination of just two common measures—distancing and mandatory masks—prevents 87% of campus COVID-19 infections and costs only $170 per infection prevented..” Frequent testing rasied the cost mor that 10 times.
January 7, 2021
SARS-CoV-2 Transmission From People Without COVID-19 Symptoms
Michael A. Johansson, PhD1,2; et al
JAMA Netw Open. 2021;4(1):e2035057. doi:10.1001/jamanetworkopen.2020.35057
“ …persons with infection who never develop symptoms may account for approximately 24% of all transmission. In this base case, 59% of all transmission came from asymptomatic transmission, comprising 35% from presymptomatic individuals and 24% from individuals who never develop symptoms. Under a broad range of values for each of these assumptions, at least 50% of new SARS-CoV-2 infections was estimated to have originated from exposure to individuals with infection but without symptoms …”
May 21, 2021
Face masks effectively limit SARS-CoV-2 transmission
by Max Planck Society
‘Don’t forget the mask’—although most people nowadays follow this advice, professionals express different opinions about the effectiveness of face masks. An international team led by researchers from the Max Planck Institute for Chemistry in Mainz, Germany, has now used observational data and model calculations to answer open questions. The study shows under which conditions and in which way masks actually reduce individual and population-average risks of being infected with COVID-19 and help mitigate the corona pandemic. In most environments and situations, even simple surgical masks effectively reduce the transmission of SARS-CoV-2 and the effective reproduction number for COVID-19. In environments with potentially high airborne virus concentrations such as medical settings and densely occupied indoor spaces, however, masks with higher filtration efficiency (N95/FFP2) should be used and combined with other protective measures such as intensive ventilation.
Some earlier investigations found that masks were apparently not effective under certain conditions.
Researchers from the Max Planck Institute for Chemistry (MPIC), the Medical Center of the Johannes Gutenberg University Mainz, and the Charité – Universitätsmedizin Berlin together with partners from China and the U.S. used observational data and a novel quantitative model of airborne virus exposure to elucidate how the efficacy of face masks depends on characteristic regimes of airborne virus concentration.
In most situations, even simple surgical masks are effective
“For the airborne transmission of SARS-CoV-2, we find that usually just a minor fraction of exhaled respiratory particles contains viruses. Most environments and contacts are under virus-limited conditions, where face masks, including simple surgical masks, have a high efficacy in preventing the spread of COVID-19,” explains Yafang Cheng, the head of a Minerva Research Group at the MPIC. “Our study provides a detailed and novel mechanistic understanding of population-average mask efficacy, which explains why regions with a higher percentage of the population wearing masks have better control of the pandemic.”
In virus-rich indoor environments with high infection probability, however, more advanced masks (N95/FFP2) and other protective equipment are required to prevent airborne transmission. The strong dependence of mask efficacy on airborne virus concentration highlights the importance of combining masks with other protective measures such as ventilation and distancing to keep the infection probability low.
“The combination of high-efficiency masks with other protective measures is particularly important for hospitals, medical centers, and other indoor environments, where high risk patients may encounter high virus concentrations,” says Christian Witt, head of the Research Area Pneumology at the Charité – Universitätsmedizin Berlin. “Masks will remain an important protective measure against SARS-Cov-2 infection—even for vaccinated persons, especially when the protection provided by vaccination decreases over time.” HRS DISAGREES with this last statement, whowing the risk to be 4 in 10,000 acquiring nasal viral colonization & even rare infection, if double vaccinated or previously infected.
The approach can be used to assess protection against more infectious mutants
“Our investigations also show that aerosol transmission does not necessarily lead to very high reproduction numbers as observed for measles, and that relatively low reproduction numbers do not rule out airborne transmission.”
Moreover, the study demonstrates how important high compliance and correct use of masks are to ensure their effectiveness in reducing the reproduction number of COVID-19. To reduce the reproduction number from ~3 as originally observed to below 1, at least 60-70% compliance would be required for surgical masks (~40% for N95/FFP2 masks). Higher rates of compliance would be required for more infectious variants of SARS-CoV-2.
“Our study explains quantitatively why and how face masks are highly effective in virus-limited environments and less effective in virus-rich environments—both at the individual and the population average level related to observed infection rates and effective reproduction numbers. This has not been achieved before and is essential to overcome inconclusive earlier results, arguments, and discussions. We are confident, that the mechanistic insights and quantitative results gained in our study constitute a scientific breakthrough that will help to settle the ongoing debate about the usefulness of masks and promote efficient mitigation of the COVID pandemic,” summarizes Ulrich Pöschl, director of the MPIC Multiphase Chemistry Department.
More information: Yafang Cheng et al, Face masks effectively limit the probability of SARS-CoV-2 transmission, Science (2021). DOI: 10.1126/science.abg6296
New SARS-CoV-2 Test Is a Simple, Cost-Effective, & Efficient Alternative for SARS-CoV-2 Testing 11/18/2020
ScienceDaily.com
Scientists from Northwell Health Laboratories have developed a new diagnostic multiplex assay that can be used for epidemiological surveillance and clinical management of COVID-19. The Northwell Health Laboratories laboratory-developed test (NWHL LDT) uses a different set of reagents than current assays and can test 91 patients at a time for SARS-CoV-2, versus a maximum of 29 patients using the modified Centers for Disease Control and Prevention (CDC) assay. The NWHL LDT performs as well as the modified CDC test with comparable analytical specificity and accuracy, report scientists in The Journal of Molecular Diagnostics.
G J Berry, PhD, Infectious Disease Diagnostics, Northwell Health Laboratories, USA School of Medicine at Hofstra/Northwell, Hempstead, NY, USA. The CDC initially developed the most-widely used NAAT assay, which includes primers and probes to detect the N1 and N2 regions of the nucleocapsid gene, a protein that plays a key role in virus enhancement, and also the human RNAse P gene to monitor RNA extraction and ensure specimen quality.
Dr. Berry and Wei Zhen, PhD, also based at Infectious Disease Diagnostics, Northwell Health Laboratories, developed the one-step real-time qualitative RT-PCR NWHL LDT test using the 7500 Fast Dx real-time PCR instrument. The NWHL LDT assay targets the S gene of SARS-CoV-2 and uses the same primers and probes for assay internal control as the modified CDC assay test.
A limit of detection (LOD) study of the NWHL LDT with inactivated virus exhibited equal performance with the modified CDC assay, with a final LOD of 1,301 ±13 genome equivalents for the NWHL LDT compared to 1,249 ± for the modified CDC assay. A clinical evaluation with 270 nasopharyngeal swab specimens from individuals suspected of having COVID-19 exhibited 98.5 percent positive agreement and 99.3 percent negative agreement compared to the modified CDC assay.
The NWHL NDT also showed significant efficiencies over the CDC assay, since the test requires only one set of primer and probe mix per specimen, compared to three sets and the use of three wells for each patient in the modified CDC assay.
The authors observed that the NWHL LDT is a single site evaluation with a single target gene, while there has been a trend toward dual-target design in commercial assays for detection of the highly contagious SARS-CoV2 pathogen.
“Ultrapotent COVID-19 vaccine candidate designed via computer
by U of Washington 10/22/20
Coronavirus proteins are added to a computer-designed nanoparticle platform to create a candidate vaccine against COVID-19 making neutralizing antibodies targeting multiple different sites on the Spike protein. The vaccine candidate was designed and tested in animal models by researchers at the University of Washington School of Medicine Credit: Ian Haydon/UW Medicine Institute for Protein Design
Compared to vaccination with the soluble SARS-CoV-2 Spike protein, which is what many leading COVID-19 vaccine candidates are based on, the new nanoparticle vaccine produced ten times more neutralizing antibodies in mice, even at a six-fold lower vaccine dose + a strong B-cell response critical for immune memory and a durable vaccine effect. This may ensure protection against mutated strains of the virus. Published in Cell lead authors are Alexandra Walls, a research scientist in the laboratory of David Veesler, who is an associate professor of biochemistry at the UW School of Medicine; and Brooke Fiala, a research scientist in the laboratory of Neil King, who is an assistant professor of biochemistry at the UW School of Medicine.
The vaccine candidate was developed using structure-based vaccine design techniques invented at UW Medicine. It is a self-assembling protein nanoparticle that displays 60 copies of the SARS-CoV-2 Spike protein’s receptor-binding domain in a highly immunogenic array. The molecular structure of the vaccine roughly mimics that of a virus, which may account for its enhanced ability to provoke an immune response. Alexandra C. Walls et al, Elicitation of potent neutralizing antibody responses by designed protein nanoparticle vaccines for SARS-CoV-2, Cell (2020)”. DOI: 10.1016/j.cell.2020.10.043
10/19/20: “The latest time-points we tracked in infected individuals were past seven months, so that is the longest period of time we can confirm immunity lasts,” Dr. Bhattacharya said. “That said, we know that people who were infected with the first SARS coronavirus, which is the most similar virus to SARS-CoV-2, are still seeing immunity 17 years after infection. If SARS-CoV-2 is anything like the first one, we expect antibodies to last at least two years, and it would be unlikely for anything much shorter.” The study began when Drs. Nikolich-Zugich and Bhattacharya, both members of the UArizona BIO5 Institute, led a UArizona Health Sciences team’
‘Half-measure’ virus vaccine intrigues experts
by Kelly MacNamara 11/23/20 in Medical Xpress
Evidence suggesting an initial half dose of the vaccine being developed by AstraZeneca and the University of Oxford is more effective than a full dose is counterintuitive, and even took the researchers by surprise.
Andrew Pollard, the director of the Oxford Vaccine Group, described the findings from the Phase 3 clinical trial as “intriguing”.
They showed that the vaccine had an efficacy of 62 percent among the people given two full doses a month apart.
But this rose to 90 percent for another group who received a half-dose first and then a full dose after a month.
Stem Cell infusion helps repair COVID-19 damage in severe cases
by University of Miami Leonard M. Miller School of Medicine 1/5/21
Camillo Ricordi, M.D., director of the Diabetes Research Institute (DRI) and Cell Transplant Center at the U of Miami Miller School of Medicine
Umbilical cord derived mesenchymal stem cell infusions safely reduce risk of death and quicken time to recovery for the severest COVID-19 patients, according to results published in STEM CELLS Translational Medicine in January 2021. Camillo Ricordi, M.D., director of the Diabetes Research Institute (DRI) and Cell Transplant Center
24 patients hospitalized at U of Miami Tower or Jackson Memorial Hospital with COVID-19 who developed severe acute respiratory distress syndrome. Each received 2 infusions given days apart of either mesenchymal stem cells or placebo. “It was a double-blind study. . “Two infusions of 100 million stem cells were delivered within three days, for a total of 200 million cells in each subject in the treatment group.” The treatment was safe, with no infusion-related serious adverse events.
Patient survival at one month was 91% in the stem cell treated group versus 42% in the control group. Among patients younger than 85 years old, 100% of those treated with mesenchymal stem cells survived at one month. Dr. Ricordi and colleagues also found time to recovery was faster among those in the treatment arm. More than half of patients treated with mesenchymal stem cell infusions recovered and went home from the hospital within two weeks after the last treatment. More than 80% of the treatment group recovered by day 30, versus less than 37% in the control group.
Mesenchymal cells not only help correct immune and inflammatory responses that go awry, they also have antimicrobial activity and have been shown to promote tissue regeneration.
“Our results confirm the powerful anti-inflammatory, immunomodulatory effect of UC-MSC. These cells have clearly inhibited the ‘cytokine storm’, a hallmark of severe COVID-19,” said Giacomo Lanzoni, Ph.D, lead author. When given intravenously, mesenchymal stem cells migrate naturally to the lungs. That’s where therapy is needed in COVID-19 patients with acute respiratory distress syndrome, a dangerous complication associated with severe inflammation and fluid buildup in the lungs.
STEM CELLS Translational Medicine (2021). dx.doi.org/10.1002/sctm.20-0472
Researchers discover drug that blocks multiple SARS-CoV-2 variants in mice
by Lauren Ingeno, Perelman School of Medicine at the University of Pennsylvania
Lung epithelial cells infected with SARS-CoV-2 (left, yellow) were successfully treated with the STING agonist diABZI s.
The drug diABZI—which activates the body’s innate immune response—was highly effective in preventing severe COVID-19 in mice that were infected with SARS-CoV-2, according to scientists in the Perelman School of Medicine at the University of Pennsylvania. The findings, published this month in Science Immunology, suggest that diABZI could also treat other respiratory coronaviruses.
“Few drugs have been identified as game-changers in blocking SARS-CoV-2 infection. This paper is the first to show that activating an early immune response therapeutically with a single dose is a promising strategy for controlling the virus, including the South African variant B.1.351, which has led to worldwide concern,” said senior author Sara Cherry, Ph.D., a professor of Pathology and Laboratory Medicine and scientific director of the High-Throughput Screening (HTS) Core at Penn Medicine.
The SARS-CoV-2 virus initially targets epithelial cells in the respiratory tract. As the first line of defense against infection, the respiratory tract’s innate immune system recognizes viral pathogens by detecting their molecular patterns. The virus is able to hide, delaying the immune system’s early recognition and response.
To identify antiviral agonists that would block SARS-CoV-2 infection, the researchers performed high throughput screening: two cyclic dinucleotides (CDNs) significantly suppressed infection by activating STING (the simulation of interferon genes).
SCDNs have low potency and make poor drugs. Cherry tested a newly-developed small molecule STING agonist called diABZI, which is not approved by the Food and Drug Administration but is currently being tested in clinical trials to treat some cancers. The researchers found that diABZI potently inhibits SARS-CoV-2 infection of diverse strains, by stimulating interferon signaling.
diABZI was administered through a nasal delivery. diABZI-treated mice showed much less weight loss than the control mice, had significantly-reduced viral loads in their lungs and nostrils, and increased cytokine production—all supporting the finding that diABZI stimulates interferon for protective immunity.
Additionally, since diABZI has been shown to inhibit human parainfluenza virus and rhinovirus replication in cultured cells.
More information: Minghua Li et al, Pharmacological activation of STING blocks SARS-CoV-2 infection, Science Immunology (2021). DOI: 10.1126/sciimmunol.abi9007
A Supercomputer Analyzed Covid-19 —a new theory: corona virus may be a blood vessel disease due to BRADYKININ
by Thomas Smith The Summit supercomputer at Oak Ridge National Lab in Tennessee set about crunching data on more than 40,000 genes from 17,000 genetic samples in an effort to better understand Covid-19. Summit is the second-fastest computer in the world, the process involved analyzing 2.5 billion genetic combinations took more than a week.
Dr. Daniel Jacobson at Oak Ridge, had an “eureka moment.” The computer revealed a new theory about how Covid-19 impacts the body: the bradykinin hypothesis and suggests 10 + potential treatments, many already FDA approved. Jacobson published in eLife 7/2020. Covid-19 infection begins as the virus enters the body through ACE2 receptors in the nose and then proceeds throughout the body where ACE2 is also present: the intestines, kidneys, and heart. Covid-19 up-regulates ACE2 receptors in places where they’re usually expressed at low or medium levels, including the lungs
The renin–angiotensin system (RAS) controls a chemical called bradykinin regulates blood pressure. When the virus tweaks the RAS, it causes the body’s mechanisms for regulating bradykinin to go haywire. Bradykinin receptors are re-sensitized: the body stops effectively breaking down bradykinin. (ACE normally degrades bradykinin, but when the virus downregulates ACE, it can’t do this as effectively.)
The end result, the researchers say, is to release a bradykinin storm — a massive, runaway buildup of bradykinin in the body. This bradykinin hypothesis is that this storm that is ultimately responsible for many of Covid-19’s deadly effects. Jacobson’s team says in their paper that “the pathology of Covid-19 is likely the result of Bradykinin Storms rather than cytokine storms,” which had been previously identified in Covid-19 patients, but that “the two may be intricately linked.” Other papers had previously identified bradykinin storms as a possible cause of Covid-19’s pathologies.
As bradykinin builds up in the body, it dramatically increases vascular permeability. In short, it makes your blood vessels leaky. This aligns with recent clinical data, which increasingly views CoV2-19 PRIMARILY AS AS VASCULAR DISEASE rather than a respiratory one. But Covid-19 still has a massive effect on the lungs. As blood vessels start to leak due to a bradykinin storm, the researchers say, the lungs can fill with fluid. Immune cells also leak out into the lungs, Jacobson’s team found, causing inflammation.
And Covid-19 increases production of hyaluronic acid (HLA) in the lungs. HLA is often used in soaps and lotions for its ability to absorb more than 1,000 times its weight in fluid. When it combines with fluid leaking into the lungs, the results are disastrous: It forms a hydrogel, which can fill the lungs in some patients. According to Jacobson, once this happens, “it’s like trying to breathe through Jell-O.” This may explain why ventilators have proven less effective in treating advanced Covid-19 than originally expected, based on experiences with other viruses: “regardless of how much oxygen you pump in, it doesn’t matter, because the alveoli in the lungs are filled with this hydrogel,” Jacobson says.
Dizziness, seizures, delirium, and stroke are present in half of hospitalized Covid-19 patients. MRI studies in France show evidence of leaky blood vessels in brains. Bradykinin — especially at high doses — can lead to a breakdown of the blood-brain barrier. Under normal circumstances, this barrier acts as a filter between the brain and the rest of the circulatory system. Similar neurological symptoms have been observed in other diseases that result from an excess of bradykinin.
ACE inhibitors, a class of drugs used to treat high blood pressure, have a similar effect on the RAS system as Covid-19, increasing bradykinin levels. In fact, Jacobson and his team note in their paper that “the virus… acts pharmacologically as an ACE inhibitor” — almost directly mirroring the actions of these drugs. ACE inhibitors are also known to cause a loss of taste and smell. Jacobson stresses, though, that this symptom is more likely due to the virus “affecting the cells surrounding olfactory nerve cells” than the direct effects of bradykinin.
The leaky vasculature caused by bradykinin storms could be responsible for “Covid toes,” a condition involving swollen, bruised toes that some Covid-19 patients experience. Bradykinin can also affects the thyroid gland, which could produce the thyroid symptoms recently observed in some patients.
The bradykinin hypothesis could also explain some of the broader demographic patterns of the disease’s spread. The researchers note that some aspects of the RAS system are sex-linked, with proteins for several receptors (such as one called TMSB4X) located on the X chromosome. This means that “women… would have twice the levels of this protein than men,” a result borne out by the researchers’ data. In their paper, Jacobson’s team concludes that this “could explain the lower incidence of Covid-19 induced mortality in women.” A genetic quirk of the RAS could be giving women extra protection against the disease.
Several drugs target aspects of the RAS and are already FDA approved to treat other conditions. They could arguably be applied to treating Covid-19 as well. Several, like danazol, stanozolol, and ecallantide, reduce bradykinin production and could potentially stop a deadly bradykinin storm. Others, like icatibant, reduce bradykinin signaling and could blunt its effects once it’s already in the body. Lanadelumab (Takhzyro) is similar to, but longer acting, than icatibant.
Vitamin D as a potentially useful Covid-19 drug. The vitamin is involved in the RAS system and could prove helpful by reducing levels of another compound, known as REN. Again, this could stop potentially deadly bradykinin storms from forming. The researchers note that vitamin D has already been shown to help those with Covid-19. CONTINUED BELOW
JAMA Network Open
October 27, 2020
Reported 11/16/20 in Medical Xpress: Oak Ridge also identified that the available hepatis C medications boceprevir and narlepravir fit into the flexible heart shaped CoV2-19 protease, stopping its ability to cut proteins in order to reproduce itself.
Original Investigation
September 17, 2020
In Vitro Efficacy of a Povidone-Iodine Nasal Antiseptic for Rapid Inactivation of SARS-CoV-2
Samantha Frank, MD1; Seth M. Brown, MD, MBA1,2; Joseph A. Capriotti, MD3; et al Jonna B. Westover, PhD4; Jesse S. Pelletier, MD5; Belachew Tessema, MD1,2
JAMA Otolaryngol Head Neck Surg. Published online September 17, 2020. doi:10.1001/jamaoto.2020.3053
Question What is the minimum contact time of povidone-iodine (PVP-I) nasal antiseptic required for inactivation of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in vitro?
Findings In this controlled in vitro laboratory research study, test media infected with SARS-CoV-2 demonstrated complete inactivation of SARS-CoV-2 by concentrations of PVP-I nasal antiseptic as low as 0.5% after 15 seconds of contact, as measured by a log reduction value of greater than 3 log10 of the 50% cell culture infectious dose of the virus.
Meaning Intranasal PVP-I rapidly inactivates SARS-CoV-2 and may play an adjunctive role in mitigating viral transmission beyond personal protective equipment.
Abstract
Importance Research is needed to demonstrate the efficacy of nasal povidone-iodine (PVP-I) against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
Objective To evaluate the in vitro efficacy of PVP-I nasal antiseptic for the inactivation of SARS-CoV-2 at clinically significant contact times of 15 and 30 seconds.
Interventions The SARS-CoV-2, USA-WA1/2020 strain, virus stock was tested against nasal antiseptic solutions consisting of aqueous PVP-I as the sole active ingredient. Povidone-iodine was tested at diluted concentrations of 0.5%, 1.25%, and 2.5% and compared with controls. The test solutions and virus were incubated at mean (SD) room temperature of 22 (2) °C for time periods of 15 and 30 seconds.
Design and Setting This controlled in vitro laboratory research study used 3 different concentrations of study solution and ethanol, 70%, as a positive control on test media infected with SARS-CoV-2. Test media without virus were added to 2 tubes of the compounds to serve as toxicity and neutralization controls. Ethanol, 70%, was tested in parallel as a positive control and water only as a negative control.
Results Povidone-iodine nasal antiseptics at concentrations (0.5%, 1.25%, and 2.5%) completely inactivated SARS-CoV-2 within 15 seconds of contact as measured by log reduction value of greater than 3 log10 of the 50% cell culture infectious dose of the virus. The ethanol, 70%, positive control did not completely inactivate SARS-CoV-2 after 15 seconds of contact. The nasal antiseptics tested performed better than the standard positive control routinely used for in vitro assessment of anti–SARS-CoV-2 agents at a contact time of 15 seconds. No cytotoxic effects on cells were observed after contact with each of the nasal antiseptics tested.
Conclusions and Relevance Povidone-iodine nasal antiseptic solutions at concentrations as low as 0.5% rapidly inactivate SARS-CoV-2 at contact times as short as 15 seconds. Intranasal use of PVP-I has demonstrated safety at concentrations of 1.25% and below and may play an adjunctive role in mitigating viral transmission beyond personal protective equipment.
Introduction
Nasal goblet and ciliated cells have the highest expression of angiotensin-converting enzyme 2 (ACE2), which is the main receptor for SARS-CoV-2.2 Recently, Hou et al6 showed that ciliated cells with ACE2 expression were the cells most susceptible to infection, rather than submucosal glandular cells.
Multiple protocols have come forth recommending intranasal use of povidone-iodine (PVP-I) in patients and health care workers.9-12 Povidone-iodine was selected given its proven in vitro efficacy against SARS-CoV and Middle East respiratory syndrome at concentrations as low as 0.23%.13,14 In vitro efficacy of an oral PVP-I antiseptic solution was recently demonstrated specifically against SARS-CoV-2 at concentrations as low as 0.5% for contact times as short as 15 seconds.15
Methods
The nasal rinse antiseptic solution consisted of various concentrations of aqueous PVP-I as the sole active ingredient (Veloce BioPharma)…. serially diluted using 8 log dilutions in test medium.
Results
Virus titers and LRV of SARS-CoV-2 when incubated with various concentrations of the manufacturer’s compounds for 15 seconds are summarized in Table 1. After the 15-second contact time, all of the PVP-I nasal rinse antiseptics tested were effective at reducing greater than 3 log10 CCID50 infectious virus, from 3.67 log10 CCID50/0.1 mL to 0.67 log10 CCID50/0.1 mL or less.
Discussion
This study demonstrates rapid inactivation of SARS-CoV-2 by PVP-I at concentrations as low as 0.5% for as little as 15 seconds of contact. These findings are consistent with those of a previous study investigating efficacy of an oral solution in the same class of PVP-I antiseptics against SARS-CoV-2.15 Clinical studies have demonstrated that lower concentrations can be administered acutely and over a period of months with no adverse effects.19 We have implemented the use of intranasal PVP-I in our practice and have updated all of our protocols to include use of 1.25% aqueous PVP-I formulations delivered to each nasal cavity in patients before any intranasal procedure.
This study demonstrates that a contact time of 15 seconds is sufficient for viral inactivation. Widespread use of PVP-I nasal antiseptic in patients prior to intranasal procedures could significantly decrease risk of virus transmission via droplet and aerosol spread. Health care professionals may also consider instructing patients to perform nasal decontamination with PVP-I prior to presenting for their procedure, which can further decrease intranasal viral load and can prevent spread in waiting areas and other common areas.
Nasal PVP-I irrigations should additionally be considered for use by health care professionals for prophylaxis. Oral mucosa decontaminated with PVP-I remains sterilized for up to 4 hours.25 Although this has not yet been proven in nasal mucosa, health care providers should consider use every 4 hours, or whenever donning or doffing a mask in high risk settings, up to 4 times daily. At concentrations of 1.25%, iodine absorption is negligible. These simple, nonbuffered, slightly acidic, complexed PVP-I solutions would further limit any transmucosal absorption of molecular iodine, providing only a minimal theoretical risk of iodine absorption. Even if some noncomplexed iodine were absorbed trans-mucosally, it would still be orders of magnitude less than the average total daily iodine intake for a healthy adult of 150 μg.19 Use of 0.08% nasal PVP-I every other day for up to 7 weeks does not result in clinical thyroid disease.20,21 Nevertheless, thyroid function testing should be considered when PVP-I is regularly administered to patients for more than 3 months. Use of intranasal PVP-I is contraindicated in patients with an allergy to iodine, patients who are pregnant, patients with active thyroid disease, and patients undergoing radioactive iodine therapy.26-28
Accepted for Publication: July 29, 2020.
Corresponding Author: Samantha Frank, MD, Division of Otolaryngology−Head and Neck Surgery, Department of Surgery, University of Connecticut School of Medicine, 263 Farmington Ave, Farmington, CT 06030 ([email protected]).
Published Online: September 17, 2020. doi:10.1001/jamaoto.2020.3053
RESTARTING CONTINUATION. Hymecromone could reduce hyaluronic acid levels, potentially stopping deadly hydrogels from forming in the lungs. And timbetasin could mimic the mechanism that the researchers believe protects women from more severe Covid-19 infections.
Baricitinib plus Remdesivir for Hospitalized Adults with Covid-19 NEJM 12/11/20
• Andre C. Kalil, MD, MPH, et al
… randomized, double-blind, placebo-controlled trial show that combination treatment with the anti-inflammatory drug baricitinib and the antiviral drug remdesivir was safe and superior to remdesivir alone for the treatment of hospitalized patients with Covid-19 pneumonia. The beneficial effects of the combination treatment were seen both in the primary outcome, with a 1-day shorter time to recovery, and in the key secondary outcome, with a greater improvement in clinical status as assessed on the ordinal scale …. both the survival rate and the time-to-death analyses favored combination treatment. These clinical benefits were observed across different age groups, sexes, ethnic groups, and races and were independent of symptom duration or disease severity at enrollment. The large proportion of Hispanic or Latino patients who were enrolled in the trial reflects the disproportionate effect of the pandemic on racial and ethnic minorities with respect to high incidences of hospitalization.14 “A majority of adult COVID-19 hospitalizations nationwide are attributable to at least one of four pre-existing conditions: obesity, hypertension, diabetes, and heart failure, in that order.”
The observed benefit of combination treatment was most evident in patients with a baseline ordinal score of 5 (supplemental oxygen) or 6 (high-flow oxygen or noninvasive ventilation), among whom the median time to recovery was, respectively, 1 and 8 days sooner with combination treatment than with placebo. Patients with a baseline ordinal score of 6 who received combination treatment were twice as likely as those in the control group to have improved clinical status at day 15 (odds ratio, 2.2; 95% CI, 1.4 to 3.6). The faster recovery in patients who received baricitinib plus remdesivir suggests that the combination treatment may have an effect in lowering the hospital-associated risk of nosocomial infections, thrombosis, and errors in hospital drug administration. Moreover, faster recovery also decreases the burden on the health care system, potentially increasing capacity, which is of critical importance during a surge of cases.
In addition, the combination treatment showed clinical benefits directly relevant to patient care, such as a difference of −17.4 percentage points in new use of oxygen (22.9% vs. 40.3%) and a difference of −5.2 percentage points in new use of mechanical ventilation or ECMO (10.0% vs. 15.2%). In fact, the odds of progression to death or invasive ventilation were 31% lower in the combination group than in the control group (hazard ratio, 0.69; 95% CI, 0.50 to 0.95), and patients in the combination group had 11 fewer days receiving new mechanical ventilation than those in the control group.
FDA authorizes monoclonal antibodies for treatment of COVID-19
World Pharma News February 10, 2021
Today, the U.S. Food and Drug Administration issued an emergency use authorization (EUA) for bamlanivimab and etesevimab administered together for the treatment of mild to moderate CoV2-19 in adults and pediatric patients (12 years of age or older weighing at least 40 kilograms [about 88 pounds]) who test positive for SARS-CoV-2 and who are at high risk for progressing to severe CoV2-19. The authorized use includes treatment for those who are 65 years of age or older or who have certain chronic medical conditions.
In a clinical trial of patients with COVID-19 at high risk for disease progression, a single intravenous infusion of bamlanivimab and etesevimab administered together significantly reduced CoV2-19-related hospitalization by 87% and no deaths during 29 days of follow-up compared to placebo.
Bamlanivimab and etesevimab are now authorized for patients who are hospitalized due to COVID-19 or require oxygen therapy due to COVID-19. Treatment with bamlanivimab and etesevimab has not been studied in patients hospitalized due to CoV2-19: worse clinical outcomes when administered to hospitalized patients with COVID-19 requiring high flow oxygen or mechanical ventilation.
CBD (cannabidiol) helps reduce lung damage from COVID by increasing levels of protective peptide: apelin
by Medical College of Georgia at Augusta University October 19, 2020
Dr. Babak Baban, DCG immunologist and associate dean for research and Dr. Jack Yu, physician scientist and chief of pediatric plastic surgery at MCG. Apelin and ACE2 also normally work together to control blood pressure, and upregulation of both may be helpful in cardiovascular disease, including heart failure, by decreasing blood pressure while increasing the heart’s ability to pump. In fact, apelin and ACE2 work together to regulate a healthy cardiovascular system and they are factors in pretty much any condition, like obesity or hypertension, that hurt the cardiovascular system, Baban says.
Like other disease, the novel coronavirus appears to upset their positive partnership. The virus’ binding to the receptor for ACE2 has been shown to decrease ACE2 levels and increase levels of the powerful blood vessel constrictor angiotensin II, because less angiotensin II gets degraded and fewer vasodilators get produced. The new finding was their first in learning more about how CBD produces the beneficial effects they saw in their model of ARDS.
Likely the virus suppresses something that suppresses apelin, they say and CBD interferes. But they doubt the apelin-CBD interaction is the only way the compound, the second most prevalent found in the marijuana plant, works in this and other scenarios. They reported this summer in the journal Cannabis and Cannabinoid Research that treatment with CBD reduced excessive lung inflammation, enabling improvements in lung function, heathier oxygen levels, and repair of some of the structural damage to the lungs that are classic with ARDS.
Anti-COVID-19 nasal spray ‘ready for use in humans’
November 19, 2020
A nasal spray that can provide effective protection against the COVID-19 virus has been developed by researchers at the U of Birmingham using products already & readily available.
The normal complex procedures to take a new product to market are greatly simplified, so the spray could be commercially available very quickly.
A pre-print (not yet peer-reviewed) study describes cell culture experiments designed to test the ability of the solution to inhibit infection. They found cell-virus cultures inhibited the infection up to 48 hours after being treated with the solution even when diluted many times.
The spray is composed of two polysaccharide polymers. The first, an antiviral agent called carrageenan, is commonly used in foods as a thickening agent, while the second a solution called gellan, was selected for its ability to stick to cells inside the nose.
The gellan, is an important component because it has the ability to be sprayed into fine droplets inside the nasal cavity, where it can cover the surface evenly, and stay at the delivery site, rather than sliding downwards and out of the nose.
The spray works in two primary ways. Firstly, it catches and coats the virus inside the nose, from where it can be eliminated via the usual routes—either nose-blowing or swallowing. Secondly, because the virus is encapsulated in the spray’s viscous coating, it is prevented from being uptaken by the body. That means it will reduce the viral load in the body, but also even if virus particles are passed on to another person via a sneeze or cough, that person is less likely to be infected by active virus particles.
The spray can also prevent the virus being passed from person to person.”
The team believe the spray could be particularly useful in areas where crowding is less avoidable, such as airplanes or classrooms. Regular application of the spray could significantly reduce disease transmission.
“Products like these don’t replace existing measures such as mask wearing and handwashing, which will continue to be vital to preventing the spread of the virus,” adds Dr. Moakes. “What this spray will do, however, is add a second layer of protection to prevent and slow virus transmission.”
More information:
Richard J. A. Moakes et al. Formulation of a composite nasal spray enabling enhanced surface coverage and prophylaxis of SARS-COV-2, (2020). DOI: 10.1101/2020.11.18.388645
FDA Authorizes Use of COVID-19 Antibody Therapy for Postexposure Prophylaxisby Diana Ernst, RPh 8/2/21
The Food and Drug Administration (FDA) has authorized the use of REGEN-COV (casirivimab and imdevimab) for postexposure prophylaxis of COVID-19 in individuals (12 years of age and older weighing at least 40kg) who are at high risk for progression to severe COVID-19. It was previously granted Emergency Use Authorization (EUA) for the treatment of mild to moderate COVID-19.
REGEN-COV consists of 2 potent, virus-neutralizing antibodies that bind noncompetitively to the receptor binding domain of the SARS-CoV-2 spike protein. Study: asymptomatic individuals who lived in the same household with a SARS-CoV-2 infected patient. A single subcutaneous or intravenous dose of casirivimab 600mg and imdevimab 600mg within 96 hours of a positive index cases of SARS-CoV-2.
Results: REGEN-COV reduced the risk of COVID-19 development by 81% in the primary analysis population (RT-qPCR negative and seronegative at baseline) compared: P <.0001). In a post-hoc analysis, a 62% reduction in risk was observed in the overall trial population.
1. FDA expands authorized use of REGEN-COV™ (casirivimab and imdevimab). News release.. Accessed August 2, 2021. https://www.prnewswire.com/news-releases/fda-expands-authorized-use-of-regen-cov-casirivimab-and-imdevimab-301345336.html.
2. REGEN-COV fact sheet for health care providers. Accessed August 2, 2021. https://www.regeneron.com/downloads/treatment-covid19-eua-fact-sheet-for-hcp.pdf.
Defeating pathological autoimmunity with kinase inhibition
by John Hewitt , Medical Xpress 12/21/20
HDAC “… several HDAC inhibitors could bind to human angiotensin I converting enzyme 2 (ACE2) on the cell surface, which in turn resulted in overall structural changes of ACE2. Since SARS-CoV-2 recognizes human ACE2 receptor by its spike protein during viral infection, such alternations inhibited the ACE2-S protein binding and prevented host cell entry of SARS-CoV-2.
Inspired by this result, the team then screened 18 commercially available HDAC inhibitors and studied their efficacy in inhibiting the entry of SARS-CoV-2 into cells. They found that four inhibitors, i.e., panobinostat, givinostat hydrochloride monohydrate, CAY10603 and sirtinol are noticeably effective…”
Top of Form
November 12, 2020
Fluvoxamine (Luvox) vs Placebo and Clinical Deterioration in Outpatients With Symptomatic COVID-19A Randomized Clinical Trial
Eric J. Lenze, MD, et al
https://jamanetwork.com/journals/jama/fullarticle/2773108?utm_source=silverchair&utm_campaign=jama_network&utm_content=covid_weekly_highlights&utm_medium=email
In this randomized trial that included 152 adult outpatients with confirmed COVID-19 and symptom onset within 7 days, clinical deterioration occurred in 0 patients treated with fluvoxamine vs 6 (8.3%) patients treated with placebo over 15 days, a difference that was statistically significant.
Design, Setting, and Participants Double-blind, randomized, fully remote (contactless) clinical trial of fluvoxamine vs placebo. Participants were community-living, non-hospitalized adults with confirmed severe acute respiratory syndrome coronavirus 2 infection, with COVID-19 symptom onset within 7 days and oxygen saturation of 92% or greater.
Interventions Participants were randomly assigned to receive 100 mg of fluvoxamine (n = 80) or placebo (n = 72) 3 times daily for 15 days.
Clinical deterioration occurred in 0 of 80 patients in the fluvoxamine group and in 6 of 72 patients in the placebo group (absolute difference, 8.7% [95% CI, 1.8%-16.4%] from survival analysis; log-rank P = .009). The fluvoxamine group had 1 serious adverse event and 11 other adverse events, whereas the placebo group had 6 serious adverse events and 12 other adverse events.
Conclusions and Relevance In this preliminary study of adult outpatients with symptomatic COVID-19, patients treated with fluvoxamine, compared with placebo, had a lower likelihood of clinical deterioration over 15 days.
Coronavirus disease 2019/COVID-19 Clinical deterioration typically occurs during the second week of illness. Early studies of COVID-19 found that hospitalization most often occurs within 8 to 10 days of initially mild to moderate symptoms.2-4
A potential mechanism for immune modulation is σ-1 receptor (S1R) agonism.7 The S1R is an endoplasmic reticulum chaperone protein with various cellular functions, including regulation of cytokine production through its interaction with the endoplasmic reticulum stress sensor inositol-requiring enzyme 1α (IRE1). Previous studies have shown that fluvoxamine, a selective serotonin reuptake inhibitor (SSRI) with high affinity for the S1R,8 reduced damaging aspects of the inflammatory response during sepsis through the S1R-IRE1 pathway, and decreased shock in murine sepsis models.9
Fluvoxamine is a strong S1R agonist,10,11 is highly lipophilic, and has rapid intracellular uptake.12 This study tested whether fluvoxamine, given as early treatment in individuals with mild COVID-19 illness, may prevent clinical deterioration.
This was a double-blind, placebo-controlled, randomized clinical trial that compared fluvoxamine with placebo in adult outpatients with confirmed SARS-CoV-2 infection.
The study included adults living in the community with SARS-CoV-2 infection confirmed by polymerase chain reaction assay and who were symptomatic within 7 days of the first dose of study medication (Figure 1).
Participants received a dose of 50 mg of fluvoxamine (or matching placebo) in the evening immediately after the baseline assessment and confirmation of eligibility, then for 2 days at a dose of 100 mg twice daily as tolerated, and then increasing to a dose of 100 mg 3 times daily as tolerated through day 15 then stopped.
The most severe presenting COVID-19 symptom varied, with fatigue (23%) and loss of sense of smell (29%) being the most common. The baseline oxygen saturation level did not differ between the groups for fluvoxamine vs 97% for placebo.
Clinical deterioration occurred in 0 of 80 patients in the fluvoxamine group and in 6 of 72 (8.3%) patients in the placebo group (absolute difference, 8.7%. In the placebo group, cases of clinical deterioration ranged from 1 to 7 days after randomization and from 3 to 12 days after the onset of COVID-19 symptoms. Four of 6 patients were hospitalized for COVID-19 illness, with the length of stay ranging from 4 to 21 days. One patient required mechanical ventilation for 10 days (Table 2) and no patients died.
In this preliminary randomized clinical trial, fluvoxamine (an S1R agonist) was associated with a reduction in clinical deterioration in adult outpatients with COVID-19. No fluvoxamine-treated patients met criteria for clinical deterioration as defined in the study, whereas 8.3% of patients taking placebo met this end point.
If fluvoxamine is determined to be effective in treating COVID-19, the underlying mechanism needs further clarification. The study was prompted by a hypothesis involving the influence of fluvoxamine on the S1R-IRE1 pathway. Anti-inflammatory (cytokine reduction) actions resulting from S1R activation would fit with recent findings of benefits of other anti-inflammatory drugs, such as colchicine and corticosteroids, for COVID-19.21,22 However, a recent study found lower levels of cytokines in patients with severe COVID-19 vs patients with bacterial sepsis.23 Alternative mechanisms of a potential fluvoxamine benefit include direct antiviral effects via its lysosomotropic properties,24 modulation of the effect of IRE1 effects on autophagy,25 and SSRI inhibition of platelet activation.26
The potential advantages of fluvoxamine for outpatient treatment of COVID-19 include its safety,27 widespread availability, low cost, and oral administration. Fluvoxamine does not promote QT prolongation unlike other SSRIs.28 However, fluvoxamine has adverse effects and can cause drug-drug interactions, particularly via inhibition of cytochromes P450 1A2 and 2C19.29
Targeting cellular response to SARS-CoV-2 holds promise as new way to fight infection
by University of Cambride 6/17/21
When a person is infected with SARS-CoV-2, the virus that causes COVID-19, it invades their cells and uses them to replicate—which puts the cells under stress. Current approaches to dealing with infection target the virus itself with antiviral drugs. But Cambridge scientists have switched focus to target the body’s cellular response to the virus instead.
In a new study, published today in the journal PLOS Pathogens, they found that all three branches of a three-pronged signalling pathway called the ‘unfolded protein response’ (UPR) are activated in lab-grown cells infected with SARS-CoV-2. Inhibiting the UPR to restore normal cell function using drugs was also found to significantly reduce virus replication.
“The virus that causes COVID-19 activates a response in our cells—called the UPR—that enables it to replicate,” said Dr. Nerea Irigoyen in the University of Cambridge’s Department of Pathology, senior author of the report.
She added: “Using drugs we were able to reverse the activation of this specific cellular pathway, and remarkably this reduced virus production inside the cells almost completely, which means the infection could not spread to other cells. This has exciting potential as an anti-viral strategy against SARS-CoV-2.”
Treatment with a drug that targets one prong of the UPR pathway had some effect in reducing virus replication. But treatment with two drugs together—called Ceapin-A7 and KIRA8—to simultaneously target two prongs of the pathway reduced virus production in the cells by 99.5%. This is the first study to show that the combination of two drugs has a much greater effect on virus replication in cells than a single drug.
The approach would not stop a person getting infected with the coronavirus, but the scientists say symptoms would be much milder, and recovery time would be quicker.
The new treatment targets the response of the infected cells; this will not change even if new variants emerge, because the virus needs this cellular response in order to replicate.
The scientists also want to see whether it works against other viruses, and illnesses such as pulmonary fibrosis and neurological disorders that also activate the UPR response in cells.
More information: PLOS Pathogens (2021). DOI: 10.1371/journal.ppat.1009644
From the NIH Director’s Blog 4/24/21:
The Montreal Heart Institute, Canada, announced preliminary results of the international COLCORONA trial, testing the use of colchicine—an anti-inflammatory drug widely used to treat gout and other conditions—for patients diagnosed with COVID-19 [4].
Their early findings in treating patients just after a confirmed diagnosis of COVID-19 suggest that colchicine might reduce the risk of death or hospitalization compared to patients given a placebo. In the more than 4,100 individuals with a proven diagnosis of COVID-19, colchicine significantly reduced hospitalizations by 25 percent, the need for mechanical ventilation by 50 percent, and deaths by 44 percent. Still, the actual numbers of individuals represented by these percentages was small.
Top of Form
Bottom of Form
Colchicine for community-treated patients with COVID-19 (COLCORONA): a phase 3, randomised, double-blinded, adaptive, placebo-controlled, multicentre trial
• May 2021, The Lancet Respiratory Medicine
• DOI:
• 10.1016/S2213-2600(21)00222-8
• Jean-Claude Tardif, et al
Abstract
Background Evidence suggests a role for excessive inflammation in COVID-19 complications. Colchicine is an oral anti-inflammatory medication beneficial in gout, pericarditis, and coronary disease. We aimed to investigate the effect of colchicine on the composite of COVID-19-related death or hospital admission. Methods The present study is a phase 3, randomized, double-blind, adaptive, placebo-controlled, multicentre trial. The study was done in Brazil, Canada, Greece, South Africa, Spain, and the USA, and was led by the Montreal Heart Institute. Patients with COVID-19 diagnosed by PCR testing or clinical criteria who were not being treated in hospital were eligible if they were at least 40 years old and had at least one high-risk characteristic. … Patients were randomly assigned to receive orally administered colchicine (0·5 mg twice per day for 3 days and then once per day for 27 days thereafter) or matching placebo. The primary efficacy endpoint was the composite of death or hospital admission for COVID-19. Vital status at the end of the study was available for 97·9% of patients. The analyses were done according to the intention-to-treat principle. … Findings: Trial enrolment began in March 23, 2020, and was completed in Dec 22, 2020. A total of 4488 patients (53·9% women; median age 54·0 years, IQR 47·0–61·0) were enrolled and 2235 patients were randomly assigned to colchicine and 2253 to placebo. The primary endpoint occurred in 104 (4·7%) of 2235 patients in the colchicine group and 131 (5·8%) of 2253 patients in the placebo group (odds ratio [OR] 0·79, 95·1% CI 0·61–1·03; p=0·081). Among the 4159 patients with PCR-confirmed COVID-19, the primary endpoint occurred in 96 (4·6%) of 2075 patients in the colchicine group and 126 (6·0%) of 2084 patients in the placebo group (OR 0·75, 0·57–0·99; p=0·042). Serious adverse events were reported in 108 (4·9%) of 2195 patients in the colchicine group and 139 (6·3%) of 2217 patients in the placebo group (p=0·051); pneumonia occurred in 63 (2·9%) of 2195 patients in the colchicine group and 92 (4·1%) of 2217 patients in the placebo group (p=0·021). Diarrhoea was reported in 300 (13·7%) of 2195 patients in the colchicine group and 161 (7·3%) of 2217 patients in the placebo group (p<0·0001). Interpretation In community-treated patients including those without a mandatory diagnostic test, the effect of colchicine on COVID-19-related clinical events was not statistically significant. Among patients with PCR-confirmed COVID-19, colchicine led to a lower rate of the composite of death or hospital admission than placebo. Given the absence of orally administered therapies to prevent COVID-19 complications in community-treated patients and the benefit of colchicine in patients with PCR-proven COVID-19, this safe and inexpensive anti-inflammatory agent could be considered for use in those at risk of complications. Notwithstanding these considerations, replication in other studies of PCR-positive community-treated patients is recommended. Funding The Government of Quebec, the Bill & Melinda Gates Foundation, the National Heart, Lung, and Blood Institute of the US National Institutes of Health, the Montreal Heart Institute Foundation, the NYU Grossman School of Medicine, the Rudin Family Foundation, and philanthropist Sophie Desmarais.
From ‘Cardiology Today’ 5/2021: revered U of Michigan Emeritus Professor of Cardiology Bertram Pitt, MD, & Moscow scientist Simon MatskeplishvillI, MD, support studies showing the heart failure treatment/potassium preserving/anti-androgenic/scarring preventive SPIRONOLACTONE is effective for the prevention for CoV2-19 lung scarring by inhibiting the transmembrane serine protease 2 (TMPRSS2) necessary for cell entry of the virus. When combined with PERSANTIN/DIPYRIDAMOLE, this anti-fibrosis/anti lung scarring effect is further enhanced as well as adding effective blood thinning/anti-platelet effect that reduced clotting without increasing bleeding. Measles, mumps-rubella, influenza, and BCG vaccinations also minimize the risk and seriousness of acquired CoV2-19 infection. For the “LONG HAULER” syndrome with multiple abnormally elevated cytokine levels is associated with too high HEART RATE of the Postural Orthostatic Tachycardia Syndrome = POTS, IVADRIBINE and/or BETA BLOCKERS such as BYSTOLIC and others can be helpful with that excess heart rate response while arising.
BMJ F Santoro Anti-platelet therapy reduces CoV2-19 morbidity and mortality: mostly just low dose aspirin. Inhospital, likely combined with full heparin (or ? NOACs) anticoagulation.
https://heart.bmj.com/content/early/2021/10/05/heartjnl-2021-319552
Multispecialty Perspectives on Long COVID-19
Jessica Nye, PhD 6/11/21
Benefits of Antiplatelet Therapy During COVID-19 Hospitalisation Without Increased Bleeding Risk
Heart (British Cardiac Society)10/24/21
https://www.practiceupdate.com/c/124928/2/2/?elsca1=emc_enews_daily-digest&elsca2=email&elsca3=practiceupdate_cardio&elsca4=cardiology&elsca5=newsletter&rid=MjA2NjE0OTY0Mzg2S0&lid=20845073
TAKE-HOME MESSAGE
• The authors reviewed data from 7824 consecutive patients with COVID-19 enrolled in a multicenter international prospective registry for antiplatelet use. Patients treated with antiplatelets showed no differences in terms of in-hospital mortality, need for invasive ventilation, embolic events, and bleeding, but had a shorter duration of mechanical ventilation (8±5 days vs 11±7 days; P = .01). On multivariable analysis, in-hospital antiplatelet therapy was associated with lower mortality risk (RR, 0.39).
• Antiplatelet therapy during hospitalisation for COVID-19 could be associated with lower mortality risk and shorter duration of mechanical ventilation, without an increased risk of bleeding.
Long-term effects associated with COVID-19 infection are being reported across multiple clinical specialties. ‘ … adverse effects include transient symptoms such as fever/chills, headache, fatigue, myalgia/arthralgia, lymphadenopathy, nausea, or local effects of swelling, erythema, or pain.2 …’ )
In the year since the global COVID-19 pandemic was declared in March 2020 by the World Health Organization,1 short-term treatments such as prone positioning of patients2 or the use of neutralizing antibody cocktails3 have been optimized, and strategies for patients who remain symptomatic for months or experience long-term sequelae4,5 are being developed.”
“Although several effective vaccines6-9 have been developed and the end of the pandemic is now within sight, the long and laborious healing process for patients with long COVID and society as a whole is only just beginning.
The Evidence: dysregulated immune activation by the vaccine or its components
A detailed follow-up of 150 patients who were treated at the University of Tours Hospital in France showed that 2 months after symptom onset, more than half (68%) of patients presented with 1 or more lingering symptom of COVID-19. The most common symptoms included diarrhea (33.3%), anosmia/ageusia (22.7%), flu-like symptoms (21.5%), a greater than 5% weight loss (17.2%), arthralgia (16.3%), chest pain (13.1%), cutaneous signs (11.5%), palpitations (10.9%), and dyspnea (7.7%).8
A longer-term, larger study of 1733 Chinese patients who were followed up at 6 months after hospital discharge revealed that most (63%) of the patients experienced fatigue and muscle weakness, and many reported difficulty sleeping (23%), below-average 6-minute walking distances (24%), anxiety or depression (23%), and diffusion impairments (22%).9
The Respiratory System
SARS-CoV-2 infection primarily affects the respiratory tract, causing pneumonia in severe cases with a proinflammatory response.10,11 “We categorize pneumonia as typical or atypical, involving different parts of the lungs. COVID-19 is an atypical pneumonia,” explained Dr Ali Gholamrezanezhad, a radiologist and assistant professor of clinical radiology with Keck Medicine of the University of Southern California, in Los Angeles.
Because COVID-19 may manifest as an atypical pneumonia, patients with severe disease can present with peripheral or posterior distribution of bilateral, multilobar ground-glass opacification, septal and/or pleural thickening, bronchiectasis, and subpleural involvement.11
In the long term, patients who present with complex lung manifestations seen on computed tomographic imaging are at increased risk for tissue scarring. “For patients who needed to be admitted into the hospital for COVID-19, the number [of patients with scarring] can be up to 30% to 40%. In patients who are not admitted, this number is much less, possibly less than 10%. It depends on the severity of the disease,” Dr Gholamrezanezhad added. For example, 6 weeks after discharge from University Hospital RWTH Aachen in Germany, 33 patients who did not require ventilation still presented with symptoms of fatigue (45%), cough (33%), and dyspnea (33%), but there was no evidence of widespread scarring.12
The Cardiovascular System
Dr Valentina Püntmann, consultant physician, cardiologist, clinical pharmacologist, and assistant professor in the Department of Cardiology at the University Hospital Frankfurt, and colleagues recruited 100 patients who had recently recovered from COVID-19. Most patients had acute illness, with only 33% requiring hospitalization (HRS notes that 33% is a high figure and distorts the calculatons of the frequency of adverse events). Using cardiovascular magnetic resonance imaging a median of 71 days after their COVID-19 diagnosis, most patients presented with persistent cardiac involvement.13
“We were surprised to see myocardial inflammation, scarring, and also pericarditis persisting a few months after the original infection.”.
In total, 78% of the patients had abnormal cardiovascular magnetic resonance scans, most commonly elevated myocardial native T1 (73%) and T2 (60%), myocardial late gadolinium (32%), and pericardial (22%) enhancement.13 These results are particularly intriguing, as they were sourced primarily from individuals who recovered at home, indicating that patients who experience even mild symptoms can have persistent myocardial inflammation and scarring for months.
“We are following up with these patients regularly, and for many patients, things do get better. They may not get better very quickly, which is perhaps something that is long-COVID specific,” stated Dr Püntmann. “But there is also a number of patients who don’t get better. I think we definitely need to get much better at understanding why some patients don’t improve, as well as work on developing effective treatments that we can administer early.”
The Nervous System
Throughout the pandemic, hospitalized patients have presented with a wide range of neurologic manifestations, thrombotic events, delirium, seizure-like events, encephalopathy, periodic discharges, ischemic lesions, and white matter lesions, among others.14,15 There has been little to no evidence collected during hospitalization to suggest there is widespread infiltration by the SARS-CoV-2 virus across the blood-brain barrier.14,16-17 With little direct evidence, it has been speculated these presentations occur as a result of neuroinflammation.15,18
During a 3-month follow-up of 61 patients admitted to Rigshospitalet, Copenhagen University Hospital in Denmark, nearly half (45.9%) of the patients had persistent central and peripheral nervous system complications.14
“Many patients actually still have affected cognitive ability. The average MOCA [Montreal Cognitive Assessment] score was 22 out of 30. Particularly, patients [who] were discharged from the intensive care unit had 19.5 out of 30, so they’re affected cognitively,” described Dr Michael E. Benros, professor of immuno-psychiatry in the Department of Immunology and Microbiology at the University of Copenhagen.
Dr Kondziella added, “There are 3 main ways by which COVID-19 might affect the nervous system. First, the virus itself has some sort of neurotropism. That means the virus crosses the blood-brain barrier and enters the brain, where it damages the tissue directly. The other option is that there is an autoimmune response by which cross-reaction toward the virus particles induces a neuroinflammatory pathway in the brain. We did find that in our study to a relatively lesser extent, compared to the third category, which is peripheral nervous system damage not directly caused by the virus or by autoimmune responses, but more because of treatment-related disorders.”
Because many of these nervous system manifestations were attributed to treatment-related complications (n=34 vs unresolved [n=4] vs para/postinfectious [n=3]),14 many of the younger, fitter patients likely will improve over time with rehabilitation after several months, predicted Dr Benros. However, patients who experience stroke or other thrombotic events may experience life-long COVID-19 complications.
Psychiatry
The neurologic presentations observed among patients with COVID-19 may have psychiatric consequences. “Symptoms from long-term COVID-19 are commonly fatigue, headache, insomnia, and brain fog,” stated Dr Samoon Ahmad, professor in the Department of Psychiatry at New York University Grossman School of Medicine and Unit Chief of Inpatient Psychiatry at Bellevue Hospital Center. “However, we don’t know whether these symptoms are neurologic or caused by depression.”
Dr Ahmad explained that approximately 30% of patients who recovered from COVID-19 went on to develop post-traumatic stress disorder (PTSD).19,20 Symptoms of PTSD also have been reported in multiple studies of highly exposed individuals working in the healthcare sector.21
The observation of PTSD among healthcare workers is important because it brings to the forefront the fact that by no means do you need to be infected with the virus to be experiencing psychiatric consequences from SARS-CoV-2.
Dr Ahmad expounded, “Using data from 2019, on average we see that around 8% of people have anxiety disorders, and 6% [have] depressive disorders. IDuring the pandemic, about 4 in 10 adults have reported symptoms of anxiety or depressive disorders.”
References
1. World Health Organization. Report of the WHO-China joint mission on coronavirus disease 2019 (COVID-19). Published February 16-24, 2020. Accessed June 1, 2021. http://who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf
2. Guérin C, Albert RK, Beitler J, et al. Prone position in ARDS patients: why, when, how and for whom. Intensive Care Med. 2020;46(12):2385-2396. doi:10.1007/s00134-020-06306-w
3. Weinreich DM, Sivapalasingam S, Norton T, et al. REGN-COV2, a neutralizing antibody cocktail, in outpatients with Covid-19. N Engl J Med. 2021;384(3):238-251. doi:10.1056/NEJMoa2035002
4. Ella R, Vadrevu KM, Jogdand H, et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBV152: a double-blind, randomised, phase 1 trial. Lancet Infect Dis. 2021;21(5):637-646. doi:10.1016/S1473-3099(20)30942-7
5. Voysey M, Clemens SAC, Madhi SA, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;397(10269):99-111. doi:10.1016/S0140-6736(20)32661-1
6. Baden LR, El Sahly HM, Essink B, et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2021;384(5):403-416. doi:10.1056/NEJMoa2035389
7. Sadoff J, Le Gars M, Shukarev G, et al. Interim results of a phase 1–2a trial of Ad26.COV2.S Covid-19 vaccine. 2021;NEJMoa2034201. N Engl J Med. doi:10.1056/NEJMoa2034201
8. Carvalho-Schneider C, Laurent E, Lemaignen A, et al. Follow-up of adults with noncritical COVID-19 two months after symptom onset. Clin Microbiol Infect. 2021;27(2):258-263. doi:10.1016/j.cmi.2020.09.052
9. Huang C, Huang L, Wang Y, et al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021;397(10270):220-232. doi:10.1016/S0140-6736(20)32656-8
10. Behzad S, Aghaghazvini L, Radmard AR, Gholamrezanezhad A. Extrapulmonary manifestations of COVID-19: radiologic and clinical overview. Clin Imaging. 2020;66:35-41. doi:10.1016/j.clinimag.2020.05.013
11. Salehi S, Abedi A, Balakrishnan S, Gholamrezanezhad A. Coronavirus disease 2019 (COVID-19): a systematic review of imaging findings in 919 Patients. AJR Am J Roentgenol. 2020;215(1):87-93. doi:10.2214/AJR.20.23034
12. Daher A, Balfanz P, Cornelissen C, et al. Follow up of patients with severe coronavirus disease 2019 (COVID-19): pulmonary and extrapulmonary disease sequelae. Respir Med. 2020;174:106197. doi:10.1016/j.rmed.2020.106197
13. Puüntmann VO, Carerj ML, Wieters I, et al. Outcomes of cardiovascular magnetic resonance imaging in patients recently recovered from coronavirus disease 2019 (COVID-19). JAMA Cardiol. 2020;5(11):1265-1273. doi:10.1001/jamacardio.2020.3557
14. Nersesjan V, Amiri M, Lebech A-M, et al. Central and peripheral nervous system complications of COVID-19: a prospective tertiary center cohort with 3-month follow-up. J Neurol. Published online January 13, 202. doi:10.1007/s00415-020-10380-x
15. Lambrecq V, Hanin A, Munoz-Musat E, et al. Association of clinical, biological, and brain magnetic resonance imaging findings with electroencephalographic findings for patients with COVID-19. JAMA Netw Open. 2021;4(3):e211489. doi:10.1001/jamanetworkopen.2021.1489
16. Moriguchi T, Harii N, Goto J, et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int J Infect Dis 2020;94:55-58. doi:10.1016/j.ijid.2020.03.062
17. Domingues RB, Mendes-Correa MC, de Moura Leite FBV, et al. First case of SARS-COV-2 sequencing in cerebrospinal fluid of a patient with suspected demyelinating disease. J Neurol. 2020;267(11):3154-3156. doi:10.1007/s00415-020-09996-w
18. Boldrini M, Canoll PD, Klein RS. How COVID-19 affects the brain. JAMA Psychiatry. Published online March 26, 2021. doi:10.1001/jamapsychiatry.2021.0500
19. Forte G, Favieri F, Tambelli R, Casagrande M. COVID-19 pandemic in the Italian population: validation of a post-traumatic stress disorder questionnaire and prevalence of PTSD symptomatology. Int J Environ Res Public Health. 2020;17(11):4151. doi:10.3390/ijerph17114151
20. Janiri D, Carfì A, Kotzalidis GD, et al. Posttraumatic stress disorder in patients after severe COVID-19 infection. JAMA Psychiatry. 2021;78(5):567-569. doi:10.1001/jamapsychiatry.2021.0109
21. Tucker P, Czapla CS. Post-COVID stress disorder: another emerging consequence of the global pandemic. Psychiatric Times. 2021;38(1).”
Long COVID symptoms likely caused by Epstein-Barr virus reactivation
by World Organization 6/23/21
The number of subjects reporting each of 13 clinical manifestations of long COVID. Credit: Jeffrey E. Gold, Ramazan A. Okyay, Warren E. Licht, and David J. Hurley
Epstein-Barr virus (EBV) reactivation resulting from the inflammatory response to coronavirus infection may be the cause of previously unexplained long COVID symptoms—such as fatigue, brain fog, and rashes—that occur in approximately 30% of patients after recovery from initial COVID-19 infection. The first evidence linking EBV reactivation to long COVID, as well as an analysis of long COVID prevalence, is outlined in a new long COVID study published in the journal Pathogens.
“We ran EBV antibody tests on recovered COVID-19 patients, comparing EBV reactivation rates of those with long COVID symptoms to those without long COVID symptoms,” said lead study author Jeffrey E. Gold of World Organization. “The majority of those with long COVID symptoms were positive for EBV reactivation, yet only 10% of controls indicated reactivation.”
The researchers began by surveying 185 randomly selected patients recovered from COVID-19 and found that 30.3% had long term symptoms consistent with long COVID after initial recovery from SARS-CoV-2 infection. This included several patients with initially asymptomatic COVID-19 cases who later went on to develop long COVID symptoms.
The researchers then found, in a subset of 68 COVID-19 patients randomly selected from those surveyed, that 66.7% of long COVID subjects versus 10% of controls were positive for EBV reactivation based on positive EBV early antigen-diffuse (EA-D) IgG or EBV viral capsid antigen (VCA) IgM titers. The difference was significant (p < 0.001, Fisher’s exact test).
“We found similar rates of EBV reactivation in those who had long COVID symptoms for months, as in those with long COVID symptoms that began just weeks after testing positive for COVID-19,” said coauthor David J. Hurley, Ph.D., a professor and molecular microbiologist at the University of Georgia. “This indicated to us that EBV reactivation likely occurs simultaneously or soon after COVID-19 infection.”
The relationship between SARS-CoV-2 and EBV reactivation described in this study opens up new possibilities for long COVID diagnosis and treatment. The researchers indicated that it may be prudent to test patients newly positive for COVID-19 for evidence of EBV reactivation indicated by positive EBV EA-D IgG, EBV VCA IgM, or serum EBV DNA tests. If patients show signs of EBV reactivation, they can be treated early to reduce the intensity and duration of EBV replication, which may help inhibit the development of long COVID.
“As evidence mounts supporting a role for EBV reactivation in the clinical manifestation of acute COVID-19, this study further implicates EBV in the development of long COVID,” said Lawrence S. Young, Ph.D., a virologist at the University of Warwick, and Editor-in-Chief of Pathogens. “If a direct role for EBV reactivation in long COVID is supported by further studies, this would provide opportunities to improve the rational diagnosis of this condition and to consider the therapeutic value of anti-herpesvirus agents such as ganciclovir.”
More information: Jeffrey E. Gold et al, Investigation of Long COVID Prevalence and Its Relationship to Epstein-Barr Virus Reactivation, Pathogens (2021). DOI: 10.3390/pathogens10060763
Teen girls suicide attempts up dramatically in pandemic: study
June 12, 2021
The US Centers for Disease Control and Prevention (CDC) said Friday that emergency department visits for suspected suicide attempts by teenage girls rose significantly last year compared to 2019, highlighting the mental health impact of the pandemic. “By early May 2020, ED visit counts for suspected suicide attempts began increasing among adolescents aged 12-17 years, especially among girls,” the health agency said in a report.
Between July 26 to August 22, 2020, the average number of visits in girls of this age was 26 percent higher than the same period in 2019. During February 21 to March 20, 2021, it had shot up an alarming 51 percent compared to the same period in 2020. For boys of the same age, the rise in emergency room visits was four percent.
Past research prior to the pandemic has also found self-reported suicide attempts are consistently higher among adolescent females than among males. But “the findings from this study suggest more severe distress among young females than has been identified in previous reports during the pandemic, reinforcing the need for increased attention to, and prevention for, this population,” the authors wrote.
The study was not designed to tease out the risk factors leading to increased suicide attempts. But it said: “Young persons might represent a group at high risk because they might have been particularly affected by mitigation measures, such as physical distancing (including a lack of connectedness to schools, teachers, and peers).”
It also noted barriers to mental health treatment, increases in substance use, and anxiety about family health and economic problems, as possible risk factors. According to the data tracking site Burbio, two percent of US school students are attending classes completely virtually, 70 percent have gone back to in-person learning, and 28 percent are learning via a hybrid of both.
The findings were not disaggregated by factors such as sex, race, income, sexual orientation and gender identity due to limitations in national data.The Trevor Project, a US suicide prevention organization for LGBTQ youth, said in January LGBTQ youth face many of the same challenges as their peers but have reported additional stress at being confined with unsupportive parents and losing access to identity-affirming spaces.
Oral COVID treatment yields promising trial data:
3/7/21 The drug molnupiravir caused a significant drop in patients’ viral load after five days of treatment, Merck said at a meeting with infectious disease experts and Wendy Painter, chief Medical Officer of the US developing firm, It causes a 50% reduction in CoV2-19 hospitalizations and death. Ridgeback Biotherapeutics
The Phase 2a (of 3) test(s) was carried out among 202 non-hospitalized people with symptoms of COVID-1; there was more than a 50 percent reduction in risk of death or respiratory trouble in patients hospitalized with moderate to severe COVID-19, the company said in January.Bottom of Form
Corona Virus incidence/severity 10/12/20: “Hospitalizations, a measure of disease severity, have been steadily declining since March, with a small bump in mid-summer. They’ve gone from a high of over 3,000 hospitalizations per week last Spring, to less than 700 per week now, according to the CDC.
In the Spring only the sickest patients were being admitted to the hospital at that time. There are no such concerns now and those hospitalized presently are not as sick, receiving hospital care much earlier in their illneess.
These case surges, in actuality, are now only positive tests. New York City is shutting down nine neighborhoods based on a positive test rate of over 3 percent for seven straight days. Yet the country as a whole has a higher test positive rate of 4.9 percent currently.” B Joondeph, MD
NEJM 10/2020: “…It is unclear whether RT-PCR is an accurate measure of viral neutralization, since viral RNA may persist for some time even in the absence of replication-competent virus….”
On-the-spot coronavirus test within spitting distance
by University of Technology, Sydney 10/21/20
A COVID-19 test that will provide results within minutes. In an Australian first, UTS scientists have used novel optical technology to design a highly sensitive saliva test for the SARS-CoV-2 virus antigens, or viral protein fragments. The test can deliver a positive result in under 15 minutes.
The rapid antigen test collects saliva in a cartridge placed in an existing hand-held device, first developed by Perth company Alcolizer for illicit drug testing. Customized iStrip technology measures the viral load in the saliva sample, even at very low levels, and displays the result on the instrument’s small screen. The device has GPS location technology and integration to cloud reporting tools to assist with contact tracing.
The test bypasses the time-consuming molecular amplification currently in use. With the quick turnaround of results and a cost of less than $25 per test, it would allow testing rates to increase. The iStrip technology is based on the pioneering work of UTS Professor Dayong Jin in using nanophotonic probes for disease diagnostics. This iStrip is sensitive enough to detect the presence of as little as a trillionth of a gram of SARS-CoV-2 viral protein.
The gold standard PCR (polymerase chain reaction) test, where samples are analyzed in a laboratory over several hours.
Professor Jin said his team’s goal is detection of the presence of SARS-CoV-2 viral protein when a person has yet to show symptoms but is highly infectious. “A person with COVID-19 may be contagious 72 hours before starting to show symptoms. “They are not sensitive enough to effectively screen people who are showing no signs of illness. They also produce a number of false negative results”. “Short of a vaccine, our best hope for containing community transmission and returning to some sort of normal life lies in a fast, highly sensitive and accurate testing regime. We believe our technology will help to realize that ambition.”
Icatibant Treatment Improves Oxygenation in COVID-19 Patients
Zahra Masoud 9/17/20
Icatibant, a bradykinin-2 receptor antagonist in a study recently published in JAMA Network Open Infectious Diseases: a small and potent study.
SARS-CoV-2 enters cells via angiotensin-converting enzyme 2 (ACE2), which is involved in degrading kinin des-Arg9-bradykinin. Kinin des-Arg9-bradykinin is a potent vasoactive peptide that can cause vascular leakage. The loss of ACE2 leads to plasma leakage and further activation of the plasma kallikrein-kinin system, resulting in more bradykinin formation and more stimulation of bradykinin-2 receptors, which would ultimately contribute to pulmonary angioedema.
3 doses of 30 mg icatibant via subcutaneous injection at 6-hour intervals. The study included patients with polymerase chain reaction assay–confirmed SARS-CoV-2, an oxygen saturation <90% without supplemental oxygen, a computed tomography severity score ≥7, and a requirement of ≥3 L/min of supplemental oxygen. The primary outcome variable was a change in oxygen need and oxygenation expressed as absolute number of liters per hour.
Icatibant was well tolerated and improved oxygenation. In all patients who received icatibant, there was a marked decrease in the need for oxygen supplementation. After 3 injections of icatibant, 4 (44%) patients were no longer dependent on oxygen supplementation within 10-35 hours. In 8 (89%) patients treated with icatibant, a reduction of ≥3 L/min in oxygen was observed after 24 hours. Of the 18 matched controls, 3 (17%) showed a spontaneous reduction of ≥3 L/min of oxygen after 24 hours. However, there was a resurgence in the need for oxygen in 3 patients treated with icatibant, which may be due to the 2-hour half-life of icatibant. Icatibant treatment was well tolerated in all 10 patients who received the drug, and no serious adverse events were reported. LANDELUMAB (Takhzyro) is a longer acting bradykinin-2 receptor antagonist than icatibant.
” … targeting the kallikrein-kinin system in patients with COVID-19, especially in the early stages of disease when patients are hypoxic and are admitted to the hospital, might be beneficial.”
Reference
van de Veerdonk FL, Kouijzer IJE, de Nooijer AH, et al. Outcomes associated with use of a kinin B2 receptor antagonist among patients with COVID-19. JAMA Network Open. 2020;3(8):e2017708. doi: 10.1001/jamanetworkopen.2020.17708
“Immune Boosting Role of Vitamins” Maturitas 8/10/20 H Shakoor. “…patients with severe COVID-19 symptoms and pneumonia, admitted to intensive care units, have been shown to have high levels of circulating pro-inflammatory cytokines such as IL-2, IL-7, G-CSF, and TNFα [11,12]…”
This 7/17/20 Journal of Experimental Medicine “Rationale for CXCR2 antagonists for the treatment of COVID-19”
by L F Koening discusses using “inhibitors of chemokine/chemokine receptor pathways to block excessive infiltration of neutrophils to interrupt the self-reinforcing hyperinflammation in severe cases of COVID-19 infection. … There is strong evidence to investigate the usage of CXCR2 antagonists in the treatment of severe COVID-19…. overreactive immune system with infiltration of inflammatory monocytes and neutrophils to the site of infection alongside an exaggerated release of proinflammatory cytokines is an important driver of severe lung damage in COVID-19 (Vabret et al., 2020).”
https://www.practiceupdate.com/c/103914/1/24/?elsca1=emc_enews_daily-digest&elsca2=email&elsca3=practiceupdate_diab&elsca4=diabetes&elsca5=newsletter&rid=MjA2NjE0OTY0Mzg2S0&lid=10332481
New insights into the cellular response to SARS-CoV-2 infection
Med Xpress 7/24/20 From the Karolinska Institute: “…hijacking the mTOR pathway can render the virus highly pathogenic, as was observed for the highly pathogenic 1918 influenza virus and the Middle East respiratory syndrome coronavirus (MERS-CoV). The mTOR pathway also plays a central role in the overall functioning of the cells and is considered a central regulator of lifespan and aging by changing the host metabolism….”
Human invasion by the corona virus is favored by its low “CpG” content. CpG is recognized by the human immune system as a foreign invader, thereby activating the ZAP neutralizing protein. Low CpG levels will escape ZAP immune attack. Thus, the virus is able to invade the human body via its ACE2 protein which is in highest concentration in the intestinal tract. But first it must join with the cell’s heparan sulfate. “Heparan Sulfate Consumption as a Potential Mechanism of Intra-Cardiac Thrombosis in SARS-CoV-2 Infection”
Heart & Lung: The Journal of Acute and Critical Care
• The interaction between the SARS-CoV-2 virus and heparan sulfate is hypothesized to drive the hypercoagulable state. There is brief a theoretical explanation for the effect of the interaction between SARS-CoV-2, the ACE2 receptor, and its co-receptor heparan sulfate.
• Heparan sulfate mediates antithrombin’s anti-inflammatory activity, and its consumption potentially drives endothelial injury and perhaps intra-cardiac thrombus formation.
More information: Molecular Biology And Evolution (2020). DOI: 10.1093/molbev/msaa094 Molecular Biology and Evolution The corona virus CoV2-19 entry into human cells is aided by an enzyme called TMPRSS2.
“ENTRANCE of the virus is gained to the human body via a protrusion on the virus’s outside called a ‘SPIKE’ by using the receptor binding domain (RBD)—which is responsible for this binding action attaches to an enzyme called ACE2 in the human body. ACE is expressed in 2 isotypes: a short and long form, with the convertase activity of the former being substantially faster than the latter, resulting in higher pathogenicity of the short form. Interestingly that is an enzyme that is blocked by certain medications used in the treatment of high blood pressure; it has been reported that patients who are on related blockers called “ARBs”, such as losartan and olmesartan, are resistant to the CoV2-19 virus infection by replenishing the low levels of ACE2 in CoV2-19 infection. But there is also debate about the safety of the use of ACE inhibitors and ARBs as being either protective versus harmful for CoV2-19 infection.”
Medical X-Press 8/19/20: “the ACE2 receptor not only provides a gateway for the coronavirus, but it also keeps vasodilatory kinins under control. The infection causes most of the ACE2 receptors of the lung cells to dysfunction. Without ACE2, these kinins have free rein and—by binding to bradykinin receptors—can make the blood vessels leaky (contributing the lung findings). Internist Frank van de Veerdonk, hospital pharmacist Roger Brüggemann and colleagues hypothesized….treatment for those with fluid in their lungs, were three doses of icatibant by subcutaneous injection at six-hour intervals. The patient in the ICU recovered sufficiently within 24 hours to be moved to the ward and was discharged after 7 days. Eight of the remaining nine patients needed less oxygen supplementation within 24 hours and the ninth after 38 hours….Brüggemann: “The drug lanadelumab remains active for much longer than icatibant, so you will probably only need to administer it only once or twice”.”
covid19-202000328-EN-1.pdf
8/28/20
MD5: d70be51fd2d0eda48d2781a60a118d6c
CiteDownload (2.47 MB)Export as PDFShareEmbed
CoV2-19 Attacks the 1-Beta Chain of Hemoglobin and Captures the Porphyrin to Inhibit Human Heme Metabolism: slightly edited
Posted 3/3/20 by Liu Wenzhong: “In this study, conserved domain analysis, homology modeling, and molecular docking were used to compare the biological roles of certain proteins of this virus. Its ORF8 and surface glycoprotein bind to the porphyrin. At the same time, orf1ab, ORF10, and ORF3a proteins coordinate the attack of the heme on the 1-beta chain of hemoglobin to dissociate the iron to form the porphyrin. This attack will cause less and less hemoglobin availability. The lung cells have extremely intense poisoning and inflammation likely due in part to the inability to exchange carbon dioxide and oxygen. This results in the ground-glass-like lung images. These traits also interfere with the normal heme anabolic pathway. Chloroquine could prevent orf1ab, ORF3a, and ORF10 from attacking heme to form the porphyrin, and inhibit the binding of ORF8 and surface glycoproteins to porphyrins to a certain extent, effectively relieving the symptoms of respiratory distress. FAVIPIRAMIR could inhibit the envelope protein and ORF7, a protein that binds to porphyrin, prevent the virus from entering host cells, and catching free porphyrins.”
INTERFERON is a virus-induced virus-attacking cytokine (a cytokine is made in many locations throughout the body, but is not made in a single area like thyroid hormone which is just made in the thyroid gland). Interferon turns on the ACE2 gene allowing more sites for CoV2-19 entry into the human body. SARS-CoV-2 is capable of blocking interferon production in the cells it infects, and it appears to be much more effective in doing so than its cousin SARS-CoV. But some patients also appear to be less capable of mounting an interferon response even in uninfected immune cells, notes Miriam Merad, who directs the Precision Medicine Institute at Mount Sinai. Either way, without a solid interferon response, the virus will persist, causing damage that activates inflammatory pathways. “The higher the damage is, the more the immune system is trying to get rid of the damage,” says Merad, “so it gets activated and at some point … it goes completely crazy.” This over activation is clearly evident in the form of high concentrations of pro-inflammatory cytokines in patients’ blood—the “cytokine storm” that COVID-19 has become known for. In a recent analysis of nearly 1,500 COVID-19 patients, Merad and her colleagues found that concentrations of IL-6, IL-8 and TNF-α in their serum upon admission correlated strongly with disease severity and death.
Hyperglycemia and COVID-19: Why Management of Glucose Levels Is Essential:
Rachael Beairsto 5/19/20 in Endocrinology Advisor: “Normalization of HYPERGLYCEMIA may be one of the most important first steps in caring for patients with CoV2-19. Hyperglycemia is linked to significantly worse outcomes. 1122 patients with COVID-19 treated in 88 hospitals across the United States, the presence of diabetes or uncontrolled hyperglycemia was linked to a longer length of hospital stay and higher mortality risk (28.8% vs 6.2% in the comparison group without hyperglycemia).
9/22/20
A New Score to Identify High-Risk, Severe COVID-19 Patients
Bradley van Paridon The BAS²IC score can predict the development of early complications by relying on easily accessible, simple, and inexpensive clinical and laboratory parameters.
Results of a new score to predict the risk of rapid progression to severe disease in hospitalized patients with COVID-19, based on easily accessible data such as age, sex, BMI, dyspnea, and inflammatory parameters, were published in Open Forum Infectious Diseases.
In a derivation cohort of 1045 patients, researchers performed a Bayesian logistic regression to identify risk factors for severe COVID-19. Investigators identified advanced age (β coefficient = .4), male sex (β coefficient = .735), overweight (β coefficient = .490), obesity (β coefficient = .776), dyspnea (β coefficient = .913), C-reactive protein level ≥ 100 and < 200 mg/L at admission (β coefficient = .489), C-reactive protein level ≥ 200 mg/L (β coefficient = 1.397), neutrophil count ≥8000/μL (β coefficient = .747) and lymphocyte count <1000/μL (β coefficient = .364) as factors associated severe disease.Kaeuffer C, Ruch Y, Fabacher T, et al. The BAS²IC score: a useful tool to identify patients at high risk of early progression to severe COVID-19 [published online September 1, 2020]. Open Forum Infect Dis. doi: 10.1093/ofid/ofaa405
Recently it has been said that the “70% death rate has changed to an 80% survival rate”: this is likely due to the use of various already available medications. 80% of those deaths occur in peole over the age of 65.
Metabolomics Profiling of Critically Ill Coronavirus Disease 2019 Patients: Identification of Diagnostic and Prognostic Biomarkers
Fraser, Douglas D. MD, PhD1–4 et al. On behalf of the Lawson COVID19 Study Team
Critical Care Explorations: October 21, 2020 – Volume 2 – Issue 10 – p e0272
doi: 10.1097/CCE.0000000000000272
“…Mortality rate for coronavirus disease 2019 positive ICU patients was 40%. Feature selection identified the top-performing metabolites for identifying coronavirus disease 2019 positive patients from healthy control subjects and was dominated by increased kynurenine and decreased arginine, sarcosine, and lysophosphatidylcholine. Arginine/kynurenine ratio alone provided 100% classification accuracy between coronavirus disease 2019 positive patients and healthy control subjects (p = 0.0002). When comparing the metabolomes between coronavirus disease 2019 positive and coronavirus
disease 2019 negative patients, kynurenine was the dominant metabolite and the arginine/kynurenine ratio provided 98% classification accuracy (p = 0.005). Feature selection identified creatinine as the top metabolite for predicting coronavirus disease 2019-associated mortality on both ICU days 1 and 3, and both creatinine and creatinine/arginine ratio accurately predicted coronavirus disease 2019-associated death with 100% accuracy (p = 0.01)….”
Inhaled Budesonide in the Treatment of Early COVID-19
The Lancet Respiratory Medicine 4/24/21
• In the phase II randomized controlled STOIC trial, the authors aimed to determine whether inhaled glucocorticoids would be an effective treatment for early acute respiratory distress syndrome (ARDS). They evaluated outcomes in patients diagnosed with mild COVID-19 who were randomized within 7 days of symptom onset to receive inhaled budesonide 800 μg versus usual care. Patients in the budesonide group had a shorter time to recovery and were less likely to need urgent medical care.
• This is a very important study that highlights an easily accessible treatment that appears to be effect against early mild COVID-19 disease.
Multiple early reports of patients admitted to hospital with COVID-19 showed that patients with chronic respiratory disease were significantly under-represented in these cohorts. We hypothesised that the widespread use of inhaled glucocorticoids among these patients was responsible for this finding, and tested if inhaled glucocorticoids would be an effective treatment for early COVID-19.
We performed an open-label, parallel-group, phase 2, randomised controlled trial (Steroids in COVID-19; STOIC) of inhaled budesonide, compared with usual care, in adults within 7 days of the onset of mild COVID-19 symptoms. The trial was done in the community in Oxfordshire, UK. Participants were randomly assigned to inhaled budsonide or usual care stratified for age (≤40 years or >40 years), sex (male or female), and number of comorbidities (≤1 and ≥2). Randomisation was done using random sequence generation in block randomisation in a 1:1 ratio. Budesonide dry powder was delivered using a turbohaler at a dose of 800 μg per actuation. Participants were asked to take two inhalations twice a day until symptom resolution. The primary endpoint was COVID-19-related urgent care visit, including emergency department assessment or hospitalization, analyzed for both the per-protocol and intention-to-treat (ITT) populations. The secondary outcomes were self-reported clinical recovery (symptom resolution), viral symptoms measured using the Common Cold Questionnare (CCQ) and the InFLUenza Patient Reported Outcome Questionnaire (FLUPro), body temperature, blood oxygen saturation, and SARS-CoV-2 viral load. The trial was stopped early after independent statistical review concluded that study outcome would not change with further participant enrollment. This trial is registered with ClinicalTrials.gov, NCT04416399.
FINDINGS
From July 16 to Dec 9, 2020, 167 participants were recruited and assessed for eligibility. 21 did not meet eligibility criteria and were excluded. 146 participants were randomly assigned-73 to usual care and 73 to budesonide. For the per-protocol population (n=139), the primary outcome occurred in ten (14%) of 70 participants in the budesonide group and one (1%) of 69 participant in the usual care group (difference in proportions 0·131, 95% CI 0·043 to 0·218; p=0·004). For the ITT population, the primary outcome occurred in 11 (15%) participants in the usual care group and two (3%) participants in the budesonide group (difference in proportions 0·123, 95% CI 0·033 to 0·213; p=0·009). The number needed to treat with inhaled budesonide to reduce COVID-19 deterioration was eight. Clinical recovery was 1 day shorter in the budesonide group compared with the usual care group (median 7 days [95% CI 6 to 9] in the budesonide group vs 8 days [7 to 11] in the usual care group; log-rank test p=0·007). The mean proportion of days with a fever in the first 14 days was lower in the budesonide group (2%, SD 6) than the usual care group (8%, SD 18; Wilcoxon test p=0·051) and the proportion of participants with at least 1 day of fever was lower in the budesonide group when compared with the usual care group. As-needed antipyretic medication was required for fewer proportion of days in the budesonide group compared with the usual care group (27% [IQR 0-50] vs 50% [15-71]; p=0·025) Fewer participants randomly assigned to budesonide had persistent symptoms at days 14 and 28 compared with participants receiving usual care (difference in proportions 0·204, 95% CI 0·075 to 0·334; p=0·003). The mean total score change in the CCQ and FLUPro over 14 days was significantly better in the budesonide group compared with the usual care group (CCQ mean difference -0·12, 95% CI -0·21 to -0·02 [p=0·016]; FLUPro mean difference -0·10, 95% CI -0·21 to -0·00 [p=0·044]). Blood oxygen saturations and SARS-CoV-2 load, measured by cycle threshold, were not different between the groups. Budesonide was safe, with only five (7%) participants reporting self-limiting adverse events.
Early administration of inhaled budesonide reduced the likelihood of needing urgent medical care and reduced time to recovery after early COVID-19.
Common diabetes drug shows promise as treatment for COVID-19 lung inflammation
by Scott Lafee 6/9/21
Metformin is a widely prescribed blood sugar-lowering drug. Metformin works by lowering glucose production in the liver, reducing blood sugar levels that, in turn, improve the body’s response to insulin. But scientists have also noted that metformin possesses anti-inflammatory properties.
In a study published online in the journal Cell Metabolism , a multi-institution team led by researchers at University of California San Diego School of Medicine identified the molecular mechanism for the anti-inflammatory activity of metformin and, in mouse studies, found that metformin prevents pulmonary or lung inflammation in animals infected with SARS-CoV-2.
Over the past year, several retrospective clinical studies had reported that metformin use by diabetic and obese patients prior to hospital admission for COVID-19 correlated to reduced severity and mortality. Notably, other drugs used to control blood sugar levels do not appear to produce a similar effect.
“The clinical studies were plagued by confounders that made conclusions hard to reach. There was some skepticism in their findings,” said corresponding study author Michael Karin, Ph.D., Distinguished Professor of Pharmacology and Pathology and Ben and Wanda Hildyard Chair for Mitochondrial and Metabolic Diseases at UC San Diego School of Medicine.
Karin, with co-senior author Elsa Sanchez-Lopez, Ph.D., an assistant professor at the Department of Orthopedic Surgery, postdoctoral fellow Hongxu Xian, Ph.D., and others, turned their focus to a mouse model of acute respiratory distress syndrome (ARDS), a life-threatening condition in which fluids leak into the lungs, making breathing difficult and restricting oxygen supply to essential organs.
ARDS is triggered by trauma and by bacterial or viral infections. It is a frequent cause of death in patients hospitalized with COVID-19. The researchers found that metformin administered to mice prior to or after exposure to bacterial endotoxin, a surrogate for bacterial pneumonia, resulted in the inhibition of ARDS onset and lessening of its symptoms. Metformin also produced a marked reduction in mortality in endotoxin-challenged mice and inhibited IL-1β production and inflammasome assembly within alveolar macrophages—immune cells found in the lungs.
IL-1β, along with IL-6, are cytokines that cause inflammation as an early immune response. Their amounts are often highly elevated in persons infected by SARS-CoV-2, creating “cytokine storms” in which the body starts attacking its own cells and tissues. They are signs of an acute immune response gone awry.
Production of IL-1β depends on a large protein complex called the inflammasome, whose presence in lung tissue is found to be highly increased in deceased COVID-19 patients, a discovery made by co-authors Moshe Arditi, MD, and Timothy R. Crother, Ph.D., at Cedars-Sinai Medical Center in Los Angeles.
Working with colleagues at The Scripps Research Institute, the UC San Diego researchers confirmed that metformin inhibited inflammasome activation and prevented SARS-CoV-2-induced pulmonary inflammation in mice.
Cell culture studies using macrophages revealed the underlying mechanism by which metformin exerts its anti-inflammatory activity: reduced production of ATP by mitochondria. ATP is the molecule that mitochondria use to store chemical energy for cells. It is essential to all cellular processes, but blunted ATP production in liver cells is responsible for the glucose lowering effect of metformin.
Lower amounts of ATP in macrophages led to inhibition of mitochondrial DNA synthesis, which had been previously identified by Karin’s lab as a critical step in NLRP3 inflammasome activation. Subsequent research found that clearing away damaged mitochondria reduced NLRP3 inflammasome activity and reduced inflammation.
UC San Diego researchers also confirmed that specific interference with mitochondrial DNA synthesis in macrophages caused by removal of the enzyme CMPK2 (cytidine monophosphate kinase 2) inhibited IL-1β (but not IL-6) production and prevented ARDS onset.
“These experiments strongly suggest that improved delivery of metformin or CMPK2 inhibitors into lung macrophages can provide new treatments for severe COVID-19 and other forms of ARDS,” said Sanchez Lopez.
The authors said the findings suggest metformin may have therapeutic potential for treating a variety of neurodegenerative and cardiovascular diseases in which NLRP3 inflammasome activation is a factor. “Inhibition of inflammasome activation may also account for the poorly explained anti-aging effect of metformin,” said Karin.
More information: Elsa Sanchez-Lopez et al, Choline Uptake and Metabolism Modulate Macrophage IL-1β and IL-18 Production, Cell Metabolism (2019). DOI: 10.1016/j.cmet.2019.03.011
“How to Avoid the Casualties of the Corona Virus War: Help to Minimize Covid-19 Infection Illness”
In its public medical public medical education role, The Preventive Medicine Center (PMC) concludes that these corona virus treatments for which there is validation would be helpful for those who need it and/or who await a vaccine. If practitioners used these already available treatment options which can be thoughtfully combined, the corona virus infection might simply be reduced to just a bad cold and rarely anything more. As this information may not be widely known, it is summarized here to help patients engage in a discussion with their health care providers.
OVID-19: Being Able to Keep Patients Well Enough To Avoid The Hospital (Updated)
Updated August 21, 2021
A combination of common medications and treatments can keep some, if not most, corona virus/CoV2-19 patients out of the hospital. This information is collated from the world’s medical literature and has been successful in all 35 of my confirmed CoV2-19 patients.
I have used 5-8 of these options in combination as a 5 day therapy since June, 2020. These readily available treatment options are routinely accessible to medical providers and can vastly reduce the symptoms and seriousness of the corona virus infection to just a bad cold and rarely anything more.
Using several of these treatments, other health care providers and I have successfully avoided hospitalization for, while aiding recovery from, CoV2-19. This information should be widely available and recommended to providers and their patients in order to engage in a discussion about, and/or use of, these treatments.
– Methylprednisolone or Decadron/also known as dexamethasone (these steroids are similar to the natural stress hormone hydrocortisone produced by the adrenal glands)
– Asmanex/Alvesco asthma inhalers or steroid similars plus another asthma inhibitor Singulair/montelukast (to reduce lung inflammation)– Stomach acid and cold/viral reducers like Pepcid/famotidine, Halodine/povidone-iodine Nasal Antiseptic liquid packets
– Colchicine – An anti-inflammatory gout medicine (to calm general inflammation)
– Ivermectin – Parasite treatment (inhibits host proteins)
– Livalo – For cholesterol lowering (statin)
– Persantin/dipyridamole – An anti-clotting pill
– Low dose aspirin – Also inhibits clotting
– Tricor/fenofibrate – A triglyceride-blood fat treatment
– Lactoferrin – A milk-derived immune enhancer that acts via inhibiting viral attachment to the cell when heparan (not heparin) used for attachment and also immune protein involvement
– Vitamin D3 5,000 IU, vitamin C, selenium, zinc, quercetin – mostly preventive, but also during infection
– Hydroxychloroquine + azithromycin + zinc – Used as an antibiotic and corona virus “ionophore” (now considered acceptable by the AMA). An option to Ivermectin
– Metformin – A diabetes medicine
– Metoprolol – A beta blocker (“calms” the heart that is under attack)
A recently published study of patients in Tlaxcala, Mexico confirmed this thinking. The aim of that study was to assess the effectiveness of a multidrug-therapy consisting of using ivermectin, azithromycin, montelukast and aspirin = acetylsalicylic acid (called “TNR4” therapy) to prevent hospitalization and death among ambulatory COVID-19 cases. The study was published in the International Journal of Infectious Disease. Nearly 85% of the patients in this study who received the TNR4 recovered within 14 days compared to 59% in the comparison group. The study concluded that the “…. likelihood of recovery within 14 days was 3.4 times greater among the TNR4 group than in the comparison group. Patients treated with TNR4 had a 75% and 81% lower risk of being hospitalized or death, respectively, than the comparison group.” In that article, the TNR4 therapy was shown to improve recovery and death as well as minimize hospitalization of COVID-19 cases.
This treatment does not keep the unvaccinated from getting the virus. Everyone who is not vaccinated, no matter their age, should continue following the common-sense preventive guidelines – double or KN-95 masking, social distancing, disinfecting contact surfaces, and thorough hand washing. Where possible, adding a high efficiency particulate filtering air purifier with UV light such as the Sun-Pure SP 60 can reduce infections by 50%. These are responsible precautions that everyone could do to avoid infection and spreading it to others until vaccinated. They should be a part of our daily lives likely until September, 2021. People will be effectively immune 14 days after their 1st Pfizer or Moderna vaccination which can nearly normalize societal behavior then. At that point, they are very unlikely to become seriously ill if colonized or even if infected. If infected, it would feel like a flu and can still be treated with the above. Adequate “total immunity” will essentially follow 14 days after the second injection or 28 days after the single Johnson and Johnson vaccine.
Current outpatient treatment includes using a statin, 81 mg chewed aspirin, montelukast, steroid inhaler, oral steroid pills, colchicine, ivermectin, Halodine (povidone-iodine) Nasal Spray, and if rapid heart beat is present, a beta blocker (ideally Bystolic): this program was independently developed and is similar to, the TNR4 program of Rene Lima-Morales as reported in the Journal of infectious disease 2021.
Morphological cell profiling of SARS-CoV-2 infection identifies drug repurposing candidates for COVID-19
Carmen Mirabelli, Jesse W. Wotring, Charles J. Zhang, Sean M. McCarty, Reid Fursmidt, Carla D. Pretto, View ORCID ProfileYuanyuan Qiao, Yuping Zhang, Tristan Frum, Namrata S. Kadambi, Anya T. Amin, Teresa R. O’Meara, Jason R. Spence, View ORCID ProfileJessie Huang, Konstantinos D. Alysandratos, Darrell N. Kotton, Samuel K. Handelman, View ORCID ProfileChristiane E. Wobus, Kevin J. Weatherwax, George A. Mashour, Matthew J. O’Meara, Arul M. Chinnaiyan, and Jonathan Z. Sexton
PNAS September 7, 2021 118 (36) e2105815118; https://doi.org/10.1073/pnas.2105815118
1. Contributed by Arul M. Chinnaiyan, July 9, 2021 (sent for review March 25, 2021; reviewed by Matthew L. Albert and Sumit Chanda)
Since its emergence in China in December 2019, SARS-CoV-2 has caused a global pandemic. Repurposing of FDA-approved drugs is a promising strategy for identifying rapidly deployable treatments for COVID-19. Herein, we developed a pipeline for quantitative, high-throughput, image-based screening of SARS-CoV-2 infection in human cells that led to the identification of several FDA-approved drugs and clinical candidates with in vitro antiviral activity.
Abstract
The global spread of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and the associated disease COVID-19, requires therapeutic interventions that can be rapidly identified and translated to clinical care. Traditional drug discovery methods have a >90% failure rate and can take 10 to 15 y from target identification to clinical use. In contrast, drug repurposing can significantly accelerate translation. We developed a quantitative high-throughput screen to identify efficacious agents against SARS-CoV-2. From a library of 1,425 US Food and Drug Administration (FDA)-approved compounds and clinical candidates, we identified 17 hits that inhibited SARS-CoV-2 infection and analyzed their antiviral activity across multiple cell lines, including lymph node carcinoma of the prostate (LNCaP) cells and a physiologically relevant model of alveolar epithelial type 2 cells (iAEC2s). Additionally, we found that inhibitors of the Ras/Raf/MEK/ERK signaling pathway exacerbate SARS-CoV-2 infection in vitro. Notably, we discovered that lactoferrin, a glycoprotein found in secretory fluids including mammalian milk, inhibits SARS-CoV-2 infection in the nanomolar range in all cell models with multiple modes of action, including blockage of virus attachment to cellular heparan sulfate and enhancement of interferon responses. Given its safety profile, lactoferrin is a readily translatable therapeutic option for the management of COVID-19.
• lactoferrin
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is an enveloped, positive-sense, single-stranded RNA betacoronavirus that emerged in Wuhan, China in November 2019 and rapidly developed into a global pandemic. The associated disease, COVID-19, manifests with an array of symptoms, ranging from flu-like illness and gastrointestinal distress (1, 2) to acute respiratory distress syndrome, heart arrhythmias, strokes, and death (3, 4). Recently, the US Food and Drug Administration (FDA) issued emergency approval of remdesivir, a nucleoside inhibitor prodrug developed for Ebola virus treatment (5). Although large-scale vaccination is ongoing worldwide, the need for safe, readily available antivirals is still a clinical priority. An antiviral compound that curbs infection and reduces COVID-19 symptoms would be highly useful to control local outbreaks or for home-based management, to protect immunocompromised patients for whom vaccination strategies are not suitable, and to slow the spread of variants of concern that could escape vaccine neutralization.
Repurposing of FDA-approved drugs is a promising strategy for identifying rapidly deployable treatments for COVID-19. Benefits of repurposing include known safety profiles, robust supply chains, and a short time frame necessary for development (6). Additionally, approved drugs can serve as chemical probes to understand the biology of viral infection and inform on the molecular targets/pathways that influence SARS-CoV-2 infection. To date, several drug repurposing screening efforts have been reported in various cell systems including nonhuman primate VeroE6 (7), Huh7.5 (8), and Caco-2 cells (9) with a significant overlap in reported drugs but with wide-ranging potencies. Here, we developed a pipeline for quantitative high-throughput image-based screening of SARS-CoV-2 infection that led to the identification of several FDA-approved drugs and clinical candidates with previously unreported in vitro antiviral activity. We also determined that inhibitors of the Ras/Raf/MEK/ERK signaling pathway exhibited proviral activity in Huh7 cells. Mechanism of action studies of lactoferrin, the most promising hit, identified that it inhibits viral attachment, enhances antiviral host cell responses, and potentiates the effects of remdesivir.
Results
Fig. 1.
Morphological profiling of SARS-CoV-2–infected Huh7 cells (MOI of 0.2 for 48 h). (A) Clockwise: Representative field with nuclei (cyan), neutral lipids (green), and SARS-CoV-2 N protein (magenta), N-protein image in the same area with “fire” false color LUT showing distinct morphologies of infected cells showing small/round cells with a hollow center, cells with protrusions, and large syncytia, CellMask image showing cell boundaries and syncytia formation. (B) UMAP embedding and phenotypic clustering of 3 million cells exhibiting viral stain show distinct morphologies, including small/bright cells (I), cells with protrusions (III), and syncytia (V). (C) Comparison of normalized cellular features in infected (brown) and uninfected (blue) cells showing differences in cytoplasmic organization, lipid content/distribution, and nuclear texture. All distributions were compared with the Mann–Whitney test and are statistically significant with P < 0.0001.
8/19/221: Proceedings of the National Academy of Sciences: Identification of FDA-Approved Drugs with Antiviral Activity Against SARS-CoV-2.
To identify compounds with antiviral activity against SARS-CoV-2, we tested a library of 1,425 FDA-approved compounds and rationally included clinical candidates (SI Appendix) in Huh7 cells in quantitative high-throughput screening (qHTS) at five concentrations (50, 250, 500, 1,000, and 2,000 nM). Compounds were selected for inclusion if they were FDA-approved and readily commercially available, and chemical probes previously reported to have any general antiviral activity. Compounds were assessed for their antiviral activity (shown schematically in Fig. 2A) using a CellProfiler (10) image analysis pipeline to 1) identify infected objects in the N protein image (from a single cell to large syncytia), 2) measure their morphologic features, and then 3) tabulate how many nuclei reside within the infected objects to calculate the total percentage of infected cells per well. To increase the likelihood of identifying true actives and decrease the false-negative rate of the assay, a liberal selection criterion was employed to choose drugs for follow-up studies (see Methods and Fig. 2A). One hundred thirty-two drugs were selected from qHTS screening or by known activity against SARS, MERS, or SARS-CoV-2 and carried forward for triplicate dose–response confirmation. Ultimately, 17 dose-responsive compounds were confirmed with IC50 values of less than 1 µM (Fig. 2B and Table 1). The remaining compounds either lacked efficacy, exhibited cytotoxicity (e.g., digoxin), or were efficacious only at concentrations above 1 µM (e.g., hydroxychloroquine, chloroquine) and were thus not prioritized for follow-up. Collectively, the 17 identified hits could be stratified by compound class as ion channel modulators (amiodarone, verapamil, clofazimine, and S1RA), nucleosides/DNA binders (remdesivir, entecavir, niclosamide, and thioguanine), kinase inhibitors (bosutinib, fedratinib, and gilterinib), and others (Table 1).
Fig. 2.
Screen to identify drugs with antiviral activity against SARS-CoV-2. (A) Schematic of the anti–SARS-CoV-2 drug repurposing screening. 1) Compounds are administered in qHTS to cells cultured on 384-well plates infected with SARS-CoV-2 and incubated for 48 h. Each plate contains 32 negative (infected) and 32 positive (noninfected) control wells. 2) Cells are fixed, stained, and imaged. Images are analyzed using Cell Profiler to identify nuclei, cell boundaries, neutral lipid content, and viral staining intensity. 3) Dose–response curves are fit to normalized percent infected cells per well. 4) Confirmation of antiviral activity in other cell lines, including a physiologically relevant iPSC-derived human alveolar epithelial cell (iAECs); (B) Dose–response curves of 17 compounds. Graphs represent median SEM of 10-point 1:2 dilution series of selected compounds for n = 3 biological replicates. IC50 values were calculated based on normalization to the control and after fitting in GraphPad Prism
Table 1.
Hit Validation in Caco-2, LNCaP, Vero E6, and an Induced Pluripotent Stem Cell–Derived Model of Alveolar Epithelial Cells, the iAEC2.
To evaluate the translatability of the 17 hits from Huh7 cells in other cell systems, we confirmed activity in LNCaP, Caco-2, and Vero E6 cell lines and in physiologically relevant induced pluripotent stem cell (iPSC)-derived alveolar epithelial type 2 cells (iAEC2s) (16). Antiviral activities across the cell systems are shown in Table 1. iAEC2s were used as a biomimetic model of the human bronchial epithelium that is involved in COVID-19 pathogenesis (17). iAEC2s are permissive to SARS-CoV-2 infection, exhibiting 10–20% N protein-positive cells at MOI of 0.2 and 50–60% positivity at MOI of 10. Upon infection, we observed long tubular protrusions that costained with viral N protein (Fig. 3A). Additionally, unlike the Huh7 model, the vast majority of infected iAEC2 cells were not present in viral syncytia, suggesting that cell-to-cell spread by cell fusion is limited in this model. Nine out of the 17 hits—amiodarone, lomitapide, ipratropium bromide, gilteritinib, fedratinib, clofazimine, remdesivir, S1RA, and bovine lactoferrin—showed dose-responsive antiviral activity against SARS-CoV-2 in iAECs (Table 1). Remarkably, even at a high MOI of 10, bovine lactoferrin, human lactoferrin, S1RA, and remdesivir retained antiviral activity, reflecting the strong efficacy of these compounds in virus-saturated infection conditions (Fig. 3B). Six compounds (amiodarone, ipatropium bromide, lactoferrin, lomitapide, remdesivir, and Z-FA-FMK) maintained efficacy across all tested cell systems (Table 1), suggesting the targets are conserved across multiple cell types.
Fig. 3.
Validation of candidate compounds in iAEC2 cells infected with SARS-CoV-2. (A) SARS-CoV-2–infected iAEC2 cells at MOI of 10, 48 h post infection (p.i.). Nuclei are in cyan, N protein in magenta, and acetylated tubulin in green. Representative image was acquired on a Yokogawa CQ1 high-content imager with a 60× lens and visualized with Fiji ImageJ. Cellular protrusions are indicated with white arrows. (B) Antiviral activity of bovine and human lactoferrin, remdesivir, and S1RA was assessed in iAEC2 cells infected with SARS-CoV-2 at MOI 10. Graphs represent median SEM of 10-point 1:2 dilution series of selected compounds for n = 3 biological replicates.
Characterization of Antiviral Hits and Identification of Compounds that Exacerbate Viral Infection.
To stratify compounds, we performed a time-of-addition study with compound added either 4 h prior to infection (as done previously in the screen) or 1 h p.i. (Fig. 4A). We infected Huh7 with SARS-CoV-2 at MOI of 1 and then quantified infection by detecting the positive-strand viral RNA genome by RNAscope (Fig. 4B). We found that verapamil, entecavir, and niclosamide lost activity under these experimental conditions (Fig. 4C). Amiodarone, clofazimine, S1RA, lomitapide, Z-FA-FMK, the other nucleoside analogs remdesivir and thioguanine, and the kinase inhibitors bosutinib, fedratinib, and gilterinib retained activity regardless of compound addition preinfection or postinfection, suggesting that they inhibit postbinding events. Two compounds, ipratropium bromide and metoclopramide, lost activity when added 1 h p.i., suggesting a role in viral binding inhibition. Although they share the same molecular target (dopamine D2 receptor), metoclopramide and domperidone seem to exert their antiviral activity with different modes of action, either by directly inhibiting binding or indirect effects on the host. Cell counts are shown in SI Appendix, Fig. S3 for drug treatments in Fig. 4 and demonstrate the lack of cytotoxicity.
Fig. 4.
Time-of-addition study with the identified antiviral hits. (A) Experimental scheme where compounds are added 4 h prior (same treatment window as drug screening) or 1 h post infection (p.i.) with SARS-CoV-2 (MOI of 1). Huh7 cells are fixed, permeabilized, and subjected to RNAscope analysis 48 h p.i. (B) Representative image of SARS-CoV-2–infected and noninfected Huh7 cells acquired on the CX5 high-content platform at 10× and analyzed with Fiji ImageJ. Viral RNA is represented in red, and nuclei in cyan. (C) Time of drug-addition for selected antiviral hits (at 10× IC50 dose) organized according to the compound class. Graphs represent the fold decrease of infection over the untreated condition. Infection was calculated on the viral RNA image after image segmentation with Cell Profiler. Graphs represent an average SEM of n = 3 biological replicates. Statistical significance determined using multiple Student’s t test with the Bonferroni–Dunn correction method, with α = 0.05. *P < 0.01.
Our screening also identified compounds that exacerbated infection. All mitogen/extracellular signal-regulated kinase (MEK) inhibitors tested (cobimetinib, trametinib, and binimetinib) resulted in a greater than twofold increase of viral infection in Huh7 (Fig. 5 A and B). To confirm this finding, we performed RNAscope on virus-infected, cobimetinib-treated versus untreated cells 24 and 48 h p.i. (Fig. 5C). The percentage of viral RNA-positive cells was increased at 48 h p.i., but not at 24 h p.i., following treatment, suggesting that these compounds could enhance virus spread. In addition, upon treatment with the three MEK inhibitors, and cobimetinib in particular, we observed an increased syncytia size (Fig. 5A) and more diffuse viral RNA and S protein staining pattern within the infected cells (Fig. 5D). These immunofluorescence staining patterns suggest a difference in viral compartmentalization and spread in MEK inhibitor-treated cells. The increased infection and the diffuse localization of viral RNA was recapitulated when treating the cells with a molecular probe, U0126 (10 μM), that is commonly used as an inhibitor of the Ras–Raf–MEK–ERK pathway (Fig. 5E). Mek inhibitors were also evaluated in Caco-2 cells, where the exacerbation effect was observed for trametinib and binimetinib, albeit at a higher concentration of 10 µM (SI Appendix, Fig. S4). Cell counts are shown in SI Appendix, Fig. S3 for drug treatments in Fig. 5 and indicate the lack of cytotoxicity at the tested doses. Taken together, these data highlight the utility of screening FDA-approved compounds as a way of identifying cellular pathways involved in viral infection.
Fig. 5.
Selective MEK inhibitors exacerbate SARS-CoV-2 infection. (
Lactoferrin Blocks SARS-CoV-2 Replication at the Entry Level.
The most broadly efficacious hit identified was lactoferrin, a protein found in colostrum and airway epithelium (18). To confirm our previous finding of inhibition of N-protein expression by lactoferrin, we infected Huh7 cells with SARS-CoV-2 (MOI of 0.2) under increasing doses of bovine lactoferrin and measured viral RNA using RT-qPCR at 48 h p.i. (Fig. 6A). Lactoferrin exhibited a dose-dependent inhibition of viral replication (Fig. 6A) and retained antiviral activity through a range of MOIs (Fig. 6B). It maintained antiviral activity even when added 1 or 24 h after infection, suggesting multiple modes of antiviral action (Fig. 6B). To rule out iron chelation as a potential mode of action, iron-saturated hololactoferrin and transferrin were tested in Huh7 cells; the former retained activity and the latter was inactive (Fig. 6C). Given the pronounced single-agent efficacy of lactoferrin, we further tested whether combinations with the FDA-approved agent remdesivir could improve the overall antiviral activity. To test for synergy between lactoferrin and remdesivir treatments, we measured the percent infection over 8 × 8 dose combinations, in two technical and two biological replicates along with single agents, DMSO vehicle, and mock infection controls. The negative control (DMSO vehicle) infection rate for the two plates were 21.6% with the 95% credible interval (CI95) of [20.8, 22.5] and 11.6% with CI95 = [11, 12.2], respectively, with no significant loss of cell viability/reduction in cell counts (SI Appendix, Fig. S3C). The maximum efficacy for each treatment and the combination were similar and not significantly different from the no-viral control of 0.004% infected cells remaining. We found the IC50 for remdesivir to be 6.7 nM with CI95 of [6.1, 7.5] and 440 nM with CI95 of [360, 520] for lactoferrin, consistent with previous experiments. To model the synergy, we fit the Bayesian MuSyC model (19) and found the synergistic potency parameter alpha to be 1.5 with CI95 = [0.6, 3.7], indicating that lactoferrin and remdesivir are not significantly synergistic or antagonistic but do exhibit additive efficacy. To visualize, we normalized the infection rate for each plate by the estimated top infection rate and plotted the median for each dose combination, with level sets of the MuSyC fit as shown in Fig. 6E. None of the treatments shown in Fig. 6 exhibited any loss of cell viability/reduction in cell counts (SI Appendix, Fig. S3C).
Fig. 6.
Lactoferrin blocks SARS-CoV-2 at the attachment step. (A) Huh7 cells were infected with SARS-CoV-2 at MOI of 0.2 for 48 h and treated with increasing concentration of lactoferrin (6.25–6,250 nM). Cells were harvested and RNA was extracted. Viral genome copies were calculated by RT-qPCR with an absolute quantification method. (B) Huh7 were infected with SARS-CoV-2 (MOI of 1, 5, and 10; MOI of 0 indicates noninfected cells) and treated with 6,250 nM lactoferrin at 1 and 24 h p.i. Bars indicate the percentage of infected cells in different conditions. Data are an average of eight replicates. Statistical significance determined using multiple Student’s t test with the Bonferroni–Dunn method, with α = 0.05. Except for MOI of 0, all conditions (untreated vs. lactoferrin, 1 h or untreated vs. lactoferrin, 24 h) differ by P < 0.0001. (C) Percentage of SARS-CoV-2–infected Huh7 cells upon treatment with bovine apolactoferrin and hololactoferrin, native human lactoferrin, and transferrin at a concentration of 6,250 nM. (D) Binding assay. Huh7 cells were preincubated on ice with compounds: lactoferrin (1,250 and 6,250 nM) and remdesivir (10 nM) as negative controls for 1 h and then infected with SARS-CoV-2 (MOI = 10) for 1 h on ice. Cells were then washed thoroughly with PBS to remove unbound virus and viral RNA was quantified by RT-qPCR. Huh7 were cultured in NaClO3 for 7 d, which strips heparan sulfate proteoglycans from the cell surface, and were subsequently used as a control for lactoferrin mode of action. (E) Synergy analysis of lactoferrin in combination with remdesivir. Cells were pretreated with combinations or single agents and infected with SARS-CoV-2 (MOI of 10) for 48 h. Data are shown after normalization to viral control (100%) and represent an average of n = 3 biological replicates with n = 2/3 technical replicates each. Unpaired t tests with Welch’s correction were performed in GraphPad Prism to determine significance. *P < 0.0001.
Previous work on lactoferrin in the context of SARS-CoV-1 suggested that lactoferrin blocks viral entry by binding heparan sulfate proteoglycans, which are viral attachment factors. Heparan sulfates are also SARS-CoV-2 attachment factors (20). Therefore, we performed a viral binding assay by incubating SARS-CoV-2 (MOI of 10) in the presence of lactoferrin (1,250 and 6,250 nM) for 1 h on ice followed by quantification of viral RNA by RT-qPCR (Fig. 6D). Remdesivir was included as a negative control as it blocks viral infection at a postbinding step. Both concentrations of lactoferrin, but not remdesivir, blocked SARS-CoV-2 attachment to Huh7 cells (Fig. 6D). As a positive control, Huh7 cells were treated with NaClO3, a protein sulfation inhibitor that depletes cells of heparan sulfate (21). SARS-CoV-2 binding to cells was reduced in NaClO3-treated cells, and additional lactoferrin treatment did not further reduce binding (Fig. 6D). These data suggest that similar to SARS-CoV-1, lactoferrin blocks viral attachment via neutralizing heparan sulfate proteoglycans.
Lactoferrin Elicits Inflammatory Responses that May Suppress Infection by SARS-CoV-2.
Another potential mechanism of action of lactoferrin is through enhancement of inflammatory responses, which can then limit viral replication within host cells (22). We evaluated mRNA levels of IFNβ and the interferon-stimulated genes ISG15, MX1, Viperin, and IFITM3 in lactoferrin-treated infected Huh7 cells (SI Appendix, Fig. S2). SARS-CoV-2 infection did not result in a robust interferon response consistent with previous studies (23). However, we did detect an up-regulation of IFNβ and interferon-stimulated gene transcripts in virus-infected and lactoferrin-treated cells, suggesting that the post-entry antiviral activity of lactoferrin may be interferon-mediated. Since Huh7 cells have defects in innate immune signaling (24), we used the more physiologically relevant model of the iAEC2 cells to detect transcriptomic changes of uninfected cells upon treatment with lactoferrin at the efficacious concentration of 6.25 µM. Our data showed significant changes in the transcription of genes after 48 h of lactoferrin treatment with 1,016 and 1,023 genes significantly up- and down-regulated, respectively (Fig. 7A). Interestingly, one of the top up-regulated genes was the secretoglobin 3A2 (SCGB3A2). It is the only secretoglobin expressed in this alveolar lineage (other secretoglobins being mostly marker of airways cells), and it has been widely reported as an anti-inflammatory protein in the context of allergic reaction to house dust mites (25). Pathway analysis revealed an induction of the anti-inflammatory TNFα pathway via NFκB (Fig. 7B) with specific up-regulation of transcripts for interferon-stimulated genes: TRIM25, ISG20, IFIT2, but also for interferon receptor α and γ (IFNAR1 and IFNARG2, respectively) (Fig. 7C). Notably, an up-regulation of IRF9 and STAT2 was also observed, consistent with the activation of a noncanonical antiviral and immunoregulatory program triggered by the combination of IFNβ and TNFα (26) (Fig. 7C). Altogether, these data suggest that lactoferrin likely exerts both direct and indirect antiviral action by inhibiting entry and by inducing an antiviral program that may suppress SARS-CoV-2 replication by host cell machinery.
Fig. 7.
Lactoferrin indirect antiviral action is mediated by the up-regulation of cellular innate immune response. iAEC2 cells were treated with lactoferrin (6,250 nM) for 48 h, and then RNA was extracted and sequenced. (A) Volcano plot of genes down-regulated (blue) and up-regulated (red) upon treatment with lactoferrin. (B) Analysis of the top up-regulated and down-regulated pathways. (C) Heatmap of selected genes. In red are highlighted genes associated with inflammation and antiviral response.
Discussion
In this study, we developed an experimental workflow based on high-content imaging and morphological profiling that allows for rapid screening of FDA-approved compounds and identified 17 compounds that inhibit SARS-CoV-2 infection in vitro. Of these, seven were previously reported and serve as a benchmark validation of our endpoints and experimental approach, and 10 were hitherto unknown. We evaluated the antiviral activity of the 17 hits identified in Huh7 in three transformed cell lines (VeroE6, Caco-2, and LNcaP) and one nontransformed cell line (iAECs) and observed six compounds (amiodarone, ipratropium bromide, lactoferrin, lomitapide, remdesivir, Z-FA-FMK) exhibiting activity across multiple cell lines.
Since the completion of this screen (June 2020), over 30 studies reporting SARS-CoV-2 antiviral activity of FDA-approved drugs have been published. A meta-analysis of these in vitro screens (including this effort) show consensus around 11 compounds, with small total overlap between studies (27). This observation suggests that drug screening of FDA-approved compounds is highly dependent on the chosen cell line and infection conditions. It is expected that compounds exerting an antiviral effect through direct binding to viral proteins would be more independent of the chosen cell system rather than drugs modulating host cell factors that can vary widely by cell type. For example, we observed conserved activity across cell systems for remdesivir, which directly inhibits the viral polymerase (28); lomitapide, which is proposed to directly inhibit SARS-CoV-2 main protease (Mpro) (29); and lactoferrin, which we have shown directly inhibits viral entry.
As most FDA-approved drugs are optimized against human molecular targets, active compounds can lead to target identification of host factors involved in SARS-CoV-2 infection. ual action theZ-FA-FMK is an irreversible cathepsin L inhibitor that exhibits potent antiviral activity in all of the five cell systems tested herein because cathepsin L has been shown to be an entry factor of SARS-CoV-2 through the late endolysosome (30, 31). Another hit in our Huh7 screen, fedratinib, was approved by the FDA in 2019 for myeloproliferative neoplasms (32) and is an orally bioavailable semiselective JAK1/JAK2 inhibitor. JAK inhibitors have been proposed for COVID-19 treatment to specifically inhibit the Th17-mediated inflammatory response (33, 34) and to block numb-associated kinase responsible for clathrin-mediated viral endocytosis (35). The JAK inhibitor baricitinib (36) in combination with remdesivir was granted emergency use authorization by the FDA in late 2020, while jakotinib (ChiCTR2000030170) and ruxolitinib (ChiCTR2000029580) are currently being evaluated in clinical trials for COVID-19 as potential drapeutics (antiviral and innate immune response inhibitors).
In contrast to the antiviral drug hits, we report the connection between MEK inhibition and exacerbated SARS-CoV-2 infection, likely by increasing cell-to-cell spread as suggested by the formation of larger syncytia and more diffuse localization of viral RNA and S protein within infected cells (Fig. 5). Intriguingly, in the context of other virus infections, including SARS-CoV-1, pharmacological inhibition of the Ras–Raf–MEK–ERK pathway results in restriction of viral infection (37). This underscores the importance of this pathway during viral infections and warrants further examination into the mechanism of action of this signaling cascade during SARS-CoV-2 infection.
This study has generated several clinically testable and readily translatable hypotheses. As an example, we observed potent antiviral activity of ipratropium bromide (Atrovent), a quaternary ammonium salt and muscarinic receptor antagonist that is commonly prescribed for asthma. It is administered via inhalation into the lungs with little systemic absorption. Given its potential mode of action as inhibitor of SARS-CoV-2 attachment, prophylaxis or postexposure treatment with ipratropium bromide may curb infection of the upper respiratory tract and drastically reduce systemic viral spread and development of severe symptoms while achieving beneficial bronchodilation. Similarly, we identified metoclopramide and domperidone, both dopamine D2 receptor antagonists used to treat gastrointestinal symptoms, as SARS-CoV-2 inhibitors. Gastrointestinal symptoms have been increasingly reported in more than half of the patients infected by SARS-CoV-2 (2). Hence, these compounds may ameliorate gastrointestinal (GI) symptoms during COVID-19 infection, and, in addition, the reduced viral load in the GI tract could also reduce fecal-oral transmission of SARS-CoV-2 (38). Consistent with our findings, the approved leprosy drug clofazamine was recently shown to not only have antiviral activity in cells, but block viral infection in a hamster model of disease (39), making it a strong candidate for clinical translation.
Most noteworthy, our screen identified bovine lactoferrin, a safe and widely available dietary supplement, with multimodal efficacy in multiple cell systems, including nontransformed and physiologically relevant iAEC2s. Our study is the only repurposing discovery effort that included several proteins (like lactoferrin), as conventional high-throughput screening is generally limited to small molecules in DMSO. Lactoferrin gene expression was shown to be highly up-regulated in response to SARS-CoV-1 infection (40), and in addition to enhancing natural killer cell and neutrophil activity, lactoferrin blocks SARS-CoV-1 attachment through binding to heparan sulfate proteoglycans (20). Here, we showed that lactoferrin likely has a multimodal mechanism of action against SARS-CoV-2 infection (Figs. 6 and 7). First, it strongly inhibited cellular binding of SARS-CoV-2 to cells via competition with heparan sulfate. Second, it modulated host cell innate immune responses through increased expression of interferon-stimulated genes and TNFα. Through heightening the innate immune response of host cells, orally administered lactoferrin could be effective in resolving the GI symptoms that are present in COVID-19 patients (41) with a mechanism similar to norovirus infection (42). In addition, lactoferrin was previously shown to decrease the production of IL-6 (43), which is one of the key players of the “cytokine storm” produced by SARS-CoV-2 infection (44, 45). Bovine lactoferrin, widely available as 250-mg gelatin capsules for oral administration, is classified by the FDA as “generally recognized as safe” and therefore may represent a promising therapy for preexposure and postexposure prophylaxis. As a therapeutic intervention for COVID-19, lactoferrin might exert direct antiviral and anti-inflammatory efficacy in the airway with alternative routes of administration like intranasal delivery or in a nebulized formulation delivered to the lungs.
Combination therapies are likely to be required for effectively treating SARS-CoV-2 infection, and this approach has already shown promise, i.e., combination therapy with interferon β-1b, lopinavir–ritonavir, and ribavirin showed efficacy against SARS-CoV-2 in a prospective, open-label, randomized, phase 2 trial (46). Here, we show that lactoferrin potentiates the antiviral activity of remdesivir and could be used in combination therapy with these drugs, which are currently being used or studied for the treatment of COVID-19. Due to its wide availability, limited cost, and strong safety profile, lactoferrin could be a rapidly deployable option for both prophylaxis and the management of COVID-19. Although our findings are promising and have led to several clinical studies, further studies are needed to confirm the efficacy of our lead antiviral compounds in animal models and/or clinical studies … C Mirabelli Proceeding fo the National Academy of Sciences PNAS (2021). DOI: 10.1073/pnas.2105815118
Aspirin use for cardiovascular disease may reduce likelihood of COVID-19 infection
by Bar-Ilan University 3/10/21
CC0 Public Domain
The use of aspirin was very popular during the 1918 Spanish Influenza pandemic. Studies showed that aspirin, in addition to its well-known anti-inflammatory effects, could modulate the innate and adaptive immune responses helping the human immune system battle some viral infections. A joint team from Leumit Health Services, Bar-Ilan University, and Barzilai Medical Center conducted an observational epidemiological study, utilizing data from Leumit Health Services, a national health maintenance organization in Israel. Their findings were recently published in The FEBS Journal.
The researchers analyzed data of 10,477 persons who had been tested for COVID-19 during the first COVID-19 wave from February 1, 2020 to June 30, 2020. Aspirin use to avoid the development of cardiovascular diseases in healthy individuals was associated with a 29% lower likelihood of COVID-19 infection, as compared to aspirin non-users. The proportion of patients treated with aspirin was significantly lower among the COVID-19-positive individuals, as compared to the COVID-19-negative ones. And those subjects who had been treated with aspirin were less associated with the likelihood of COVID-19 infection than those who were not. Moreover, the group observed that the conversion time of SARS-CoV-2 PCR test results from positive to negative among aspirin-using COVID-positive patients was significantly shorter, and the disease duration was two-three days shorter, depending upon the patients’ pre-existing conditions.
“This observation of the possible beneficial effect of low doses of aspirin on COVID-19 infection is preliminary but seems very promising,” says Prof. Eli Magen from the Barzilai Medical Center, who led the study.
More information: Eugene Merzon et al, The use of aspirin for primary prevention of cardiovascular disease is associated with a lower likelihood of COVID‐19 infection, The FEBS Journal (2021). DOI: 10.1111/febs.15784
Provided by Bar-Ilan Univer
Myocarditis VAERS Analysis Sparks Social Media Uproar
— Doctors worry misinterpretation will fuel misinformation campaigns
by Kristina Fiore, Director of Enterprise & Investigative Reporting, MedPage Today September 15, 2021
Researchers are getting pushback over a medRxiv preprint that relied heavily on Vaccine Adverse Event Reporting System (VAERS) data to characterize myocarditis risk with the COVID-19 vaccine in adolescents, particularly young boys.
The report by Tracy Høeg, MD, PhD, of the University of California Davis, and colleagues found that rates of “cardiac adverse events” after the second dose were higher than previous CDC estimates, at 162 per million among boys ages 12 to 15 and 94 per million among boys ages 16 to 17. (Rates were much lower for girls, at about 13 per million for each age group.)
The authors also concluded that the risk of hospitalization for cardiac adverse events following vaccination is higher than the risk of being hospitalized with COVID for healthy boys in both age groups.
The findings led to an uproar by physicians on social media, who pointed out that they’re unreliable due to the nature of VAERS and its known limitations — and that the authors are running the risk of serious misinterpretation of their findings by groups with bad intentions.
VAERS is an early warning system that can generate hypotheses that require adjudication of reports; it was never intended to be used as a research dataset, said blogger David Gorski, MD, PhD, of Wayne State University in Detroit.
Gorski also raised concerns about confirmation bias
Høeg responded to questions from MedPage Today via email. (John Mandrola, MD, the cardiologist on the team, recused himself because he writes for one of MedPage Today’s competitors.)
Høeg said the group used the same definition of myocarditis that the CDC did for its previous estimates using VAERS data, and that cardiologist Mandrola was key in vetting reports. She also pointed to the established fact that VAERS often provides an underestimate of the true prevalence of an adverse event. HRS states, Mandrola is very thoughtful.
Several physicians, however, have asked why this group would bother repeating the CDC’s own analysis, especially if they are using the same definition of myocarditis — and working without CDC’s adjudicated data.
“We repeated the project because we were concerned about the symptom search criteria the CDC used being too narrow,” Høeg told MedPage Today. “We found around 40% of our cases simply using expanded symptom search criteria from theirs but requiring the same objective evidence they did of myocarditis (which we called in our paper ‘cardiac adverse event’).”
She said they also wanted to stratify risk by age groups 12-15 and 16-17, which CDC hadn’t done: “As a mom of 10- and 13-year-old boys, this was important information for me to see and I know many other parents feel the same way.”
Nonetheless, outside researchers noted that Høeg and colleagues could have assessed previously adjudicated reports from the CDC, or that they could have assessed data from the CDC’s Vaccine Safety Datalink (VSD) research dataset instead.
“The benefit of using the VAERS first was this is a rare event, which we were just beginning to track in the U.S., and VAERS gave us quick access to the largest number of reported cases,” Høeg said. “We could also easily identify instances of post-vaccination cardiac injury in terms of troponin levels, along with case descriptions (which we have made publicly available) and directly compare our rates with those that the CDC found, demonstrating that there may be a significant amount of missed post-vaccination cardiac damage in this age group, specifically in the youngest group, compared to what the CDC had initially reported.”
Høeg has a PhD in epidemiology and public health from the University of Copenhagen, but her medical specialty is physical medicine & rehabilitation. She is employed by Northern California Orthopaedic Associates. Her bio for that group says she’s a “Voluntary Assistant Professor at UC Davis.”
Mandrola is a cardiac electrophysiologist affiliated with Baptist Health in Louisville, Kentucky.
The other two authors on the paper appear to be medical communications professionals. Allison Krug, MPH, is an epidemiologist and principal of Artemis Biomedical Communications.
Josh Stevenson lists Truth in Data, as his primary affiliation on the paper, and is also listed under the “team” page at Rational Ground, where he is described as a “data visualization expert who focuses on creating easy to understand charts and dashboards with data,” with a background in computer systems engineering.
Rational Ground has been known for challenging mask mandates and lockdowns. For instance, it offers sample letters to summer camps, school boards, and college presidents, describing how the COVID pandemic has “drummed up unnecessary panic.”
“Did the authors’ biases lead to this analysis?” Gorski wrote in his blog. “Who knows? I just know that the result is definitely not good, and the involvement of Josh Stevenson in particular makes this whole study very suspect indeed.”
Gorski emphasized the typical pathway for evaluating a hypothesis generated by VAERS: CDC will follow up to see if the event is actually associated with vaccination, and if correlation is established, it will investigate whether causation is plausible.
“Other databases, active surveillance system databases like the VSD and [Clinical Immunization Safety Assessment], are required to test these hypotheses, because again — VAERS data are, by the design of the database, unsuitable for this purpose,” he noted.
Gorski, like other physicians on social media, acknowledged that early data, including from other countries such as Israel, do seem to indicate an increased risk of myocarditis in younger boys.
“There’s a signal,” he wrote. But, from the evidence we have thus far: “It’s not yet clear how large the risk of myocarditis is in this age group.”
COVID-19 Outcomes Tied to Hospital,
Not Just Race
Patrice Wendling 11/19/20
Researchers studied 7868 patients hospitalized across 88 sites from January 1 to July 22 of this year in the American Heart Association’s COVID-19 Cardiovascular Disease (CVD) Registry, established early in the pandemic to better understand hospital outcomes and CV complications. Over the study period, there were 1447 deaths (18.4%) and 768 of these, or 53%, were among Black and Hispanic patients.
In-hospital deaths occurred in 17.6% of Black, 16% of Hispanic, and 19.3% of Asian patients, compared with 21.1% of non-Hispanic White patients (P < .001).
Contrary to expectations, race and ethnicity were not associated with mortality in logistic regression analyses that adjusted for sociodemographic, clinical, and presentation factors and included a random intercept for hospitals to account for variation within and across hospitals.
The fully adjusted odds ratios (ORs) for mortality were 0.93 (95% CI, 0.76 – 1.14) for Black individuals, 0.90 (95% CI, 0.73 – 1.11) for Hispanic patients, and 1.31 (95% CI, 0.96 – 1.80) for Asian persons, compared with non-Hispanic White patients.
“Our headline is not [that] there’s no racial or ethnic differences in mortality,” Fatima Rodriguez, MD, MPH, assistant professor of medicine (cardiovascular), Stanford University Medical Center, California, told theheart.org | Medscape Cardiology. Rodriguez pointed out that smaller studies have also found no association between race/ethnicity and mortality.
Results were similar for the secondary outcome of major adverse cardiovascular events (MACE), which occurred in 21.4% of patients and was defined as death, myocardial infarction, stroke, new-onset heart failure, or cardiogenic shock. Unadjusted MACE rates were lower in Black (21.4%) and Hispanic (17.7%) patients than in White patients (24.7%, P <.001) but were no longer different after full adjustment.
In the final model, the odds ratios were 0.99 for Black (95% CI, 0.82 – 1.20), 0.88 for Hispanic (95% CI, 0.72 – 1.08), and 1.28 for Asian (95% CI, 0.95 – 1.72) patients, compared with White patients.
Most of the feared cardiovascular complications of COVID-19 did not occur as often as anticipated, Rodriguez noted. “Rates of myocarditis were very low; even rates of blood clots, DVTs/pulmonary embolisms were relatively low, under 5%. So that was surprising.”
At admission, Black and Hispanic patients were substantially younger, at 60 and 57 years, than White and Asian patients, at 69 and 64 years, and had more adverse socioeconomic factors.
Black patients had the highest prevalence of obesity, hypertension, diabetes, prior cerebrovascular disease, and advanced kidney disease, whereas White patients had the highest prevalence of prior coronary artery disease and pulmonary disease. In hypertension, immune cells are pre-activated: CoV2-19 increases this activation.
Hispanic patients did not have more comorbidities than other racial/ethnic groups. Asian patients had the highest cardiorespiratory disease severity at presentation.
With regard to COVID-19 specific therapies, hydroxychloroquine was the most common. Remdesivir was infrequently used, particularly among Black patients despite a greater need for mechanical ventilation. This may be explained by more advanced kidney disease and lower rates of COVID-19 trial participation among Blacks, Rodriguez said.
Table. COVID-19 Specific Therapies
Therapy
Non-Hispanic White
Black
Hispanic
Asian/Pacific Islander
Hydroxychloroquine (%)
40.0
42.2
44.4
46.6
Remdesivir (%)
8.0
6.1
9.5
9.2
Tocilizumab (%)
7.5
7.1
6.6
9.4
Steroids (%)
22.3
22.3
19.0
24.1
Convalescent serum (%)
2.9
3.3
2.3
2.8
The results were presented alongside other registry findings during the 2020 virtual AHA Scientific Sessions and came days after the AHA issued an advisory statement on structural racism.
American Heart Association 2020 Scientific Sessions. Presented November 17, 2020.
Circulation. Published online November 17, 2020. Abstract
35% of excess deaths in pandemic’s early months tied to causes other than COVID-19
Journal of the American Medical Association (2020). DOI: 10.1001/jama.2020.19545
Provided by Virginia Commonwealth University
COVID-19 not the sole cause of excess U.S. deaths in 2020
by University of Pennsylvania 4/13/21
By the year 2017, the United States was already suffering more excess deaths and more life years lost each year than those associated with the COVID-19 pandemic in 2020, according to research from demographers Samuel Preston of the University of Pennsylvania and Yana Vierboom of the Max Planck Institute for Demographic Research.
In 2017, the United States suffered an estimated 401,000 total excess deaths, those beyond the “normal” number of deaths expected to have occurred. The Centers for Disease Control and Prevention reports 376,504 deaths related to COVID-19 in 2020.
“We do not want to diminish the tremendous losses due to COVID-19 in the U.S. and elsewhere,” says Vierboom, a postdoctoral research scientist in Max Planck’s Laboratory of Population Health. “It’s just a helpful tool to put the U.S. mortality disadvantage into perspective.”
The researchers published their findings in the journal Proceedings of the National Academy of Sciences.
“It is not commonly recognized how far we have fallen behind our European counterparts in mortality and survival,” says Preston. “A notable U.S. disadvantage in 2000 has ballooned in the course of the 21st century.”
To make age-specific comparisons of U.S. and European mortality from 2000 to 2017, Preston and Vierboom used data from the Human Mortality Database to create three indexes. For the comparison, they selected the five largest European countries—Germany, England and Wales together, France, Italy, and Spain—whose combined population nears that of the U.S. “We chose these larger countries because small countries may have unique features such as climate, diet, social history, and health care that make them inappropriate models for larger and more diverse populations,” Preston says.
Even more striking was the mortality comparison between the U.S. and those five European countries when using the measure known as “years of life lost.” This accounts for the age at which death occurs, giving more weight to death at a younger age. In 2020, 4.41 million years of life were lost to COVID-19, yet that’s only about one-third of the 13.02 million life years lost to excess mortality in the United States in 2017.
“Our results underscore the routine and persistent daily health hazards that Americans face,” Vierboom says. “Identifying and remediating the factors that contribute to this massive loss of life should be a national priority.”
U.S. COVID deaths may be underestimated by 36%: HRS, MD, comments: “or overestimated by 25%”
More information: Samuel H. Preston et al. Excess mortality in the United States in the 21st century, Proceedings of the National Academy of Sciences (2021). DOI: 10.1073/pnas.2024850118
The psychological toll of the pandemic
University of British Columbia October 12, 20
From facing fears of contracting the virus, coping with extended separation from loved ones, and combatting the everyday emotional and financial repercussions of COVID-19, the pandemic is having an unprecedented impact on our mental health and wellbeing. Dr. Lakshmi Yatham, professor and head of the UBC department of psychiatry on the psychological toll of COVID-19.
COVID-related delays to colorectal cancer screening causing 11.9% rise in death rates, research reveals
by Spink Health 10/12/20
New research presented today at UEG Week Virtual 2020 has shown that delays in colorectal cancer (CRC) screening caused by COVID-19 has resulted in significantly increased death rates for the cancer. Researchers at the University of Bologna
Nasal Povidone-Iodine Solutions Effectively Inactivate SARS-CoV-2 In Vitro
Brandon May October 2, 2020
All 3 nasal povidone-iodine solutions completely inactivated SARS-CoV-2 within 15 seconds of contact.
Nasal povidone-iodine (PVP-I) solutions at concentrations between 0.5% and 2.5% were capable of rapidly inactivating the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in vitro at relatively short contact times, according to study results published in JAMA Otolaryngology–Head & Neck Surgery.
High viral loads of SARS-CoV-2, the virus that causes coronavirus disease 2019 (COVID-19), have been detected in both the nasopharynx and oropharynx of asymptomatic and symptomatic carriers. Physical barriers and personal protective equipment are typically employed to reduce transmission of the virus through aspiration, but some research has supported the use of intranasal PVP-I solutions as an effective agent against SARS-CoV-2.
In this in vitro laboratory study, researchers tested nasal antiseptic solutions composed of aqueous PVP-I as the active ingredient against the fully sequenced USA-WA1/2020 strain of SARS-CoV-2. Diluted concentrations of PVP-I at 0.5%, 1.25%, and 2.5% were studied, and efficacy of these solutions was compared with controls. The positive control consisted of 70% ethanol on test media infected with SARS-CoV-2. The virus-absent test media were added to 2 tubes of the compounds, which served as toxicity and neutralization controls.
Investigators incubated both the test solutions and the virus at a mean room temperature of 22 °C for 15 & 30 seconds. The log reduction value following 15 seconds and 30 seconds of the given treatment comprised the primary outcome.
All 3 PVP-I solutions completely inactivated SARS-CoV-2 within 15 seconds of contact, as represented by a reduction of greater than 3log10 of the 50% cell culture infectious dose (CCID50) of the virus (3.67 log10 CCID50/0.1 mL to ≤0.67 log10 CCID50/0.1 mL). In contrast, the positive ethanol control did not completely inactivate the virus after this same time period. There were no cytotoxic effects observed on cells following contact with the tested nasal antiseptics.
A limitation of the study included the lack of in vivo assessment of the efficacy and safety of the PVP-I solutions against SARS-CoV-2.
Based on their findings, the investigators concluded that “povidone-iodine nasal irrigation may be beneficial for the population at large as an adjunct to mask usage as a means of virus mitigation.”
Frank S, Brown SM, Capriotti JA, Westover JB, Pelletier JS, Tessema B. In vitro efficacy of a povidone-iodine nasal antiseptic for rapid inactivation of SARS-CoV-2. JAMA Otolaryngol Head Neck Surg. Published online September 17, 2020. doi:10.1001/jamaoto.2020.3053
Medical Xpress 7/27/20: “In a proof-of-concept trial involving 31 patients with COVID-19, DIPYRIMADOLE supplementation was associated with significantly decreased concentrations of D-dimers (P<0.05), increased lymphocyte and platelet recovery in the circulation, and markedly improved clinical outcomes for the severely ill patients in comparison to the control patients.
7/31/20: Iloprost May Be Useful in the Treatment of COVID-19 Vasculopathy
7/31/20 Rheumtology Advisor: Iloprost may help reduce lung damage and thrombotic complications observed in some patients with COVID-19.
Prostacyclin receptor agonist iloprost may be a useful adjunctive treatment for coronavirus disease 2019 (COVID-19) vasculopathy, according to a report published in Lancet Rheumatology.1
3 patients with severe COVID-19 and hypoxemia who tested positive for CoV2-19, 2 had digital ischemia; all 3 received supportive oxygen treatment. Based on clinical diagnoses and persistent oxygen requirement, patients received a 5-day intravenous infusion of iloprost (0.5 mg/kg/min).
Following continuous treatment with iloprost, patients showed sustained clinical improvement in digital ischemia and cardiovascular and respiratory parameters, decreasing oxygen requirements, and increasing Pa02:FiO2 ratio. It was noted that none of the patients required mechanical ventilation or had any serious adverse events. Any complications, including mild rebound tachycardia, observed upon iloprost cessation were resolved before patients were discharged from the hospital.”
References
1. Moezinia CJ, Ji-Xu A, Azari A, Horlick S, Denton C, Stratton R. Iloprost for COVID-19-related vasculopathy [published online July 10, 2020]. Lancet Rheumatol. doi:10.1016/S2665-9913(20)30232-0
2. Hughes M, Ong VH, Anderson ME, et al. Consensus best practice pathway of the UK Scleroderma Study Group: digital vasculopathy in systemic sclerosis. Rheumatology. 2015;54(11):2015-2024.
In cell studies, seaweed extract outperforms remdesivir in blocking COVID-19 virus by Mary L. Martialay, Rensselaer Polytechnic Institute (RPI)
Medical Express 7/24/20: An extract from edible seaweeds WITH no cellular toxicity even at the highest concentrations outperformed remdesivir, the current standard antiviral used to combat the disease. Heparin, a common blood thinner, and a heparin variant stripped of its anticoagulant properties, performed on par with remdesivir in inhibiting SARS-CoV-2 infection in mammalian cells. This is an example of a decoy, from the RPI Center for Biotechnology & Interdisciplinary Studies (CBIS). This decoy technique also works in trapping other viruses, including dengue, Zika, and influenza. There is an ESCO dose response: shorthand for the effective concentration of the compound that inhibits 50% of viral infectivity: a lower value signals a more potent compound.
The seaweed extracts RPI-27 yielded an EC50 value of approximately 83 nanomolar, while a similar in vitro test of remdesivir on the same mammalian cells yielded an EC50 of 770 nanomolar. Heparin yielded an EC50 of 2.1 micromolar, or about one-third as active as remdesivir, and a non-anticoagulant analog of heparin yielded an EC50 of 5.0 micromolar, about one-fifth as active as remdesivir.
These substances could be the basis for a nasal spray or as an oral delivery approach to address potential gastrointestinal infection. “Sulfated polysaccharides effectively inhibit SARS-CoV-2 in vitro” was published in Cell Discovery (2020). DOI: 10.1038/s41421-020-00192-8 Paul S. Kwon et al. Sulfated polysaccharides effectively inhibit SARS-CoV-2 in vitro, Cell Discovery
The rapid inflammatory response and increased glycosylation of the angiotensin-converting enzyme 2 (ACE2) receptor increases CoV2-19 disease severity via a higher propensity for cellular intrusion SARS-CoV-2 using the ACE2 receptor. The amount of glycosylated ACE2, not just the amount of ACE2 is associated with viral binding and fusion. Hyperglycemia also produces a cytokine release and favors the nonenzymatic glycosylation of the ACE2 receptor.
The glycosylation of ACE2 induced by hyperglycemia is needed for the linkage of the virus to this cellular receptor. This mechanism, at the very early stage, is reversible using the Updated Yale Insulin Infusion Protocol for managing hyperglycemia during critical illness.
1. Bode B, Garrett V, Messler J, et al. Glycemic characteristics and clinical outcomes of COVID-19 patients hospitalized in the United States [published online April 12, 2020]. J Diabetes Sci Technol. 2020; in press.
2. Ceriello A. Hyperglycemia and the worse prognosis of COVID-19. Why a fast blood glucose control should be mandatory [published online 4/28/20]. Editorial. Diabetes Res Clin Pract. doi:10.1016/j.diabres.2020.108186
Jon Barron on LUNG INVASION: “COVID-19 appears to have a preference for two specific types of lung cells: goblet cells and ciliated cells. Goblet cells produce the mucus that both keeps your lungs moist and also captures particles, bacteria, and even viruses that you might inhale. Ciliated cells, on the other hand, have little hairs on them that move in a wavelike manner pushing the mucus (and anything it captured) up and out of the lungs into the back of the throat, where you can cough it out.” In the HEART, myocardial cellular targets for SARS-CoV-2 = pericytes, cardiomyocytes, fibroblasts, and immune cells such as resident macrophages. “upregulation of 6 proinflammatory genes (tumor necrosis factor, interferon γ, CCL4, and interleukin 6, 8, and 18) in the 16 myocardial samples with the high viral RNA levels” Bonow et al in JAMA Network 9/22/29/20. The S1 subunit of spike proteins expressed at the surface of SARS-CoV-2 is known to bind to angiotensin-converting enzyme 2 (ACE2) on target cells. Once the virus is bound to ACE2, the TMPRSS2 protease facilitates viral entry into the host cell. These mechanisms of SARS-CoV-2 highlight 3 potential therapeutic targets: antibodies against S1, as well as ACE2, and TMPRSS2. I Wilson & D Burton of the Scripps Clinic using X-ray crystallography published in the 7/13/20 Science that “a set of B cell originated antibodies contain an unusually short variant of the CDR H3 loop, normally a key target-binding element, is particularly powerful at neutralizing the virus—and these potent antibodies are all encoded, in part, by the same antibody gene, IGHV3-53.”
M Wadman et al. in 4/17/20 Science “Front-line white blood cells release inflammatory molecules called CHEMOKINES, which in turn SUMMON more immune cells that target and kill virus-infected cells, leaving a STEW OF FLUID AND DEAD CELLS-PUS behind in the alveoli (air sacs). This is the UNDERLYING PATHOLOGY of the CoV2-19 PNEUMONIA, with its corresponding symptoms: coughing; fever; and rapid, shallow respiration.” Severe cases have multiorgan dysfunction, and hemodynamic instability (unstable circulation), as well as cardiovascular complications including myocardial injury, myocarditis, acute myocardial infarction (heart attack), heart failure, dysrhythmias (irregular heart beat), and venous thromboembolic events (clots). “…The most critical patients showed signs of organ function damage, including ARDS in 67%, pneumothorax in 2%, acute kidney injury in 29%, cardiac injury in 23%, and liver dysfunction in 29%.3 Moreover, in a study of 416 patients who required hospitalization due to COVID-19, 20% demonstrated signs of cardiac injury.3 Results of another study among 138 hospitalized patients with COVID-19 showed that 44% demonstrated a cardiac arrhythmia, and 38% had abnormal blood clotting ….”
JAMA 11/23/20 re clotting in CoV1-19 G Piazza, et al: “Diagnosis … Arterial and Venous Thrombosis….”
“Although the pathophysiology is not fully defined, prothrombotic abnormalities have been identified in patients with COVID-19. In a study of 19 critically ill patients with COVID-19, elevated levels of the following markers of hypercoagulability were identified: D-dimer in 100%, fibrinogen in 74%, and factor VIII in 100%.3 Antiphospholipid antibodies were detected in 53%, and decreased protein C, protein S, and antithrombin levels were detected in all.3”
Why patients develop life-threatening blood clots 8/26/20 MDLinx
Critical Care Explorations
Dr. Douglas Fraser, lead researcher from Lawson and Western’s Schulich School of Medicine & Dentistry, and Critical Care Physician at at London Health Sciences Centre (LHSC) measured 1,161 plasma proteins from the blood of 30 participants: 10 COVID-19 patients and 10 patients with other infections admitted to LHSC’s ICU, as well as 10 healthy control participants.
A major complication occurring in most critically ill COVID-19 patients is clotting in the lung’s small blood vessels which leads to low oxygen levels. “Most suspect the clotting mechanisms in our blood are put into overdrive and so many clinicians have been treating with anticoagulant therapies like the drug heparin,” said Dr. Fraser. “But we’ve uncovered an entirely different mechanism.” The team further analyzed the blood samples from their 30 participants, and found evidence suggests that the inner linings of small blood vessels are becoming damaged and inflamed, making them a welcoming environment for platelets to stick.
Three molecules (hyaluronic acid, syndecan-1 and P-selectin) identified the risk. The first two molecules are products broken down from small hair-like structures (the glycocalyx) which line the inside of the blood vessels. The glycocalyx is broken down by FURIN, a PCSK-3 protein. The 1st 2’s presence suggests the glycocalyx is being damaged with its breakdown products sent into the bloodstream. The presence of P-selectin is also significant as this molecule helps to make both platelets and the inner lining of blood vessels adhere to one another.
“The glycocalyx keeps platelets from touching the inside wall of the blood vessel and helps facilitate the production of nitric oxide, which has an important role in preventing platelets from sticking,” explained Dr. Fraser. “We suspect the body’s immune response is producing enzymes (such as FURIN) that shear off these little hair-like structures, inflaming blood vessels and making them a welcoming environment for platelets to form clots.” This change in vascular permeability is then acted upon mast cell-released bradykinin that further enhances vascular permeability, dilates blood vessels (causing hypotension and is one of the origins of the unoxygenated bubble-laden blood flowing through the lungs), allows seepage of hyaluronate, P-selectin, syndecan into pulmonary air sacs, and thereby predisposing to the hydrogels that cause the COv2-19 pneumonia.
SARS-CoV-2 replication targets nasal ciliated cells early in COVID-19 infection
by Institute for Basic Science 7/1/21
(A) en-face view of ACE2 protein signal in human nasal respiratory, nasopharynx transitional, and oral squamous epitheliums. Scale bars, 50 μm. (B) Cross-sectional view of human nasal epithelium showing robust ACE2 protein in acetylated-α-tubulin+ ciliated epithelium
Understanding how viral infection occurs can provide important clues for researchers to develop strategies to prevent viral transmission and develop effective therapeutic agents and vaccines. SARS-CoV-2, the causative agent of COVID-19, enters the host cells through interaction between the virus’s spike protein and the extracellular receptor binding domain of ACE2. The viral entry into the cells is completed by various proteases, which allow the viral and cell membranes to fuse together. While it is known that the upper respiratory tract becomes compromised in the early infection, the exact types of the cells that the virus infects at the earliest stage have not yet been identified.
Led by Director Koh Gou Young, scientists from the Center for Vascular Research within the Institute for Basic Science, South Korea, have recently uncovered the processes involved in the earliest stages of COVID-19 infection. The group accomplished this by combining immunofluorescence staining (IFS) and single-cell RNA-sequencing (scRNA-seq) of the molecules that are involved in the viral entry process. Through this, Koh and his colleagues identified the exact target of the coronavirus at the cellular level by comparing the results of the subjects infected with COVID-19 with those of healthy controls.
The researchers first looked for the presence of ACE2, TMPRSS2, and FURIN, which are the predominant SARS-CoV-2 entry-related molecules, on the surfaces of various types of cells in the nasal epithelium. It was found that these proteins were present in high quantities on ciliated cells. Moreover, the apical (upper) sides of these cells showed higher levels of these molecules in comparison to the basal (bottom) side. The levels of these proteins were highest in fully differentiated multiciliated cells. On the other hand, viral entry proteins were not found in the undifferentiated epithelial cells or secretory cells such as the goblet cells.
Next, the researchers further studied these nasal epithelial cells using scRNA-seq and visualized the cells into different clusters using UMAP. Samples were collected from four patients on the first day of their COVID-19 diagnosis, which were compared against two samples from healthy donors. It was found that the fraction of multicilial cells was greatly reduced in the samples from infected patients, while there was an increase in the secretory cells and differentiating cells. This hinted that multicilial cells were the first to be attacked and killed off by the virus, which are then replaced by the latter types of cells.
Multiciliated epithelial cells are the main site for SARS-CoV-2 replication and shedding in nasal respiratory epithelium (A) Schematic image depicting relative levels and tissue distributions of mRNA and protein of SARS-CoV-2 entry-related host molecules in human nasal mucosa epithelial cells. Note that the protein but not mRNA of SARS-CoV-2 entry-related host molecules are co-localized in the apical side of nasal multiciliated epithelial cells. (B) Infected SARS-CoV-2 highly replicates and accumulates in nasal multiciliated epithelial cells, which rapidly shatter and scatter the viruses in the early stage of COVID-19. Credit: Institute for Basic Science
The study also measured the level of SARS-CoV-2 mRNA transcripts within the various types of cells. Among all the epithelial cells in COVID-19 infected patients, 38% of the cells were labeled as SARS-CoV-2hi cells. Up to 75% of the detected genes in these cells were of viral origin, compared to less than 1% for other clusters of cells. This means that these cells serve as the main factories for the mass production of SARS-CoV-2 viruses. While it was not possible to directly identify these cells through RNA seq due to the fact that they primarily produce viral mRNA, the researchers employed pseudo-time-trajectory analysis to predict their differentiation paths. The differentiation trajectory showed that SARS-CoV-2hi cells likely originated from ciliated cells. Further IFS analysis on the infected patients’ samples conclusively determined that up to 85% of SARS-CoV-2 factories were in fact multiciliated cells.This study was able to determine that multiciliated cells in the nasal epithelium are the first cells that are targeted in the early COVID-19 infection. This implies that targeting these cells using specific treatments, such as through nasal sprays, can be an ideal strategy to curb COVID-19 infection in the early stages.
Identification of two pathways for SARS-CoV-2 entry into cells
More information: Ji Hoon Ahn et al, Nasal ciliated cells are primary targets for SARS-CoV-2 replication in the early stage of COVID-19, Journal of Clinical Investigation (2021). DOI: 10.1172/JCI148517
An EXCELLENT (!) and lucid (!) 7/20/20 discussion of INATE & ADAPTIVE IMMUNITY, T & B-CELL roles from Newsletter Science X is:
https://medicalxpress.com/news/2020-07-coronavirus-cells.html?utm_source=nwletter&utm_medium=email&utm_campaign=daily-nwletter
Postmortem Examination of Patients With COVID-19
Tina Schaller and Rainer Claus et al JAMA. Published online 5/21/20. doi:10.1001/jama.2020.8907
In all cases, including 6 patients who did not receive invasive ventilation, disseminated diffuse alveolar damage at different stages (the histopathological correlate of acute respiratory distress syndrome) was the major histologic finding. Diffuse alveolar damage was detectable in all lobes but appeared unevenly distributed with pronounced manifestation in middle and lower lung fieldS (Figure, A-B). Signs of exudative early-phase acute diffuse alveolar damage with hyaline membrane formation, intra-alveolar edema, and thickened alveolar septa with perivascular lymphocyte-plasmocytic infiltration were consistently found. Organizing-stage diffuse alveolar damage with pronounced fibroblastic proliferation, partial fibrosis, pneumocyte hyperplasia leading to interstitial thickening and collapsed alveoles, and patchy lymphocyte infiltration was the predominant finding. In areas of organizing diffuse alveolar damage, reactive osseous and squamous metaplasia were observed (Figure, C-G). Fully established fibrosis was most prominent in patient 1, ultimately leading to almost complete destruction of pulmonary parenchyma. In 5 patients, minor neutrophil infiltration was indicative of secondary infection and/or aspiration.
See the comment of world-famous virologist David Ho below.
10/2020 Financial Times SUPERB article AND GRAPHS/ILLUSTRATIONS on CoV2-19 TIMELINE:
https://ig.ft.com/coronavirus-global-data/
KEY DATES IN COv2-19 The daily telegraph Newspaper
HRS inserts: Satellite data confirms increased traffic near Wuhan Hospitals as early as October, 2019.
11/9/15: Wuhan (China) Institute of Virology publish a study revealing they created a new virus in the lab from SARS-CoV2-19.
12/6/19: Five days after a man linked to Wuhan’s seafood market presented pneumonia-like symptoms, his wife contracts it, suggesting human to human transmission. (studying patients with COVID-19 in Germany, there were no viable isolates of the virus after the first week of symptoms.21)
12/27/19: China’s health authorities told a novel disease, then affecting some 180 patients, was caused by a new coronavirus.
12/26-30/19: Evidence of new virus emerges from Wuhan patient data.
12/31/19: Chinese internet authorities begin censoring terms from social media such as Wuhan Unknown Pneumonia.
1/1/20: Eight Wuhan doctors who warned about new virus are detained and condemned.
1/3/20: China’s top health authority issues a gag order.
1/5/20: Wuhan Municipal Health Commission stops releasing daily updates on new cases. Continues until January 18.
HRS inserts: in JANUARY 2020, imports to China of surgical face masks were increased by 278% and surgical gowns by 72 %, China also decreased its global exports of gloves, gowns and face masks, exports of medical ventilators decreased by 45 %: these conclusions are based on the 95% probability that the changes in imports and exports were not normal for this time of year.
1/10/20: PRC official Wang Guangfa said outbreak “under control” and mostly a “mild condition”.
1/12/20: Professor Zhang Yongzhen’s lab in Shanghai is closed by authorities for “rectification”, one day after it shares genomic sequence data with the world for the first time.
1/14/20: PRC National Health Commission chief Ma Xiaowei privately warns colleagues the virus is likely to develop into a major public health event.
1/24/20: Officials in Beijing prevent the Wuhan Institute of Virology from sharing sample isolates with the University of Texas.
2/6/20: China’s internet watchdog tightens controls on social media platforms.
2/9/20: Citizen-journalist and local businessman Fang Bin disappears.
4/17/20: Wuhan belatedly raises its official fatalities by 1290.
China notified the World Health Organization/WHO of this infection 12/31/19 saying “the (corona virus 19/CoV2-19) disease is preventable and controllable” incorrectly at that time. 7 million people had been in, and then left Wuhan to go else(every)where outside of China beginning December, 2019. Flights were completely stopped from Wuhan to Shanghai, Beijing, and the rest of China, but not elsewhere, during that period of time. As noted, in contrast to the rest of the world where every large city has been affected by the corona virus CoV2-19, ALL other large Chinese cites were REPORTED to have had virtually no incidence of the corona virus because of that policy. January, 21, 2020, China stated, and the World Health Organization (WHO)’s Dr. Tedros repeated, that this corona virus could not be transmitted from person to person: that, too, was wrong. The first overseas CoV2-19 case was documented 1/15/20. The first USA case was found 1/21/20. 1/19/20, a 35-year-old man with a 4-day history of cough, fever, and recent travel history to Wuhan, China, presented to an urgent care clinic in Snohomish County, Washington. 1/20/20 the CDC confirmed that samples from the patient’s nasopharyngeal and oropharyngeal swabs tested positive for CoV2-19 making this man the first confirmed case of the coronavirus disease of 2019 (COVID-19) in the USA.
The city of Wuhan, China, (not the whole country) lockdown began 1/31/20. The shutdown of immigration to the USA for non-Americans began 1/31/20. The virus is in 213 countries in the world.
In 1348, the BLACK PLAGUE killed 1/3 to 1/2 (50%!) of the WORLD’s population. That was due to the bacteria Pasteurella or Yersinia pestis carried by the rat flea. The quite recent MERS = Middle Eastern Severe Respiratory disease was 33% lethal! For a more recent perspective, the HONG KONG 1968-1969 FLU ravaged the world; it killed more than one million (1,000,000) worldwide, over 100,00 in the USA with no lockdown nor closure of businesses, but with social distancing: this memory may seem unbelievable, yet it is true and similar to now.
The 2009-2010 SWINE FLU H1N1 virus originated in Mexico when there was an open Southern USA border. The WHO declared the H1N1 virus an imminent threat on 4/29/09, however, the WHO waited until 6/11/2009 to declare it a world pandemic. The Swine Flu virus was in the United States 7 months before the government called it a national emergency and that was several months after it was named a pandemic by the World Health Organization (WHO). Learning from the past, the current national emergency was so named 2 months after the virus was first here 1/21/20 and several days after the WHO called it a pandemic. As for the seasonal flu, the CDC website for 2019-2020 now projected 48,000,000 illnesses, 22,000,000 medical visits, 550,000 hospitalizations and 43,000 deaths. In harsher flu years, 60-80,000 Americans are lost. “America endured six million (6,000,000) traffic accidents and 36,000 fatalities, over 16,000 and 90 per day respectively. During the current CoV2-19 the number of deaths as of 7/20/21 is 625,000; that is overall 1.5 in 10,000 for the total USA population. 4,000,000 have recovered. Kurt Silverfiddle: “Every human activity has a fatality rate, and a responsible society works to minimize death and injury. Zero traffic deaths is a laudable goal but not economically feasible. No one in the U.S. bats an eye at 35,000 to 60,000 influenza deaths each year.” The 1957 Asian flu, a form of H2N2 influenza that is believed to have originated in China, is estimated to have killed 116,000 Americans, the equivalent of roughly 200,000 in today’s larger America. The corona virus attacks the elderly and the infirm, seldom children. Influenza, in contrast, affects the younger and children.
As further perspective, in 2015, Ian Goldin, an Oxford University professor, in his book The Butterfly Defect, warned of the risks of a “global pandemic in a modern, interdependent world”: no one in the USA federal/state/local governments and other governments, listened or prepared for the future thus affecting our ability to appropriately respond now. These are historical facts.
8/2021: …The latest CDC data reveals that the total of children under 18 who have died in the U.S. from CoV2-19 since the start of the pandemic sixteen months ago is 361 — in a country of 330 million people, including 74.2 million people under 18. Instead, the “devastating toll” refers to multi-layered harm to children from the various lockdowns, isolation measures, stay-at-home orders, school closures, economic suffering and various other harms that have come from policies enacted to prevent the spread of the virus …”
The 2019-2020 FLU vs CoV2-19: the latter is more serious. See this comparison link.
https://www.nola.com/news/coronavirus/article_e33c0cf0-7090-11ea-b3da-53f5ab31dd4b.html
There is no prior experience with this current CoV2-19 virus; it spreads relatively easily. That spread has slowed significantly in China. 95+% of the Chinese population are reported to not have contracted the virus. In China, infectivity decreased from 2 to 3 other people per infected case to 1.5 per case. In the beginning, the infection rate doubled every five days as documented with increased testing. Newer information from the USA’s Los Alamos National Laboratory states that 5 people are infected per case. Influenza (the flu) spreads to 1.7 people per original case. A wide screen in San Diego, Calif, implies that there may be 30-50 million in the USA who are already infected. Justin D. Silverman et al, using influenza surveillance networks to estimate state-specific prevalence of SARS-CoV-2 in the United States, 6/22/20 believes that there may 50 times more cases of CoV2-19 than are currently identified, Science Translational Medicine. DOI: 10.1126/scitranslmed.abc1126 , stm.sciencemag.org/content/ear … scitranslmed.abc1126 . JAMA NETWORK 7/21/20 Tyler S Brown: “The estimated total number of infections (extrapolated from the measured proportion of individuals with positive SARS-CoV-2 serologies) was between 6- and 24-fold higher than the number of confirmed COVID-19 cases reported in each location prior to the study.”
Social distancing to break the chain of viral transmission continues, but individual states began to return to normal as of 4/17/20. “We are past the point of being able to contain or eliminate the virus”. 7/5/20: the WHO PRAISED Sweden’s handling of the pandemic, saying that Sweden “deserves respect”!! 5/25/20 Newsletter Science X: Sweden, which has a 16% lower incidence of obesity, shut down minimally = social distancing + protection of the elderly. The Swedish fatality rate was initially similar to the USA (with NO ((!)) lockdown). Sweden’s open economy death toll surpassed neighboring Nordic countries that imposed more restrictive containment measures. According to website Worldometer, Sweden’s virus death rate of 399 per million inhabitants WAS INITIALLY HIGHER than Norway’s death rate of 43 per million, Denmark’s at 97, or Finland’s at 56-all with closed economies. However, it is still lower than France’s 435, the UK’s and Italy’s 542, and Spain’s 615-all with closed economies. Critics accused Sweden of gambling with the lives of citizens by not imposing strict stay-at-home measures. Sweden’s death rate in 8-10/2020 is 1/6th the USA rate. The obesity rate is 16% lower in Sweden than in the USA, Predisposing risks are reduced and I (HRS) believe that social distancing is much more achievable in Sweden’s low population-large area nation, combined with likely societally encouraged more appropriate/risk averse public behavior. The Swedish Public Health Agency stated via Anders Tegnell, stated that their more relaxed approach is sustainable in the long-term and it rejected drastic short-term measures as too ineffective to justify their impact on society. Sweden kept schools open for children under the age of 16, along with cafes, bars, restaurants and businesses, while urging people to respect social distancing and hygiene guidelines. “As the report from Business Insider confirms, the Swedish economy “was the least harmed in Europe”, the “best of the bunch” (and) “the only major economy to grow in the first quarter of the year.” Sweden was progressively ramping up its activity while the United States was still stuck.” State epidemiologist Tegnell of the Public Health Agency stated that stricter measures would not have saved more
lives. Medical Xpress 7/21/20: “Swedish officials have argued that lockdowns only work temporarily and that drastic short-term measures are too ineffective to justify their impact.” T-cell immunity is the likely answer and double that of antibody immunity.
Medical Xpress 10/14/20: “Sweden, which didn’t have a compulsory lockdown at all but put in place many voluntary measures. It had considerably more excess deaths than its Scandinavian neighbors that did lock down, and this pattern remained for longer than almost all these countries. Overall though, Sweden had fewer excess deaths than several countries that did lock down—possibly due in part to relatively low levels of other illnesses, possibly because of high levels of compliance with the voluntary measures.”
Sweden tops EU virus case rates
Credit: CC0 Public Domain5/20/21
For the first time this year, Sweden’s incidence rate of coronavirus cases has topped European Union figures, the EU’s health authority said Thursday, but associated deaths are among the lowest. The country had a 14-day-notification rate of 577 cases per 100,000 inhabitants, according to the European Centre for Disease Prevention and Control. The vast majority of positive cases in Sweden are asymptomatic or minimally symptomatic.
Passing one million cases in early May, Sweden has been near the top, after Cyprus, for weeks with new cases subsiding quicker in the rest of Europe. The Scandinavian country has never imposed the type of lockdown seen elsewhere in Europe, controversially relying on mostly non-coercive measures.
It has however gradually tightened restrictions since November, including a ban on alcohol sales after eight pm and on public gatherings of more than eight people. Since March, cafes, bars and restaurants have also been required to shut by 8:30 pm.
The 14-day-notification for deaths however was much lower than many other countries, with 12 cases per million inhabitants. In comparison, Hungary and Croatia saw death rates of 133 and 128 per million inhabitants respectively.
The total number of deaths associated with COVID-19 since the start of pandemic reached 14,351 on Thursday, putting Sweden in the middle of the pack in Europe, although well ahead of Nordic neighbours Finland, Norway and Denmark,
Mortality statistics also show Sweden had a lower than average excess mortality in 2020, compared to the rest of Europe.
SWEDEN and CoV2-19 AS OF 8/8/21
While many U.S. policy makers are suggesting that states return to lockdowns and impose vaccine or mask mandates to engage in everyday life as new coronavirus variants surface, Sweden’s population of 10.3 million fared better than expected after implementing no lockdowns over the last year.
In April of last year, Björn Olsen, professor of infectious medicine at the University of Uppsala, issued a report suggesting that if lockdowns weren’t implemented a “tragedy” would soon befall Sweden. An Imperial College model suggested that 85,000 people would die without a lockdown, and an Uppsala University team projected that 40,000 people would die from COVID-19 by May 1, 2020 and nearly 100,000 by June.
But by May, Sweden reported roughly six deaths for every one million people, according to the Financial Times, with 48.9% of its initial coronavirus deaths taking place in nursing homes, according to an analysis by the Swedish Public Health Agency. More than a year later, Sweden recorded 1.1 million coronavirus cases with 1.07 million people having recovered from the virus, and 14,620 coronavirus-linked deaths, according to woldometers.info as of Aug. 8, 2021.
Of the currently 12,248 people who have tested positive for COVID-19, 12, 219 are experiencing mild symptoms (99.8%) and 29 (0.2%) are in serious or critical condition, according to woldometers.info.
While the Financial Times warned that Sweden wouldn’t likely see an economic benefit from its no-lockdown approach, in May 2020, Sweden fared better than expected. According to an analysis by Nordetrade.com: “Covid-19 has had a rather limited impact on [Sweden’s] economy compared with most other European countries. Softer preventative restrictions against Covid-19 earlier in the year and a strong recovery in the third quarter contained the GDP contraction.”
With the rise of the so-called Delta variant, Sweden has held fast in not imposing a lockdown or a mask mandate, having lifted its last face covering requirement on public transit July 1. Still, its population had a total of eight coronavirus-related deaths in July, or an average of 0.26 coronavirus-linked deaths a day. Daily hospitalizations were also close to zero in July, with between zero and two COVID-19 cases requiring hospital treatment nationwide.
Sweden’s numbers are considerably lower compared to 74 and 329 coronavirus-related daily fatalities in the U.K. and U.S., respectively, over the same period. Sweden announced it plans to administer booster doses of the COVID-19 shots for its most vulnerable citizens beginning in September. “The assessment is that it is not possible to eradicate the virus and therefore vaccination work should be long-term and focused on reducing serious illness and death,” Anders Tegnell, Sweden’s chief epidemiologist recently announced.
Sweden’s Public Health Agency expects its entire adult population will have received two doses of COVID-19 shots by the fall, and that many will receive a third shot next year. Tegnell said there is “a lot we do not know” about Delta and that it had been circulating in Sweden “for quite some time” with little impact, especially in high-risk environments like nursing homes.
His remarks came after the Centers for Disease Control and Prevention suggested the Delta strain is more transmissible than the original coronavirus, even as contagious as chicken pox, and could potentially cause severe illness. The agency has not explained its testing methodology, as critics have pointed out tests to determine variants are not available on a national scale, making it difficult to determine if someone who tests positive for the coronavirus has a variant or not.
“It is difficult to say how contagious Delta is, [as] when it comes to chickenpox, we have been able to follow the disease for several years,” Tegnell told Sweden’s Aftonbladet. “The infectivity [of Delta] seems to be very uneven — in some cases, one person infects a hundred people, then we have other occasions when an infected person does not infect anyone at all.”
Sweden’s public health agency has recommended that Swedes continue to social distance and work remotely but has not issued mask or face covering mandates in public spaces. Sweden remains open while other countries are considering re-instituting lock-downs, and still others like France, have announced vaccine mandates as a condition to be able to participate in society.
These countries got COVID-19 under control. Here’s 3 things they did right (condensed)
Aria Bendix in Business Insider.11/4/20: an organized federal response and the populace trusting and willing to comply is most important…” Monica Gandhi at the U of California, San Francisco, told Business Insider.
Emma Hodcroft, a scientist from Basel, Switzerland sees a common trend among governments that got the virus under control: They have a plan in place in case cases rise, communicate it clearly to the public, and enact it quickly whenever numbers start going up, avoiding the common approach with too much inertia.
Even Sweden, recently adopted stricter guidelines about gatherings and non-essential recreation after cases there surged 70 percent in one week. Australia, China, & New Zealand used location-targeted lockdowns to over the last several months, requiring shutdowns only in cities and regions experiencing outbreaks.
Japan’s government put out messaging early on instructing citizens to avoid the 3 C’s: closed spaces, crowded places, and close-contact settings. Then the country used a comprehensive system of regional healthcare facilities to expand testing and public-health communication. Combined, these actions meant Japan didn’t have to lock down at all.
October 26, 2021
Sweden as CoV2-19 Outlier or Success 10/21/21
Sweden which has stood out among European nations for its comparatively hands-off response to the pandemic, has passed the threshold of 15,000 deaths with COVID-19 since the beginning of the pandemic, according to official figures released Tuesday. Thomas Linden of the National Board of Health and Welfare, told Swedish public radio that it was difficult to say whether Sweden has an unusually high excess mortality due to the virus.
“Internationally, Sweden has not had a higher mortality rate. But if you compare with the other Nordic countries, we are significantly higher,” Linden told SR. In comparison, Denmark has recorded 2,703 deaths, Norway 895 and Finland nearly 1,150. Each of those countries has slightly over half as many people as Sweden.
According to the Public Health Agency of Sweden, 15,002 people—6,793 women and 8,209 men—have died with COVID-19 since the beginning of the pandemic.
Sweden had opted for keeping large sections of society open. It has not gone into lockdowns or closed businesses, relying instead on citizens’ sense of civic duty to control infections.
JAPAN & CoV2-19 An amazing statistic: 5/5/21 NYT, D Leonhardt
What did Japan do right?
It already had a culture of mask wearing to prevent illness, and masks became almost ubiquitous early last year. (This article by Motoko, from almost a year ago, compares mask habits in Japan and the U.S.) The government also virtually closed its borders. And it was quick to focus on the settings where the coronavirus was most likely to spread, warning people to avoid the “three C’s” — closed spaces, crowded places and close contact.
Carbon dioxide levels among children wearing face masks exceed healthy limits.
HealthDay News — Carbon dioxide levels among children wearing face masks may exceed healthy limits, according to a research letter published online 6/30/21 in JAMA Pediatrics.
Harald Walach, Ph.D., from the Poznan University of the Medical Sciences in Poland, and colleagues assessed whether nose and mouth coverings increase carbon dioxide in inhaled air. The analysis included 45 children (mean age, 10.7 years) who tested two types of nose and mouth coverings (a surgical mask and a filtering facepiece 2 mask) in a laboratory-like setting.
The researchers found that levels were similar between the masks, with means ranging from 13,120 to 13,910 ppm of carbon dioxide. Only age was associated with carbon dioxide content in inhaled air. The youngest children had the highest carbon dioxide values, with carbon dioxide level measured at up to 25,000 ppm. The limit of 0.2 percent by volume (2,000 ppm) was exceeded by more than threefold among children with the lowest carbon dioxide levels.
“We suggest that decision-makers weigh the hard evidence produced by these experimental measurements accordingly, which suggest that children should not be forced to wear face masks,” the authors write.
By The New York Times | Sources: Governments, health agencies and hospitals
Japan has not defeated Covid. Cases have risen over the past two months, and the government declared a state of emergency in several major cities, urging new restrictions on activity. “Japan has recently lost a little control of the caseloads,” Motoko says. “Of course, it’s nothing like New Delhi, but it’s not like Sydney or Taipei, either.”
By The New York Times | Sources: Governments, health agencies and hospitals
South Korea, for its part, leveraged smartphone technology to communicate its response and give the public clear information. Its government provided free apps that sent people emergency text alerts about spikes in infections in their local area, granted access to telemedicine, and informed users about the number and type of face masks available at stores for purchase.
Science Newsletter X 8/21/20: “…coronavirus cases have been dropping sharply in the United States for week. After exceeding 70,000 confirmed infections per day in July, 2020, the country recorded 43,000 cases 8/20/20. Hospitalizations have fallen by a third. “I do think we’re going to start to see significant declines in the mortality across the country in the hopefully next week,” said Robert Redfield, director of the Centers for Disease Control and Prevention during a talk hosted by the medical journal JAMA….” 100,000 a day in the USA by 11/6/20: BUT younger, handled much better, and less severe in general.
Delaying Herd Immunity Is Costing Lives” 4/29/20 by bio-statistician Martin Kuldorff, PhD of Harvard Medical School’s Brigham and Women’s Hospital
“…The question is not whether to aim for herd immunity as a strategy, because we will all eventually get there. The question is how to minimize casualties until we get there. Since Covid-19 mortality varies greatly by age, this can only be accomplished through age-specific countermeasures. We need to shield older people and other high-risk groups until they are protected by herd immunity.
Among the individuals exposed to Covid-19, people aged in their 70s have roughly twice the mortality of those in their 60s, 10 times the mortality of those in their 50s, 40 times that of those in their 40s, 100 times that of those in their 30s, and 300 times that of those in their 20s. The over-70s have a mortality that is more than 3,000 times higher than children have. For young people, the risk of death is so low that any reduced levels of mortality during the lock down might not be due to fewer Covid-19 deaths, but due to fewer traffic accidents….” … “Under-18s have accounted for just two percent of hospitalized COVID-19 cases and less than 0.1 percent of all deaths in the United States, according to statistics from the Centers for Disease Control and Prevention (CDC).
A total of 45 children died from the coronavirus in the United States between February 1 and August 1—compared to 105 who died from seasonal flu—out of a total of 13,000 children who died of all of causes over the period.”
After 6 months without lockdown, Sweden’s COVID-19 deaths, infections bottom out
Updated: 8/2/20 By Daniel Payne
After months without lockdowns, school closures and other mitigation measures widely imposed across the world, Sweden’s coronavirus cases and deaths have fallen to such minimal levels as to revive the debate over its so-called herd immunity strategy.
Some Swedish officials are far from declaring victory, warning there could be a second wave and that too many elderly died in the country during its comparatively lax pandemic restrictions. The country’s population-adjusted death rate, meanwhile, is in the top 10 worldwide, but lower than the rates for Italy, Spain and even New York, where heavy lockdowns prevailed.
And the dramatic drop in new cases and deaths in that country point to a rapidly improving situation there in defiance of many earlier predictions. The Swedish government has engaged in minimal interventions and imposed relatively few restrictions upon its citizenry for the duration of the pandemic.
There is relatively little evidence to support social distancing, masks, and lockdown measures, many governments — particularly those in Western Europe and the United States — have for several months imposed these mandatory lockdown, masking and social distancing orders on their respective citizens, with numerous European heads of state and U.S. governors indicating that these measures may remain in place until an effective vaccine is developed, a process that could take years, if ever, to come to fruition.
Heavy mitigation has no ‘historical scientific basis’
Sweden largely avoided such policies. Throughout March, as much of the Western world was shutting down large swaths of its economies and strictly limiting individual mobility with stay-at-home orders, Sweden opted for a much lighter touch, refusing to close down service industries, leaving schools largely open, and allowing its borders to remain open. It did restrict large gatherings for a time, some schools were closed.
According to the World Health Organization, Sweden’s daily deaths peaked in late April and have been declining ever since; on some recent days, the country has recorded as few as nine deaths. Daily new cases were in the low-to-mid-hundreds for most of July, and a few days no new cases were recorded at all.
Anders Tegnell is the country’s chief epidemiologist.
The country has been the subject of withering criticism since March. In July, the New York Times said that the country was a “cautionary tale” for the world. The National Post in June said the Swedish model “failed” and that the country “took the pain, but realized no gain.” In May, Wired stated that the country’s epidemiological experiment “well and truly failed.”
In justifying those claims, numerous commentators correctly pointed out that Sweden’s population-adjusted death rate is significantly elevated compared to its Scandinavian neighbors Finland and Denmark, both of which have death rates 10 to 12 times lower than Sweden’s. Indeed, Sweden’s adjusted death rate is No. 8 in the world, 20% higher than the United States. Tegnell has blamed those high numbers partly on the number of elderly deaths in the country, claiming Sweden did not do enough early on to protect nursing homes and retirement communities from infections.
Perhaps more pointedly, in countries that imposed some of the more draconian lockdowns in the Western world — the U.K., Italy, Spain — the pandemic at times seemed to rage out of control compared with Sweden’s relatively level epidemic curve. Italy and Spain at the height of their outbreaks recorded many hundreds of deaths per day and saw, at times, local health systems on the verge of collapse.
Both countries have population-adjusted fatality rates above that of Sweden; smaller government authorities, such as those of New York and New Jersey, also have fatality rates far above Sweden’s. New York State, where Gov. Andrew Cuomo instituted one of the strongest and most restrictive lockdowns in the U.S., has a population-adjusted death rate three times that of Sweden. Tegnell and others have argued that lockdown countries likely have significantly lower immunity levels than Sweden’s, and that those countries may have to lock down again in the fall if and when the disease returns.
Many experts have claimed that COVID-19, like some other respiratory diseases, will only be subject to the effects of herd immunity when anywhere from 60-80% of the population recovers from it; reaching those levels, it is argued, would require unacceptable levels of deaths.
Yet there are indications that the herd immunity threshold for the coronavirus may be much lower than that, possibly due to the presence of protective “T cells” gained from earlier epsoure to other types of coronaviruses. That theory, if true, could explain why Sweden’s death rate declined even as cases increased there.
Much of the world effort to determine the disease’s herd immunity threshold has hinged upon testing individuals for COVID-19 antibodies, which signify a patient once had the disease and has since recovered from it. A study released in mid-May found that just 7.3% of Stockholm residents had coronavirus antibodies, far below even the lower bounds of most estimates of the herd immunity threshold.
But peak infections in Sweden occurred roughly a month after that study even as deaths there continued to plummet — an unlikely scenario if 92.7% or more of the country had absolutely no immunity to the disease.
Sweden’s steadily declining deaths and cases, then, may represent an advantage obtained by few other countries at this point. Indeed, other countries that appeared to have earlier success in clamping down on the pandemic — such as Australia, Japan, Belgium, the Netherlands, Morocco, and others — have all lately posted rising numbers of cases, suggesting those populations may at this time be more susceptible to the virus than is Sweden’s.
Other countries in Europe, meanwhile — including Germany, France, Spain, and Belgium —have all seen recent spikes in cases after several months of declining and plateaued infection rates, suggesting that those populations may have not achieved the same degree of herd immunity that Sweden appears to have developed.
Tegnell has suggested as much, though he has expressed optimism over Sweden’s prospects moving into the fall. “In the autumn there will be a second wave,” he told Financial Times earlier this year. “Sweden will have a high level of immunity and the number of cases will probably be quite low.”
A notable body of medical literature has found large-scale quarantine efforts are ineffective at stopping the spread of respiratory diseases. A 2006 paper in Biosecurity and Bioterrorism, for instance, found “no historical observations or scientific studies that support the confinement by quarantine of groups of possibly infected people for extended periods in order to slow the spread of influenza,” a disease which, like COVID-19, is spread through respiratory droplets.
That same year, meanwhile, a World Health Organization writing group surveyed the evidence in favor of quarantining individuals during pandemic influenza and found support for those policies lacking. Throughout the pandemic, Swedish authorities have insisted that their country’s approach was one rooted in years of epidemiological research and that much of the rest of the world abandoned that data in favor of panic and hysteria.
“It was as if the whole world had gone mad,” Tegnell said several weeks ago, citing the worldwide rush to lock down and quarantine. “The cases became too many, and the political pressure got too strong. And then Sweden stood there rather alone.”
A Different Perspective, more critical, of Sweden: Strategy
Norway
“Sweden: an article from 1/7/21: QUITE pointed. HRS (this author) is not sure of the accuracy or the interpretations here, but these 2 articles are powerful
What are the specific differences between Sweden’s and Norway’s Covid-19 strategies? Looking for details here not a general summary. For context: I am a Canadian living in Sweden and wish to understand how we can learn from the Norwegian strategy.
First wave
The initial Norwegian response strategy to the Covid-19 pandemic that was hastily implemented on March 12 of 2020 was quite firm, and mostly followed what Denmark had done the previous days. The measures included:
• Near-total shutdown of schools, kindergartens and universities, with only the children of people performing criticial societal functions allowed to attend.
• Shutdown of cultural and sports events, gyms, hair dressers, bars, many restaurants and similar places where lots of people are usually gathered.
• Quarantine for anyone entering Norway.
• Strict access control at health institutions where individuals in risk groups were living.
The measures were slightly more lenient than in Denmark, where all restaurants and shopping centers were closed, and the borders were shut. None of these measures were implemented in Sweden. Perhaps the strictest measure in Sweden was a maximum limit of 500 people gathering outdoors, whereas the limits in Denmark and Norway were 10 and 5 people respectively.
By March 24, The Norwegian Institute of Public Health (NIPH), a research institute, published a report ordered by the government, outlining three possible strategies to tackle the pandemic going forward.
• “Release” with an estimated reproduction rate (R) of 2.4
• “Restrain” with an estimated R of 1.3
• “Suppress” with an estimated R of 0.9
Realistically, only the last two strategies were considered by the Norwegian Directorate of Health and the government, which had the ultimate say in how to move forward. The government announced that Norway would go ahead with a forceful suppression strategy, aiming to “knock down” the pandemic, even though NIPH hinted strongly that the burden of the measures required in this strategy would be enormous on the Norwegian population. In practice, this meant a continuation of the measures that were already in effect.
Meanwhile, the Swedish strategy was somewhere between Release and Restrain, but probably closer to Release. This was unique in Western Europe, where most countries were implementing even stricter measures than Norway, with the exception of Great Britain, which followed a similar strategy to Sweden in the early stages.
Summer break
The suppression strategy was even more effectful than NIPH had anticipated. By May, the R number in Norway was down to 0.4. The strategy had widespread support in the Norwegian population, and most citizens abided by the strict rules. By mid-summer, there were only 10–20 new cases daily in all of Norway, and most of the restrictions were lifted. Meanwhile, Sweden had more than a thousand cases a day in June and a few hundred in July.
Second wave
Since late 2020, Scandinavia has experienced a second wave of the pandemic even worse than the first. Norway implemented stricter measures again in the weeks leading up to Christmas holiday, hoping that we would be able to loosen up again in time for Christmas, but the numbers kept increasing.
In the first week of 2021, the numbers were higher than ever, and very strict measures were introduced again. The situation is now similar to March/April last year, with the exception of schools and kindergartens staying open. After the first wave, NIPH concluded that the effect of closing these were minimal. At the same time, it was the most burdensome measure. Another difference is an increased use of face masks, which was not enforced during the first wave.
Sweden has been hit much harder than Norway in the second wave as well. The measures in Sweden are stricter this time, with home schooling for high school students, strict regulations for restaurants and no alcohol sales after 8 in the evening.
Eamon Lynch, former UN Peacekeeper and Delegate to the EU
1. Death’s per million is many times higher in Sweden than Norway. Norway and neighbouring Finland are amongst the developed countries with the lowest death rate of COVID well down the first quartile of death rates.
2. Sweden is well up the third quartile in deaths per million. WorldOMeters sourced today records for Jan 6, 2021 104 deaths per million population for Finland.
3. WorldOMeters recorded 86 deaths per million population for Norway. WorldOMeters recorded 250 deaths per million population for Denmark.
4. The average for Sweden’s three neighbouring countries is 147 deaths per million population. WorldOMeters recorded 911 deaths per million population for Sweden.
5. The death rate from COVID-19 in Sweden is more than ten times higher than in Norway. The death rate from COVID-19 in Sweden is more than six times higher the average of its three adjoining countries.
6. Let us examine the facts underlying this disparity. The population of Sweden is 10.1 million. The population of Norway is 5.4 million. The population of Denmark is 5.8 million. The population of Finland is 5.5 million. With nearly twice the population of Norway about twenty times more Swedes have died of Covid-19 than Norwegians.
7. The land area of Norway is 625,217 sq km. The land area of Sweden is 447,430 sq km. The land area of Finland is 338,450 sq km. The land area of Denmark is 42.920 sq km.
With a land area half as big again as Sweden, Norway has a less dense population spread than Sweden. However, the population density in Denmark and Finland is significantly more dense than Sweden. Population density is a factor in COViD spread. In the case of Sweden it does not provide us with much more than a very small part of the explanation for Sweden’s high death rate from COVID-19.
Between January and July 2020 as COVID became a pandemic and the number one issue in the world Sweden stayed open and was one of the few countries to try to continue “as normal” whatever that is wherever. This approach had value as an experiment if nothing else. As an experiment it has proven to produce a poorer outcome than alternative strategies.
Experts who know much more than I do suggest that the Swedish deaths are or could be (very slightly) overstated because Sweden adopts a reporting regime that initially places doubtful COVID deaths in the COVID column. However, these experts do not include Norway, Denmark nor Finland in any category that might record cases significantly differently than in Sweden.
Sweden has about 20 per cent of its population over 65 years of age. Norway has about 17.5 per cent. The age of the population must therefore be examined as a causative factor. However, Finland has a higher percentage over 65 and Denmark has a similar level over 65. We can therefore rule out the population age profile as a substantial contributor to excess increased COVID morbidity.
if we examine all mortality levels from 2015 to late 2020 we see that mortality levels in Norway during 2020 remained at the same as 2015–2019 levels. In Sweden there was a higher mortality rate in 2020 compared to 2015–2019 levels (Juul et al, 2020). That study found Sweden had a lower than expected mortality before the pandemic and excess mortality during the first wave and lower than expected mortality after the first wave. The current wave, if the study was ongoing, would also show excess mortality.
The key errors in Sweden’s COVID-19 strategy (having explained and measured some main but not all surrounding factors), in 2020 were: Keeping schools open. Keeping restaurants open. Keeping Gyms open. Banning gatherings of over 50 people and asking people to self isolate failed as an effective policy and strategy.
Sweden did not ban outside travelers. Norway banned outside travelers and closed tourist attractions Finland restricted border traffic and closed schools. Finland was better prepared with PPE than Sweden. Finnish policy had been to constantly stockpile medical supplies, a policy continued since the Cold War.
Finland, Denmark and Norway were better off in the number of their ICU beds. Sweden has 5.8 ICU beds per 100,000. Norway has 8 ICU beds per 100,000. Sweden had a State Epidemiologist who created their strategy and while he defends it with statements of plateauing the figures clearly show now that the strategy was plain wrong in almost every way.
When other experts suggested change (2,300 academics signed an open letter). The Swedish approach places responsibility in the hands of the public. Therefore, the Swedish approach has three underlying strengths. It is democratic, it will result in less stress and depressive illness, and, when it is over, Sweden will recover faster.
None of these three factors are compelling to a Canadian living in Sweden.”
Read the Comments in this article for a countering of the just above article here: “Mortality in Norway and Sweden before and after the Covid-19 outbreak: a cohort study”
https://www.medrxiv.org/content/10.1101/2020.11.11.20229708v1#disqus_thread
View ORCID ProfileFrederik E Juul, View ORCID ProfileHenriette C Jodal, View ORCID ProfileIshita Barua, View ORCID ProfileErle Refsum, Ørjan Olsvik, View ORCID ProfileLise M Helsingen, View ORCID ProfileMagnus Løberg, View ORCID ProfileMichael Bretthauer, View ORCID ProfileMette K
James Todaro, MD: 8/10/20 There is growing evidence that T-cell immunity allows populations to reach herd immunity once only 10-20% are infected with SARS-CoV-2. This would explain why a highly transmissible virus in densely populated areas peaked at 10-20% infected regardless of lockdowns or masks. The pervasive misconception is that we have zero immunity against COVID-19. Based on this flawed understanding, epidemiologists projected that herd immunity is not reached until 60-70% are infected.This is almost certainly wrong.
Of course, the media ignores this research3/. While antibodies against COVID-19 may only last months, T cell immunity can remain protective for years. In a study of 23 people who survived SARS in 2003, every single one had memory T cells that recognized the SARS virus 17 years later. (Nature) https://www.nature.com/articles/s41586-020-2550-z …4/ Moreover, blood samples from all 23 individuals showed “robust cross-reactivity” against SARS-CoV-2. This can be called crossover immunity. Crossover immunity is not limited to just people who were infected with SARS years ago though.5/ In the same study, in 37 persons with no history of SARS or COVID-19 (negative serology and/or samples taken before COVID-19), over 50% had SARS-CoV-2 specific T cells. This is not surprising because there are at least 4 strains of coronaviruses that cause the “common cold”.6/
The above study is not the only one to show this level of cross-reactivity.In a study from April 2020, in 68 healthy donors never exposed to COVID-19, 34% had T cells that reacted to SARS-CoV-2. https://www.medrxiv.org/content/10.1101/2020.04.17.20061440v1 …7/ This finding was confirmed in yet another study published in Cell in June 2020 showing that 40-60% of unexposed individuals had T cell recognition of SARS-CoV-2. The authors hypothesized that crossover immunity came from “common cold” coronaviruses. https://www.sciencedirect.com/science/article/pii/S0092867420306103?via%3Dihub …8/ Crossover immunity may explain why so many young and middle-aged individuals are asymptomatic even when testing positive for coronavirus. It is likely that their T cells recognized the virus and mounted an immune response before even mild symptoms surfaced.9/ What does this mean? All those runny noses from the common cold prepared our T cells to fight COVID-19.Although it has been ominously called the “novel-coronavirus”, SARS-
CoV-2 is yet another coronavirus with many similarities in structure to the common cold coronaviruses.10/ Why are the elderly hit so hard by COVID-19 though? Indeed the strain of coronavirus that we faced in 2020 is more lethal than those in the past, specifically in the elderly and immunocompromised…11/ …With an understanding of T cell immunity, it makes sense that the elderly are more affected by COVID-19.It is well known that persons in advanced age and/or who are immunocompromised lose T cells https://medicalxpress.com/news/2020-06-cell-immunity-elderly.html …12/ Let’s get back to herd immunity via T cells. If ~50% of people had T cell immunity prior to SARS-CoV-2, then that leaves 50% of the population susceptible.T helper cells seem to be the longest lasting form of CoV2-19 immunity.
In the regions hit hardest by COVID-19, serology studies show new cases and deaths peaked at around 10-20% infected.13/ Adding the 50% who already had T cell immunity from common cold viruses to the newly infected 10-20% equals about 60-70% immunity. Not coincidentally, 60-70% is the percentage epidemiologists project is necessary for herd immunity with a respiratory virus.14/ It is likely that many of the hardest hit regions of the world (e.g. Lombardy, NYC, Madrid, London, Stockholm) are now at herd immunity. Lock-downs and mask ordinances (mostly coming after the peak) likely had little effect, with the exception of perhaps prolonging the spread.15/ Sweden is an example of what herd immunity looks like without lockdowns or masks. Based on serology testing, ~20% of Stockholm was infected by April. Deaths peaked in Sweden in April. Today, the pandemic is over in Sweden with zero deaths per day & subsiding new infections16/ Lockdown advocates will challenge this thesis and point to Indian slums and areas in Peru that reached much higher infection prevalences.However, malnourishment is rampant in these very poor regions…And it is well known that T cell function is reduced in the malnourished.17/ This research on T cell immunity is largely being ignored by the mainstream media, possibly due to political and pharmaceutical interests. Hint: assuming $35 per vaccine dose (Moderna’s price), vaccinating just the USA alone will result in a revenue of ~ $10 billion annually. Considering that the coronavirus vaccine industry has the potential to be the biggest profit maker big pharma has ever seen, it is not surprising that we are seeing an overly aggressive push for lockdowns and masks until there is a vaccine—no matter the cost. Special thanks to the efforts of @dockaurG, @ProfKarolSikora, @FatEmperor, Dr. Beda Stadler (former director of the Institute for Immunology at the University of Bern) and many others who have been talking about T cell immunity for quite some time now. (Source: https://threader.app/thread/1292873236716433416)
Tyler Durden 7/11/20: “Stanford’s Nobel-laureate Dr. Michael Levitt on 5/4/20 was interviewed by the Stanford Daily: he advocated for Sweden’s approach of letting COVID-19 spread naturally through the community until you arrive at HIT (herd immunity). Levitt stated ‘If the Sweden death frequency stops at about 5,000 or 6,000 deaths, we will know that they’ve reached herd immunity, and we didn’t need to do any kind of lockdown.’ ” Statistics are showing this to be true presently.
Durden again: “Sweden appears to have reached HERD IMMUNITY at 10-20% infected, instead of the touted necessity of 60-70% to reach herd immunity. Apparently, many have had exposure to previous corona virus infections/or may have partial native T-CELL immunity and thus are NOT susceptible to infection: neutralizing antibodies are from B-cells. Medical Xpress 7/16/20 Resistant or Immune T-Cells from recovered SARS CoV2-19 patients discussion:
https://medicalxpress.com/ns ews/2020-07-scientists-uncover-sars-cov-specific-cell-immunity.html?utm_source=nwletter&utm_medium=email&utm_campaign=daily-nwletter
Up to 81% can mount a strong response to CoV2-19 without ever having been exposed to it before. On 7/8/20, USA new cases rose to 62,000 in one day while deaths are down 75% from the April peak. This shouldn’t come as a surprise because the pattern has been the same as in countries around the world. The trajectory of infections was mapped out long ago by UK epidemiologist and statistician, William Farr. “Farr shows us that once peak infection has been reached then it will roughly follow the same symmetrical pattern on the downward slope. However, under testing and variations in testing regimes means we have no way of knowing when the peak of infections occurred. In this situation, we should use the data on deaths to predict the peak. There is a predicted time lag from infection to COVID deaths of approximately 21 to 28 days.” In an Icelandic study, children under 10 apparently seldom become infected or transmit the infection: in fact, there was no documented case of transmission to parents as reported in the National Review.
The 7/2020 Federal decision to have testing reports sent to the the Department of Health and Human Services (DHS) instead of the CDC appears correct: “When told how the CDC chose to lump the results of both serum and nasal-oral tests together to determine CoV2-10 freqeuncy, director of the Harvard’s Global Health Institute, Ashish Jha, MD, told The Atlantic, “You’ve got to be kidding me … How could the CDC make that mistake? This is a mess.”
Multiple tests MAY be necessary to prove if a patient is infected. On 3/30/20 there was a report from Wuhan, China, of CoV2-19 corona virus reinfection. This is now also true of Singapore and South Korea, even as returnees also increase the number of cases. BUT, there is no actual proof of reinfection.
As reported by Anthony Fauci, MD, Chief of the National Institutes of Health (NIH) Infectious Disease section, the current influenza virus is 0.1% lethal and this CoV2-19 virus is 3.4% lethal, although in Germany it was found to be 0.37% lethal in the symptomatic; while not 3.4% lethal, still it is 4 times more lethal than the seasonal flu, AND in a more CONDENSED period of time.
0-49 years old: 0.05%
50-64 years old: 0.2%
65+ years old: 1.3%
From Heather MacDonald in The Spectator: “…Neil Ferguson, Director of the Imperial College model that triggered lockdowns in Great Britain and the US, has conceded that as many as two-thirds of all people who die of coronavirus in 2020 would have died by the end of the year anyway.
Middle-aged and the young are at minimal risk from the coronavirus. The median age of coronavirus death in most countries was 80, now 35! Political analyst Phil Kerpen had found that Pennsylvania has more COVID-19 deaths among people over 100 than among people under age 45, more deaths over age 95 than under age 60, and more deaths over 85 than under 80. An analysis of Spanish data found that the fatality rate for the infected was 0.052 % for people under 60 — half of that for the seasonal flu. The typical coronavirus case is asymptomatic, and appears to have no lasting effect on the sufferer…” As of 10/14/20 a report states that 45% of Cov2-19 cases are asymptomatic and those are associated with high levels of beneficial lymphocytes.
Jon Barron offers this information: “the mortality rate is as low is 2.1% in Turkey and 2.5% in Germany. But it’s as high as 12.8% in Belgium (due to better tracking in Belgium) and Italy. The US mortality rate is 4.1 -> 1.3 %, and the global average is disturbingly high at 6%.” 90% of those admitted have underlying chronic health conditions. In the USA, ONLY 5% will need the ICU. 20% of those hospitalized will go to the ICU & 20% of those who go to the ICU will die. 20% of ICU admissions last 28 days & 10% last 42 days. Michael Greger, MD: statistics “from South Korea: of confirmed cases, about 1 in 1,000 died in their 30-40s, 1 in 150 of those in their 50s, 1 in 50 in their 60s, 1 in 15 in their 70s, and 1 in 5 in their 80s. Harlan Krumholz of Yale University said “Its ferocity is breathtaking and humbling.”
It has recently been estimated that only 6% of cases have been identified (Lancet Infectious Diseases 2020. DOI: 10.1016/S1473-3099(20)30243-7). This means there are already 30-50 million infected in the USA. The true death rate is unknown because the actual number of infected is based on those tested and MANY more who have this infection but are not yet identified as they have not been tested and found. In 80% of the infected it will be a mild cold or flu. 25% are asymptomatic carriers and likely infectious. “More than half of COVID-19 transmission is driven by people who show no symptoms, according to research published in the 7/6/20 Proceedings of the National Academy of Sciences.” 15-20% of those admitted to the hospital will be sent to the ICU/intensive care unit, 15% of those admitted to the hospital will require invasive mechanical ventilation: 40% on ventilators will die. 20% of those admitted to the hospital died. CONTINUED below starting at “Average duration…”
The Covid Tracking Project data as of 11/8/20
Covid-19 is at least three rimes as lethal as the current influenza.
@COVID19Tracking
Our daily update is published. States reported 1.1 million tests, 103k cases, and 57k people currently hospitalized with COVID-19. The death toll was 462.
Blood test could identify COVID-19 patients at risk of cytokine storm
by University of Southampton 10/14/20
High levels of cytokines IL-6, IL-8, TNF, IL-1β and IL-33 in the patients’ blood on admission were associated with greater chance of needing intensive care, artificial ventilation and of dying. IL-1β and IL-33 showed the biggest effect.
Average duration of illness onset for those who died was 18-28 days. While it can affect the young adversely and lethally, it is most severe in those over 65, those with high blood pressure, overweight, diabetes, high population density living- inter- or multigenerational living together, a higher score on the Charlson especially Comorbidity Index, elevated respiratory rate of 24, elevated levels of venous lactate, creatinine, procalcitonin, LDH, low platelet or lymphocyte counts, asthma, heart disease, exposure to air pollution, immune immobilization, etc. Recent identification of “counties” at risk for developing the corona virus disease includes those that are poor and rural, have low education status, high housing debt, and where there is inadequate sleep. Follow the latest news on the coronavirus (COVID-19) outbreak
More information: The entire high-risk county sets analysis can be viewed in more detail on this website: akaikaeru.com/covid-19-1
JAMA 8/24/20 S T Nyberg “Weight management….” : “In the UK Biobank of 387,109 men and women, mutually adjusted hazard ratio for COVID-19 hospitalizations was 25% to 30% lower in those who never smoked compared with ex-smokers and current smokers, 25% lower for physically active compared with physically inactive people, and 50% lower in individuals with a healthy weight compared with those with obesity.”
The IMHE (Institute for Health Metrics and Evaluation) model that advises the Federal government on the corona virus has been remarkably incorrect. This model predicted that over 121,000 Americans would be hospitalized by 4/1/20: the actual number was 31,142. The scariest predictions of lethality have NOT come to pass which is in part to social distancing and quarantining (quarantine originally meant 40 days of isolation) + effective treatment.
“The 50-59 age group had a 0.1% fatality rate, the level often cited as the overall death rate for the seasonal flu. Those are all lower odds than an individual has of dying in a giving year of any cause and in the case of an average 50-year-old, five times lower. Children under 20 were not tested, but their fatality rate is likely near zero.
While the Netherlands is an entirely different country, it has actually experienced a 30% higher death rate per capita than America. So the numbers are likely not any higher here for those under 70, especially because the macro serology tests showing a 0.2% fatality rate (but grossly distorted by the death rate of those over 80). Data from prisons and ships in younger populations harmonize with this data. A report from France shows very similar estimates of fatality rates, at least for those under 60.”
From Wired online Magazine by G M Graff : “An Oral History of the Day Everything Changed, 3/11/20” “… History will record that Wednesday, March 11, the 71st day of 2020, proved to be unlike any other in American history—the pivot point on which weeks of winter unease about the looming novel coronavirus turned in a matter of hours into a sudden, wrenching, nation-altering halt to daily life and routine. Just a day earlier, Americans across much of the country were still going into the office, meeting friends for drinks, and shaking hands in meetings. That morning, the number of coronavirus cases in the US crossed the 1,000 mark, up 10-fold from the prior week. Only 29 Americans had died.
But on that Wednesday, the World Health Organization, which had only begun referring to the virus as Covid-19 a month earlier, declared the disease a global pandemic. Every hour seemed to bring major new developments: On Wall Street, after days of huge up-and-down gyrations, the Dow Jones Industrial Average fell 1,465 points and officially entered bear territory; Capitol Hill faced its first confirmed Covid-19 case; the NCAA announced it would play its basketball tournament without fans; and then, in rapid-fire succession that evening, President Trump gave an Oval Office address, announcing a travel ban from Europe, the NBA suspended its season after player Rudy Gobert tested positive for the virus, and Tom Hanks and his wife, Rita, posted on Instagram that they too had been diagnosed with Co V2-19 while in Australia and were recuperating….”
TRENDING: “This is the Beginning of the End of the Pandemic” – Dr. Stephen Smith Announces Hydroxy-Chloroquine Study that is “Game Changer” in Battle Against Coronavirus (VIDEO)
See also the more optimistic 3/25/20 Wall Street Journal article by Bendavid and Bhattacharya.
B. Hume: ““I think it’s time to consider the possibility… that this lockdown, as opposed to the more moderate mitigation efforts… is a colossal public policy calamity.” Economist Scott Grannis observed 4/12/2020: “Almost overnight, we have wiped out all the net job gains of the past 14 years.” Grannis bluntly concluded that, “The shutdown of the U.S. economy will prove to be the most expensive self-inflicted injury in the history of mankind.”
N J Kaster 4/25/20: “Despite their air of authority, the experts never had enough knowledge about this virus to make reliable calculations about the future. But the real problem with the models weren’t that they proved to be false, but rather that they were promoted with false certitude.” “I confess that I prefer true but imperfect knowledge,” economist Friedrich Hayek once said, “to a pretense of exact knowledge that is likely (proved later to be) to be false.”
Hayek’s remark, given as he was accepting the Nobel Prize in 1974, was that thinking of economics as a “science” might lead to “a pretense of knowledge,” the idea that any one person might know enough to engineer society successfully, unmindful of unintended consequences.
“There is danger in the exuberant feeling of ever-growing power which the advance of the physical sciences has engendered and which tempts man to try, ‘dizzy with success’… to subject not only our natural but also our human environment to the control of a human will. The recognition of the insuperable limits to his knowledge ought indeed to teach the student of society a lesson of humility which should guard him against becoming an accomplice in men’s fatal striving to control society–a striving which makes him not only a tyrant over his fellows, but which may well make him the destroyer of a civilization which no brain has designed but which has grown from the free efforts of millions of individuals.”
In contrast to the USA, this is essentially the approach Sweden has chosen. In an article in the UK Spectator, Fredrik Erixon, the director of the European Centre for International Political Economy in Brussel, explained that about Covid-19. Many people work from home. Restaurants are open, but not bustling. Keeping two metres apart at bus stops is something Swedes were pretty good at before the crisis: we don’t need much encouragement now. We’re careful. But our approach to fighting the pandemic starts from something more fundamental: in a liberal democracy you have to convince and not command people into action. If you lose that principle, you will lose your soul.”
So far, the Swedish strategy of allowing some exposure to the virus in order to build immunity among the general population while protecting high-risk groups like the elderly appears to be paying off. The country’s chief epidemiologist reported “herd immunity” could be reached in the capital of Stockholm in a matter of weeks. Moreover, Sweden has achieved this while taking a less economic hit than other countries in Europe. Sweden’s approach was a mixture of epidemiology and principle. Erixon noted that the concept of a national lockdown is “deeply illiberal — and, until now, untested.” He allowed that Sweden may change if facts warrant. “But,” he wrote, “the vast majority, for now, want Sweden to keep its cool. We don’t want to remember 2020 as the time when we caused irreparable harm to our liberties — or lost them entirely” Kaster finished.
Medical XPress 7/6/20: “New research from Karolinska Institute and Karolinska University Hospital shows that many people with mild or asymptomatic COVID-19 demonstrate so-called T-cell-mediated immunity to the new coronavirus, even if they have not tested positively for antibodies. According to the researchers, this means that public immunity is probably higher than antibody tests suggest. The article is freely available on the bioRxiv server and has been submitted for publication in a scientific journal.”
T cells take the lead in controlling SARS-CoV-2 and reducing COVID-19 disease severity
MedicalXpress 9/17/20
Ever since SARS-CoV-2 first appeared, researchers have been trying to understand whether sometimes the immune system does more harm than good during the acute phase of COVID-19. The latest study by researchers at La Jolla Institute for Immunology clearly argues in favor of the immune system.
Their work, published in the 9/16/20, online issue of Cell, confirms that a multi-layered, virus-specific immune response is important for controlling the virus during the acute phase of the infection and reducing COVID-19 disease severity, with the bulk of the evidence pointing to a much bigger role for T-cells than antibodies. A weak or uncoordinated immune response, on the other hand, predicts a poor disease outcome. The findings suggest that vaccine candidates should aim to elicit a broad immune response that include antibodies, helper and killer T cells to ensure protective immunity. 10/2020: an entirely NEW approach that generates antibodies and T-CELL activation is that of Dr. Partick Soon-Shiong’s Immunity Bio.
“Our observations could also explain why older COVID-19 patients are much more vulnerable to the disease,” says senior author Shane Crotty, Ph.D., who co-led the study with Alessandro Sette, Dr. Biol.Sci., both professors in LJI’s Center for Infectious Disease and Vaccine Research. “With increasing age, the reservoir of T cells that can be activated against a specific virus declines and the body’s immune response becomes less coordinated, which looks to be one factor making older people drastically more susceptible to severe or fatal COVID-19.”
Adds Sette, “What we didn’t see was any evidence that T cells contribute to a cytokine storm, which is more likely mediated by the innate immune system.”
When SARS-CoV-2 or any other virus infiltrates the body, the innate immune system is first on the scene and launches a broad and unspecific attack against the intruder. It releases waves of signaling molecules that incite inflammation and alert the immune system’s precision forces to the presence of a pathogen.
Within days, the so-called adaptive immune system tools up and moves with pinpoint precision against the virus, intercepting viral particles and killing infected cells.
The adaptive immune system consists of three branches: antibodies; helper T cells (Th), which assist B cell to make protective antibodies; and killer T cells (CTL), which seek out virus-infected cells and eliminate them. Co-first author Sydney Ramirez, MD/PhD spearheaded the sample collection.
What the team found was that similar to their previous study all fully recovered individuals had measurable antibody, helper and killer T-cell responses, while the adaptive immune response in acute COVID-19 patients varied more widely with some lacking neutralizing antibodies, others helper or killer T cells or any combination thereof.
“When we looked at a combination of all of our data across all 111 measured parameters we found that in general, people who mounted a broader and well-coordinated adaptive response tended to do better. A strong SARS-CoV-2 specific T cell response, in particular, was predictive of milder disease,” says co-first author Carolyn Moderbacher, PhD, “Individuals whose immune response was less coordinated tended to have poorer outcomes.”
The effect was magnified when the researchers broke down the dataset by age. “People over the age of 65 were much more likely to have poor T cell responses, and a poorly coordinated immune response, and thus have much more severe or fatal COVID-19,” says Crotty. “Thus, part of the massive susceptibility of the elderly to COVID-19 appears to be a weak adaptive immune response, which may be because of fewer naïve T cells in the elderly.”
Naïve T cells are inexperienced T cells that have not met their viral match yet and are waiting to be called up. As we age, the immune system’s supply of deployable naïve T cells dwindles.
In line with what other research teams had found before, antibodies don’t seem to play an important role in controlling acute COVID-19. Instead, T cells and helper T cells in particular are associated with protective immune responses. “This was perplexing to many people,” says Crotty, “but controlling a primary infection is not the same as vaccine-induced immunity, where the adaptive immune system is ready to pounce at time zero.”
If a vaccination is successful, vaccine-induced antibodies are ready to intercept the virus when it shows up at the doorstep. In contrast, in a normal infection the virus gets a head start because the immune system has never seen anything like it. By the time the adaptive immune system is ready to go during a primary infection, the virus has already replicated inside cells and antibodies can’t get to it.
“Thus, these findings indicate it is plausible T cells are more important in natural SARS-CoV-2 infection, and antibodies more important in a COVID-19 vaccine,” says Crotty, “although it is also plausible that T cell responses against this virus are important in both cases.”
7/6/20: Oxford epidemiologist pushes herd immunity by JACQUELIN MAGNAY
“One of the world’s top epidemiologists has urged Australia to abandon its lockdown strategy against coronavirus and look to the Swedish model of developing herd immunity. Dr. Sunetra Gupta, professor of theoretical epidemiology in the Department of Zoology at the University of Oxford, says Australia is adopting a “selfish’’ and “self-congratulatory’’ approach which is misguided and will have negative long-term consequences and urged the country to look at the latest evidence to decide its tactics. (HRS comments: She supported the Swedish method.)
She said if the Australian government changed its approach and let the virus, which 80-90 % of the population will only get it asymptomatically — spread naturally, with intense protections for those most vulnerable, it would in the long term help protect all of Australians from future viral threats and also avoid the most damaging short-term economic impacts for the underprivileged.
The most recent scientific research shows that 30-81 % has natural immunity to coronavirus because the body’s T-cells recognize the threat from having had other cold and flu viruses. Scientists believe having coronavirus causes people’s immune systems to develop antibodies and T-cell responses to future viruses. She warned that suppression of the virus did not work and lockdown simply resulted in some parts of the population being more exposed to the virus when it next flared up.
She said: “There is no way lockdown can eliminate the virus … and so it’s not at all surprising once you lift lockdown in areas it will flare up again, (HRS now inserts, unless there are effective treatments as now exists and/or vaccines). That is what we are seeing in the southern United States, and in Australia. In places where it has already swept through, a proportion of people are immune and you are not seeing it come back.’’
Newsletter X Science 5/22/20: Despite never declaring a general lockdown, Uruguay had recorded 749 cases and 20 deaths by Thursday among a population of 3.4 million. In Costa Rica (https://qcostarica.com/hydroxychloroquine-the-drug-costa-rica-uses-successfully-to-fight-covid-19/) there have been just 903 cases and 10 deaths in a country of five million. The numbers don’t lie, and the outbreak in Uruguay “is currently under control,” said epidemiologist Julio Vignolo, citing the country’s rapid response.
By David Leonhardt NYT 5/15/20
Almost every country across Europe and North America put in place a kind of lockdown. Not every country has experienced the same sharp increase in unemployment the USA has.
3 million more Americans filed for jobless benefits last week. The total over the past two months is now 36.5 million. See the chart:
The U.S. has more joblessness than other countries.
This article was reviewed for me by an economist who said that the statistics are quite misleading due to different payment techniques affecting the definition of “employment”. The countries with the smallest increases in unemployment put in place programs that directly pay companies to retain their workers.
Australia, Denmark and New Zealand created new programs. France and Germany expanded existing programs. All maintained the connection between employer and employee as much of the economy is temporarily shut down.
The United States took a different approach. The $2 trillion stimulus program passed in March did include a version of the approach other countries are taking: $350 billion Paycheck Protection Program quickly finished with high demand.
Top Israeli Mathematician Claims COVID-19 Peaks After 40 Days With or Without Economic Lockdown: leading doctor dismisses his claims
By Times of Israel 4/14/2020
A prominent Israeli mathematician, analyst and former general claims simple statistical analysis, Isaac Ben-Israel, demonstrates that the spread of COVID-19 peaks after about 40 days and declines to almost zero after 70 days — no matter where it strikes, and no matter what measures governments impose to try to thwart it. Prof Isaac Ben-Israel (I B-I), head of the Security Studies program in Tel Aviv University and the chairman of the National Council for Research and Development said that research he conducted with a fellow professor, analyzing the growth and decline of new cases in countries around the world, showed repeatedly that “there’s a set pattern” and “the numbers speak for themselves.”
While he said he supports social distancing, the widespread shuttering of economies worldwide constitutes a demonstrable error in light of those statistics. In Israel’s case, he noted, about 140 people normally die every day. To have shuttered much of the economy because of a virus that is killing one or two a day is a radical error that is unnecessarily costing Israel 20% of its GDP, he charged. I B-I stated that the closure policies of the quarantine countries can be replaced by more moderate social distancing policies. “The numbers simply do not support quarantine or economic closure.”
Proper quarantine is, however, reviewed alternatively in this Beijing, China, study: “Starting mass quarantine and case isolation earlier greatly improves the effectiveness of disease suppression and also demands fewer health care resources,” Dr. Baoguo said.
Another important finding was that quarantine must be implemented at a rate of at least 50 percent, and a maximum of 10 weeks after the start of a COVID-19 outbreak to have a chance at reversing the infection rate. If less that 40 percent of the population remains quarantined or the quarantine begins more than 11 weeks after onset of the outbreak, it is too late.”
More information: Tianbing Wang et al, A four-compartment model for the COVID-19 infection—implications on infection kinetics, control measures, and lockdown exit strategies, Precision Clinical Medicine (2020). DOI: 10.1093/pcmedi/pbaa018
Medical Xpress 11/23/20 “…Knowing that there were no safe or proven treatments or an effective vaccine, China relied on proven nonpharmaceutical interventions to conquer the epidemic. First and foremost was containing the virus through controlling the sources of infection and blocking transmission. This was accomplished through early detection (testing), isolation, treatment and tracing the close contacts of any infected individual.
This strategy was aided by the three field hospitals (fancang) the government built to isolate patients with mild to moderate symptoms from their families. Strict quarantine measures were also central to preventing the spread of this epidemic. This was paired with compulsory mask-wearing, promotion of personal hygiene (hand-washing, home disinfection, ventilation), self-monitoring of body temperature, universal compulsory stay-at-home orders for all residents, and universal symptom surveys conducted by community workers and volunteers.”
On the reasonableness of Israel’s unprecedented quarantine and closure, he commented to the news agency, “I think it’s mass hysteria. I have no other way to describe it.” Prof. Gabi Barbash, a hospital director and the former Health Ministry director general, insisted in an exchange that Ben-Israel is mistaken, and that the death tolls would have been far higher if Israel and other countries had not taken the steps they did.
But Ben-Israel said the figures — notably from countries, such as Singapore, Taiwan, and Sweden, which did not take such radical measures to shutter their economies — proved his point. (He posted a paper in Hebrew to this effect on Facebook, with graphs showing the trajectories). When Barbash cited New York as ostensible proof that Ben-Israel was mistaken, I B-I noted the latest indications from New York were precisely in line with his statistics that indicate daily new cases figures peaking and starting to fall after about 40 days.
Asked to explain the phenomenon, I B-I, who also heads Israel’s Space Agency, later said: “I have no explanation. There are all kinds of speculations. Maybe it’s related to climate, or the virus has a life-span of its own.” He said the policy of lockdowns and closures was a case of “mass hysteria.” Simple social distancing would be sufficient, he said. Stanford University infectious disease epidemiologist of note, John Ionnadis stated his “view of lockdown” as a “drug with dangerous side effects when its use is prolonged. It’s an extreme measure — a last resort, the nuclear option.”
If the lockdowns instituted in Israel and elsewhere were not causing such immense economic havoc, there wouldn’t be a problem with them, he said. “But you shouldn’t be closing down the entire country when most of the population is not at high risk.” Asked to explain why the virus had caused such a high death toll in countries such as Italy, he said the Italian health service was already overwhelmed. “It collapsed in 2017 because of the flu,” he said. Barbash, speaking after Ben-Israel had left the studio, insisted that “we’re going to be living with the coronavirus for the next year.”
The six foot rule: ”…if the six-foot rule is not arbitrary, why does the World Health Organization suggest a three-foot distance and why Austria, Norway, Sweden, and Finland have adopted that 3 foot rule, and why Germany and other countries use a 4.5-foot rule. Does the coronavirus behave differently in Europe?”
“Close to 60% of ALL infections in the US are within a 350 mile radius of NYC. The majority of the remaining US infections are also concentrated in urban metro areas like Detroit, New Orleans, Philadelphia, Atlanta etc. In New Jersey, its seven “commuter” counties closest to NYC contain 75% of the state’s positive infections. The majority of deaths are people with:
pre-existing medical conditions and/or who are over 65 and/or who are living in nursing, dementia care, or assisted living facilities and/or are addicted to drugs/alcohol. The county infection data confirms this is primarily an urban/metro area pandemic: very few healthy addiction-free people under 65 are dying from Covid-19 infection.”
10/3/21: A CRUCIAL YOUTUBE video ABOUT COUNTRIES REOPEING COMPLETELY TO NORMALCY AND “LIVING WITH” THE VIRUS (as best possible) https://www.youtube.com/watch?v=_ItkYhFiGB
REOPENING in GERMANY, NORWAY, the CZECH Republic and DENMARK all lifted some restrictions 4/20/20: figures published by German disease control agency Robert Koch Institute 4/16/20 stated that the person-to-person infection rate has dropped to 0.7. Shops up to 800 square meters (8,600 square feet) will be allowed to reopen if they uphold hygiene rules, Chancellor Merkel said 4/15/20. Schools reopened 5/4/20 in Germany with priority given to pupils taking exams soon. Rules will remain in force preventing groups of more than two people from gathering in public, other than family groups who live together, while large public events remain banned until 8/31/20.
HONG KONG (HK)MODEL: “testing, contact tracing, and population behavioral change were far less disruptive socially and economically than total lockdown. HK averted a major COVID-19 outbreak up to 3/31/20, by adopting far less drastic control measures than most other countries using a combination of border entry restrictions, quarantine and isolation of cases and contacts, together with some degree of social distancing, as reported in The Lancet Public Health journal. As of 3/31/20, HK had 715 confirmed COVID-19 cases including 94 asymptomatic infections, and 4 deaths in a population of about 7.5 million.”
Advance Care Planning and “The Love Song of J. Alfred Prufrock”
Daniel P. Sulmasy, MD, PhD1 JAMA Intern Med. Online April 13, 2020 doi:10.1001/jamainternmed.2020.0796 “…T. S. Eliot’s poem, “The Love Song of J. Alfred Prufrock”7 (eAppendix in the Supplement), first published in 1915, considers the need to act under uncertainty and in the face of our certain mortality. The poem can help us to understand why personal and cultural transformation are more important than legal documents, planning, scripted conversations, or AI….”
SYMPTOMS initially are fever (50% in the beginning and later 90%), dry cough, mild shortness of breath, malaise, headache, reddish eyes (conjunctivitis), 30% will have loss of the sense of SMELL and TASTE, but much less of runny nose, diarrhea, or vomiting. X-Ray/thin slice CT SCAN findings show “ground glass” bilaterally in both lungs, no pneumothorax, effusions or lymphadenopathy. 80% of non-severe cases have normal chest X-rays or CT scans. Chest ULTRAOUND can also visualize and follow the course of the CoV2-19 pneumonia. Incubation is 2 to 11 days, for an average of five days; that is, symptoms develop on average 5 days after exposure, infectivity tends to last 14 days after symptoms develop. Reports of NEUROMUSCULAR complications are an axonal peripheral neuropathy (nerve toxicity) or a myopathy (muscle toxicity) with elevated blood creatinine kinase muscle enzyme. Pathology showed widespread VASCULITIS and disseminated clotting in many organs, including striated muscle. These clinical features might be part of the corona virus infection more than just nonspecific complications of any severe illness. There was a report of a patient with olfactory neuropathy (disorder of smell).
Five of 206 patients in Singapore developed large-vessel strokes. Four of these patients had their strokes in the setting of critical illness and 3 were associated with hypotension/low blood pressure. The elevated cardiac blood tests SuPAR (soluble urokinase plasminogen activated receptor) is a sign of immune activation, ferritin, LDH, procalcitonin, D-dimer (reference range <500 ng/mL), direct bilirubin, CRP, and 8/22/20 Triad of molecules could predict severity of COVID-19
Troponin strongly predicted mortality; interleukin-13 is especially predictive of mortality. 6/25/20 The Lawson Research Institute found that six molecules were uniquely elevated in COVID-19 ICU patients (tumor necrosis factor, granzyme B, heat shock protein 70, interleukin-18, interferon-gamma-inducible protein 10 and elastase 2). The team also used AI to validate their results. They found that inflammation profiling was able to predict the presence of COVID-19 in critically ill patients with 98 percent accuracy. They also found that one of the molecules (heat shock protein 70) was strongly associated with an increased risk of death when measured in the blood early during the illness. Douglas D. Fraser et al, Inflammation Profiling of Critically Ill Coronavirus Disease 2019 Patients , Critical Care Explorations (2020). DOI: 10.1097/CCE.0000000000000144
MedicalXpress 8/2020 Researchers at the Francis Crick Institute, King’s College London and Guy’s and St Thomas’ NHS Foundation Trust, published in Nature Medicine that a common immune signature in patients with COVID-19 can be used to predict how severely ill a patient will become. The team analyzed 63 patients and identified 3 results that indicate how the disease will progress: the “triad” is IP-10, interleukin-10 and interleukin-6.
Reuters Health Medical News 5/15/20: low T-cell subset counts, especially of CD4+ and CD8+ T cells, are associated with more severe illness in CoV2-19 patients, especially CD3+, CD4+, CD8+ T cells, and natural killer (NK) cells. B-cell counts did not differ significantly from those in the control group. Severe CoV2-19 patients had significantly lower CD3+, CD4+, and CD8+ T-cell count: Journal of Infectious Diseases by Dr. Wan et al. CD3+, CD4+, and CD8+ T-cell counts recovered dramatically whose SARS-CoV-2 nucleic acid tests turned negative but did not change in patients with persistently positive tests. NK and B-cell counts did not change significantly. CD8+ T-cell counts best discriminated between COVID-19 patients and healthy controls, whereas CD4+ T-cell counts were slightly more accurate for differentiating between patients with severe illness and patients with mild-to-moderate illness.
Scientists discover ‘immune scars’ on patients with lung infections
by Patrick Galey 5/18/20 Newsletter Science X. “Studies show that the body’s immune response is temporarily switched off after some severe infections. Patients recovering from severe lung infections develop “immunological scars” that stifle the body’s immune response and then heighten their risk of contracting pneumonia, a common killer of COVID-19 patients. Cells that form the immune system’s first line of defense—macrophages (raise an internal alarm that sends immune cells rushing to the site of infection)—were “paralyzed” after severe infection. Antoine Roquilly, from the University Hospital of Nantes, also identified the trigger or “switch” for reanimating the macrophages, a receptor known as SIRP-alpha. Most COVID-19 deaths occur due to a cytokine storm—a process whereby the body’s own immune response runs wild causing acute and often fatal inflammation.”
• Onset to recovery is 12-32 days. Patients at high RISK are over age 65, have high blood pressure, a d-dimer blood test greater than 1000 ng or 1 ug/mL (reference range <500 ng/mL) implying the now documented diffuse intravascular & pulmonary clotting, and who have an adverse SOFA sepsis score. “Apixaban (Eliquis, a NOAC), given as prophylaxis or therapy in COVID patients, appeared beneficial for improving survival when D-dimer levels were 1-3 µg/mL. Full apixaban therapy was also tied to better survival when D-dimer levels exceeded 10 µg/mL.” One is SAFE 3 days after having no fever + resolved respiratory symptoms + improved chest CT scan + 2 negative PCR (molecular or nucleic acid) tests for the virus separated by 1 day. Viral shedding can occur for up to 37 days after onset of symptoms. Viral RNA can persist in the blood for up to 29 days and does not correlate with symptoms. It is (medically) believed that an ALKALINE cellular chemistry (“alkalinization of the phagolysosomes”) impedes the virus: that is thought to be a mechanism for how HYDROXYCHLORQUINE (HCQ) & AZITHROMYCIN (AZITH) work: increasing alkalinity inside the cell. The corona virus attaches to its ACE2 receptor(s) and then is internalized by microphagic vesicles, which eventually fuse with lysosomes, strip the viral genome from its envelopes and set it free. HCQ is a Toll-like receptor antagonist that inhibits the fusion of lysosomal and endosomal vacuoles which may be another mode of action of HCQ. See Mechanism of action of antimalarial drugs: inhibition of antigen processing and presentation. Fox RI, Kang HI. Lupus. 1993 Feb;2 Suppl 1:S9-12. PMID: 8097945 Review. Zinc sulphate is a part of that treatment regimen. HCQ was approved for use in 1955 by the FDA. Is
An analysis with too much theatrical language, but nonetheless discusses the incorrect/much TOO HIGH doses of HCQ chosen for studies by high prestige authors/institutions/high impact journals that spoke incorrectly (“designed to fail”) against the efficacy of HCQ: https://c-vine.com/blog/2020/08/07/faulty-hcq-research-based-on-death-by-toxic-overdosing/
Mefloquine: A promising drug ‘soldier’ in the battle against COVID-19
by Tokyo University of Science 7/1/21
Scientists propose the re-purposing of anti-malarial drug mefloquine to fight the novel coronavirus. Credit: Tokyo University of Science
Early 2020 saw the world break into what has been described as a “war-like situation”: A pandemic, caused by the severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2), the likes of which majority of the living generations across most of the planet have not ever seen. This pandemic has downed economies and resulted in hundreds of thousands of deaths. At the dawn of 2021, vaccines were deployed, but before populations can be sufficiently vaccinated, effective treatments remain as a pressing need.
Thus, other than fast-tracking research into novel drugs, scientists have also been exploring their arsenals of existing medicines in a bid to find anything that could work against COVID-19. Some approved drugs, like hydroxychloroquine, lopinavir, and interferon, have already been put to clinical use against SARS-CoV-2 without well establishing their clinical efficacies, due to the severity of the pandemic. Subsequent randomized trials have not been able to yield a consensus on the efficacy of these drugs. Only remdesivir has been approved for clinical use against severe COVID-19, although its efficacy is still being debated.
In a breakthrough study, a team of scientists—comprising Dr. Koichi Watashi, Kaho Shionoya, Masako Yamasaki, Dr. Hirofumi Ohashi, Dr. Shin Aoki, Dr. Kouji Kuramochi, and Dr. Tomohiro Tanaka from Tokyo University of Science (along with scientists from the National Institute of Infectious Diseases, Kyushu University, The University of Tokyo, Kyoto University, Japanese Foundation for Cancer Research, and Science Groove Inc.)—have identified an anti-malarial drug, mefloquine (which is incidentally a derivative of hydrochloroquine), that is effective against SARS-CoV-2. Their findings are published in Frontiers in Microbiology.
Detailing their modus operandi, lead scientist in the team Dr. Watashi says, “To identify drugs with higher antiviral potency than existing antivirals, we first screened approved anti-parasitic/anti-protozoal drugs. We found that mefloquine had the highest anti-SARS-CoV-2 activity among the tested compounds. Upon testing it against other quinoline derivatives, such as hydrochloroquine, in a cell line mimicking the cell-based environments of human lung cells, we found it to be better.”
The team further explored mefloquine’s mechanism of action. Dr. Watashi explains the process, “In our cell assays, mefloquine readily reduced the viral RNA levels when applied at the viral entry phase but showed no activity during virus-cell attachment. This shows that mefloquine is effective on SARS-COV-2 entry into cells after attachment on cell surface.”Thus, to bolster mefloquine’s anti-viral activity, the scientists looked into the possibility of combining it with a drug that inhibits the replication step of SARS-CoV-2: Nelfinavir. Interestingly, they observed that the two drugs acted in “synergy” and the drug combination showed greater anti-viral activity than either showed alone, without being toxic to the cells in the cell lines themselves.
The scientists also mathematically modeled the effectiveness of mefloquine to predict its potential real-world impact if applied to treat COVID-19. What they predicted was that mefloquine could reduce the overall viral load in affected patients to under 7% and shorten the “time-till-virus-elimination” by 6.1 days.
This study must be succeeded by clinical trials, but the world can hope that mefloquine becomes a drug used to effectively treat patients with COVID-19.
The dream team: Scientists find drug duo that may cure COVID-19 together
More information: Kaho Shionoya et al, Mefloquine, a Potent Anti-severe Acute Respiratory Syndrome-Related Coronavirus 2 (SARS-CoV-2) Drug as an Entry Inhibitor in vitro, Frontiers in Microbiology (2021). DOI: 10.3389/fmicb.2021.651403
Meta-analysis Suggests Hydroxychloroquine Benefit Outside Hospitals
By Reuters Staff 10/5/20
When data from five randomized controlled trials of hydroxychloroquine for COVID-19 prevention or treatment were pooled for a meta-analysis, researchers found that early use of the drug by people who were not hospitalized yielded a statistically significant 24% reduction in risk of infection, hospitalization or death.
In the individual trials, hydroxychloroquine did not show a statistically significant impact on prevention or treatment.
“The meta-analysis pools together the studies and increases statistical power,” said Dr. Joseph Ladapo of the David Geffen School of Medicine at UCLA, coauthor of a report posted on Wednesday on medRxiv ahead of peer review.
A weakness of the meta-analysis, Ladapo acknowledged, is that infections, hospitalizations and deaths were grouped together into a composite outcome. Combining all those events into one big number makes it more likely researchers will find that treatment had a significant effect.
Coauthor Dr. Harvey Risch of the Yale School of Public Health noted that seven nonrandomized controlled trials have also shown “statistically significant reduced risks with early outpatient use of hydroxychloroquine.” Along with the meta-analysis, he told Reuters, “This is extremely strong evidence of benefit.”
SOURCE: https://bit.ly/2SlHEeE medRxiv, online September 30, 2020.
Cite this: Meta-analysis Suggests Hydroxychloroquine Benefit Outside Hospitals – Medscape – Oct 02, 2020.
Full length article| Volume 101, P283-289, December 01, 2020
Early hydroxychloroquine but not chloroquine use reduces ICU admission in COVID-19 patients
• A.J.J. Lammers
• R.M. Brohet
• R.E.P. Theunissen
•
• H. Bax
• D.F. Postma
• P.H.P. Groeneveld
• Show all authors
Open AccessPublished:September 29, 2020DOI:https://doi.org/10.1016/j.ijid.2020.09.1460
• Following a global push for the use of hydroxychloroquine and chloroquine, there is ongoing discussion about the effectivity of these drugs.
• The findings of this observational study provide crucial data on a potential protective effect of hydroxychloroquine in non-ICU, hospitalized, COVID-19 patients.
• Early treatment with HCQ on the first day of admission is associated with a 53% reduction in risk of transfer to the ICU for mechanical ventilation.
• This protective effect was not observed for chloroquine; therefore, these drugs cannot be regarded as interchangeable.
Abstract
Background
The global push for the use of hydroxychloroquine (HCQ) and chloroquine (CQ) against COVID-19 has resulted in an ongoing discussion about the effectivity and toxicity of these drugs. Recent studies report no effect of (H)CQ on 28-day mortality. We investigated the effect of HCQ and CQ in hospitalized patients on the non-ICU COVID-ward.
Methods
A nationwide, observational cohort study was performed in The Netherlands. Hospitals were given the opportunity to decide independently on the use of three different COVID-19 treatment strategies: HCQ, CQ, or no treatment. We compared the outcomes between these groups. The primary outcomes were 1) death on the COVID-19 ward, and 2) transfer to the intensive care unit (ICU).
Results
The analysis included 1064 patients from 14 hospitals: 566 patients received treatment with either HCQ (n = 189) or CQ (n = 377), and 498 patients received no treatment. In a multivariate propensity-matched weighted competing regression analysis, there was no significant effect of (H)CQ on mortality on the COVID ward. However, HCQ was associated with a significantly decreased risk of transfer to the ICU (hazard ratio (HR) = 0.47, 95% CI = 0.27–0.82, p = 0.008) when compared with controls. This effect was not found in the CQ group (HR = 0.80, 95% CI = 0.55–1.15, p = 0.207), and remained significant after competing risk analysis.
Conclusion
The results of this observational study demonstrate a lack of effect of (H)CQ on non-ICU mortality. However, we show that the use of HCQ — but not CQ — is associated with a 53% reduction in risk of transfer of COVID-19 patients from the regular ward to the ICU. Recent prospective studies have reported on 28-day, all-cause mortality only; therefore, additional prospective data on the early effects of HCQ in preventing transfer to the ICU are still needed.
Introduction
After the emergence of SARS-CoV-2 in December 2019, the new coronavirus spread around the world, resulting in a pandemic. Unfortunately, there is still no proven effective drug or vaccine available against COVID-19, and hospitalized patients with COVID-19 are at high risk for admission to the ICU (10–20%), with 3–10% of patients requiring intubation, and 2–5% of patients dying (Guan et al., 2020a ). Among the drug candidates for treating COVID-19 are hydroxychloroquine (HCQ) and chloroquine (CQ) (Sanders et al., 2020 ). Insights into the underlying mechanisms of action of HCQ and CQ are still emerging. Both drugs have a large volume of distribution (Zhou et al., 2020, Schrezenmeier and Dörner, 2020.). Their molecular structures are comparable, except that HCQ has an extra hydroxyl group. Both interfere with lysosomal activity and decrease membrane stability, reduce signaling pathways for Toll-like-receptors 7 and 9, and impact on transcriptional activity, inhibiting cytokine production )Schrezenmeier and Dörner, 2020 ).
There are only a few differences between the drugs, of which the most important is drug clearance (Schrezenmeier and Dörner, 2020. Some observational studies on the efficacy of (H)CQ report clinical benefits and antiviral effects (Gao et al., 2020 Gautret et al., 2020, Arshad et al., 2020,Cortegiani et al., 2020), while others do not (Geleris et al., 2020, Mahevas et al., 2020). A few small, controlled trials have been inconclusive (Tang et al., 2020 , Chen et al., 2020). The Recovery study included 176 UK hospitals, comprising 1395 patients receiving high doses of HCQ (9200 mg cumulative dose), and reported no beneficial effects on all-cause mortality at 28 days (26.8% of treated patients versus 25% of controls) (Horby, 2020). The risk of admission to the ICU could not be calculated, since 17–60% of patients were already on (non-invasive) ventilation at randomization. A recent systematic review and meta-analysis, including 11 932 patients on HCQ, found that its use was not associated with reduced mortality (pooled relative risk of RCTs for HCQ use of 1.09) (Fiolet et al., 2020). Results of other prospective trials are not expected, since the European Discovery and the WHO Solidarity trials have discontinued their HCQ treatment arms because of lack of effect on mortality. Meanwhile, the US FDA and the Infectious Diseases Society of America (IDSA) advise against the use of (H)CQ outside the context of a clinical trial (Swank and McCarten, 2020, Infectious Diseases Society of America Guidelines, 2020 ). Based on the available evidence present at the start of the outbreak, a Dutch treatment guideline was developed (RIVM, 2020). Off-label use of both HCQ and CQ was offered as a treatment option; however, the guidelines did not endorse either treatment in particular. Consequently, hospitals decided independently on a treatment protocol with either HCQ or CQ, or to give no treatment. This policy created a unique situation for comparing the efficacy of HCQ and CQ with no treatment in hospitalized non-ICU patients, with a reduction of potential bias by indication.
Methods
Study design
The study was designed as an observational, multicenter, cohort study of hospitalized COVID-19 patients. Before the first patients were admitted, Dutch hospitals independently implemented a treatment protocol with or without (H)CQ. As a consequence, Dutch patients were geographically allocated to their local hospital with or without the intention to treat with (H)CQ. Eligible patients were included retrospectively over the period from February 28 to April 1, 2020. Patients were followed up until they reached one of the clinical endpoints: (1) discharge for cured infection to home or rehabilitation center; (2) transfer from the COVID ward to the intensive care unit (ICU); or (3) death, either during their hospital stay on the ward (non-ICU) or following transfer to a hospice facility. Secondary outcomes were the effects of the use of azithromycin (AZM) and angiotensin-receptor blockers (ARB) on outcome.
Participating hospitals
All hospitals in The Netherlands were considered eligible to participate in the study, including academic hospitals as well as non-teaching hospitals. These hospitals were asked to participate early in the outbreak. All participating hospitals shared their data with the coordinating hospital (Isala, Zwolle), where the statistical analysis was performed. Data-sharing agreements were signed, and the Medical Ethics Review Committee (METC) of Isala approved a waiver for informed consent.
Patients
Inclusion and exclusion criteria were designed to select a study sample of hospitalized patients with moderate to severe COVID-19. New confirmed COVID-19 cases were included if they were aged > 18 years and if they were admitted to the emergency department (ED) and subsequently hospitalized on the non-ICU hospital COVID-19 ward. Exclusion criteria were age < 18 years, admission to the ICU, or death within 24 h after presentation at the ED. Patients transferred between Dutch hospitals, for example due to capacity issues, were also excluded. Confirmed COVID-19 infection was defined as either positive SARS-CoV-2 real-time reverse transcriptase polymerase chain reaction (PCR) on swab material, sputum, or bronchoalveolar lavage samples (
Corman et al., 2020
), or typical findings on chest computed tomography (CT). Typical CT findings were defined as CO-RAD 4–5, using the CO-RAD classification system (COVID-19 Reporting and Data System, developed by the Dutch Radiology Society to describe levels of suspicion for COVID-19 infection) (
Prokop et al., 2020
). Routine blood tests were carried out for hematological and biochemical analysis, according to standard hospital laboratory techniques. Since the use of (H)CQ for COVID-19 was off-label, patients were started on (H)CQ only after giving informed consent.
Data collection
Data were extracted from Electronic Health Records (EHR) in all participating hospitals by medical students and/or infectious disease (ID) physicians. Data were collected on site using a standardized data-collection form on a secured website of the coordinating hospital. Patient data were immediately anonymized and encoded upon entry into the online research manager program. Collected data included patient characteristics, such as comorbidities, registered ICU-restrictive policy by treating physician, routine laboratory results, SARS-Cov2-PCR and chest CT-scan results, medical treatment before admission, and antibiotic treatment during hospitalization.
Statistical analysis
Differences between HCQ and CQ users (cases) and non-users (controls) were compared using χ2 statistics or the Fisher exact test for categorical variables, and the independent t-test or Mann-Whitney U test for continuous variables. The data were analyzed within a Cox proportional hazard regression framework. Follow-up commenced from the date of hospital admission and ended on the dates of death or ICU admission, and patients were censored at the time they were discharged from hospital. Hazard ratios were calculated for (H)CQ use in relation to the primary endpoints of death and ICU admission, or a combination of these endpoints denoted as a composite adverse endpoint. Death and ICU admission are competing risk events; therefore, competing risk regression analysis was conducted for these two endpoints according to the method developed by
Fine and Gray (1999)
. Instead of KM survival curves, survival data were summarized using the cumulative incidence function (CIF) or cumulative risks of an event, which indicate the probability of the event at a given time. The proportional hazards assumption was confirmed by Schoenfeld’s global test and inspection of log (―log [survival]) curves. Propensity score (PS) matching was used for making causal inferences for the treatment on the clinical outcome. A set of pre-test covariates that were associated with the treatment was selected and PS scores were estimated using logistic regression, with treatment as the outcome measure. Separate PS-matched Cox regression models with and without adjustment for potential confounders were used (see Appendix), but only the results of the overall and inverse-probability-of-treatment-weighted (IPTW) Cox regression analysis are shown. Analyses were adjusted for gender, age, comorbidity CVA, comorbidity diabetes, comorbidity asthma/COPD, use of broad-spectrum antibiotics, therapeutic anticoagulation, prophylactic anticoagulation, first day at ED, and ICU restriction. The combined endpoint risk regression analyses were stratified by ICU restriction, because of the distinctive patient characteristics in this group. For PS estimation and matching the PS matching R package in SPSS and the PSMATCH2 package in Stata were used. All tests were two-sided and p < 0.05 was considered statistically significant. Statistical analyses were performed using SPSS version 24.0 and the STATA version 14 statistical package (Stata Corporation, College Station, TX).
Results
Inclusion and baseline characteristics
Between February 28 and April 1, 2020, 1130 patients admitted to the 14 participating hospitals in The Netherlands met the inclusion criteria; 1106 patients were eligible for inclusion. After propensity score matching the analytic cohort consisted of 1064 patients, comprising 566 (53.2%) treated patients, both with HCQ (N = 189; 17.8%) and CQ (N = 377; 35.4%), and 498 (46.8%) untreated controls (see Figure 1).
Figure 1Number of included COVID-19 patients.
Table 1 shows the characteristics of the study population. The distribution of patients over the three hospital groups was as follows: 270 patients (25.4%) were admitted to an HCQ hospital, 532 (50%) to a CQ hospital, and 262 (24.6%) to a hospital with a protocol of no additional treatment. In both HCQ and CQ hospitals at least 70% of patients received treatment. Median time from admission to receipt of treatment was short: 1 day in both groups (HCQ 1.00, SD 1.5 days; CQ 1.00, SD 1.19 days). Most patients were male (60%) and body mass index (BMI) was 28 in all three groups. Comorbidities were comparable, except for cardiac disease, which saw a higher incidence in the non-treated group. Some patients had an ICU-restrictive policy, for instance due to comorbidity or high age: in the HCQ group 36% of patients had an ICU restriction (68/189), in the CQ group 30.5% (115/377), and 48.5% of patients without treatment (242/498) were not considered eligible for admission to the ICU. During follow-up, 191 patients (18%) died, 147 (13.8%) were admitted to the ICU, and 726 (68.2%) were discharged from the hospital upon recovery.
Table 1Characteristics of the study population.
Total (N = 1064)
Chloroquine centers (N = 532)
Hydroxychloroquine centers (N = 270)
No therapy centers (N = 262)
Variable
Treated
No treatment
Missing
p
Treated
No treatment
Missing
p
All
Missing
Total: N, %
377
35.4
155
14.6
189
17.8
81
7.6
262
24.6
Gender (male): N, %
244
64.7
78
50.3
0
0.002*
123
65.1
43
53.1
0
0.063*
156
59.5
0
Age: M, SD
66.4
13.5
71.8
15.3
0
0.000‡
64.7
14.5
63.9
17.2
0
0.944‡
68.8
14.8
0
BMI: M, SD
28.2
4.9
28.1
5.3
98
0.996‡
27.5
4.1
28.5
6.2
147
0.537‡
27.7
5.4
69
ICU restriction: N, %
115
30.8
86
55.5
0
0.000*
68
36
29
36
0
0.978*
127
48.5
0
Comorbidities: N, %
Hypertension
133
35.3
65
41.9
0
0.149*
62
32.8
25
30.9
0
0.755*
103
39.3
0
Heart failure
15
4
24
15.5
0
0.000*
12
6.3
11
13.6
0
0.051*
36
13.7
0
Myocardial infarction
29
7.7
16
10.3
0
0.322*
6
3.2
7
8.6
0
0.054*
27
10.3
0
Atrial fibrillation
43
11.4
41
26.5
0
0.000*
22
11.6
13
16
0
0.323*
34
13
0
CVA
31
8.2
22
14.2
0
0.037*
10
5.3
3
3.7
0
0.577*
20
7.6
0
Diabetes type 1 or 2
69
18.3
46
29.7
0
0.004*
47
24.9
17
21
0
0.492*
49
18.7
0
Asthma or COPD
80
21.2
35
22.6
0
0.729*
21
11.1
17
21
0
0.032*
54
20.6
0
OSAS
24
6.4
6
3.9
0
0.261*
9
4.8
2
2.5
0
0.382*
18
6.9
0
Chronic kidney disease (creat. > 150 µmol/L)
14
3.7
7
4.5
1
0.670*
12
6.3
5
6.2
0
0.956*
20
7.6
1
Active malignancy
29
7.7
12
7.7
0
0.984
14
7.4
6
7.4
0
1*
17
6.5
0
Muscle disease
5
1.3
1
0.6
0
0.499*
1
0.5
1
1.2
0
0.536*
6
2.3
1
History of DVT/LE
23
6.1
8
5.2
0
0.686*
11
5.8
5
6.2
0
0.91*
23
8.8
0
Immunosuppressive
23
6.1
8
5.2
0
0.674*
8
4.2
1
1.2
0
0.208*
32
12.2
0
Diagnosis based on…: N, %
PCR
359
95.2
145
93.5
0
0.431*
180
95.2
79
96.3
0
0.699*
252
96.2
0
CT
16
4.2
8
5.2
0
0.643*
9
4.8
2
2.5
0
0.382*
9
3.4
0
Clinical judgement
2
0.5
2
1.3
0
0.357*
0
0
0
0
0
N/A
1
0.4
0
Vitals and laboratory results at presentation: M (N), SD
Temperature
38.1
1.0
37.9
1.0
1
0.009§
38.1
1.0
38.0
1.0
1
0.476‡
38.0
1.05
1
Oxygen needed: N, %
326
86.5
93
60
0
0.000*
167
88.4
56
69.1
0
0.000*
163
62.2
0
CRP
97
72.9
83.1
75.8
2
0.003‡
105.3
76.9
64.1
48.5
28
0.0000‡
88.3
74.9
3
Leucocytes
7.0.
3.1
6.9
3.4
6
0.313‡
7.0
5.1
7.3
4.0
29
0.524‡
7.0
3.0
3
Lymphocytes
1.0
1.4
1.0
1.0
20
0.901‡
1.3
4.4
1.3
1.1
63
0.006‡
1.1
1.0
37
Platelets
207.9
83.5
204.9
81.2
11
0.443‡
205.6
95.68
177.6
107.4
67
0.357‡
203.3
86.2
6
Creatinine
93.1
44.7
106
68.1
3
0.090‡
92.8
73.5
103.0
112.6
29
0.096‡
107.9
107.6
4
LDH at presentation
356.2
142.3
312.2
118.5
40
0.000‡
346.7
148.1
340.1
140.1
54
0.692‡
347.2
143.6
22
Pre-hospital medication: N, %
ACE inhibitors
55
14.6
34
22.1
2
0.037*
30
16.0
15
18.8
0
0.588*
52
20.1
3
Angiotensine-2 receptor antagonists
48
12.8
24
15.6
2
0.390*
25
13.4
9
11.3
4
0.624*
27
10.5
4
Therapeutic anticoag.
50
13.3
37
24
2
0.002*
29
15.8
17
21.5
7
0.26*
51
19.9
6
In-hospital medication
Broad-spectrum antibiotics: N, %
327
86.7
99
63.9
0
0.000*
185
97.9
71
87.7
0
0.0010*
196
74.8
0
Azithromycin: N, %
31
8.2
33
21.3
0
0.000*
48
25.4
45
55.6
0
0.0000*
53
20.2
0
Cumulative dosage AZM: M (N), SD
833.3
461.1
1241.9
560.8
3
0.001‡
2020.8
1115.5
1661.1
834.5
0
0.137‡
2264.4
925.4
1
Cumulative dosage CQ/HCQ: M (N), SD
2179.5
897.6
N/A
N/A
1
N/A
1823.5
636.1
N/A
N/A
19
N/A
N/A
N/A
N/A
Therapeutic anticoag.: N, %
66
17.5
51
32.9
0
0.000*
38
20.1
19
23.5
0
0.536*
56
21.4
0
Prophylactic anticoag.: N, %
318
84.4
99
63.9
0
0.000*
161
85.2
57
70.4
0
0.005*
148
56.5
0
Deep venous thrombosis: N, %
1
0.3
0
0
3
0.519*
0
0
0
0
1
N/A
2
0.8
3
Pulmonary embolism: N, %
6
2.1
1
0.8
115
0.355*
3
1.6
0
0
4
0.253*
4
1.5
3
Endpoints
Discharged for cured infection: N, %
245
65.0
107
69.0
0
0.370*
139
73.5
58
71.6
0
0.742*
177
67.6
0
ICU admission: N, %
72
19.1
10
6.5
0
0.000*
20
10.6
3
3.7
0
0.064*
42
16.4
0
Death or hospice: N, %
60
15.9
38
24.5
0
0.020*
30
15.9
20
24.7
0
0.087*
43
16.0
0
M = mean, SD = standard deviation, *= χ2 test, † = Fisher exact test, § = independent t-test, ‡ = non-parametric Mann-Whitney test, HCQ = hydroxychloroquine, CQ = chloroquine, AZM = azithromycin, BMI = body mass index, ICU = intensive care unit, CVA = cerebrovascular accident, OSAS = obstructive sleep apnea syndrome.
• Open table in a new tab
Primary outcomes
Table 2 shows the results of the unadjusted and adjusted overall and weighted competing risk analyses for the different endpoints by type of medication. Figure 2A and B show the corresponding cumulative incidence functions (CIF). Multivariate analysis proves that both CQ and HCQ use were not statistically associated with a risk of death on the non-ICU COVID ward (for CQ, hazard ratio (HR) = 0.99, 95% CI = 0.70–1.43; for HCQ, HR = 0.96, 95% CI = 0.63–1.45). However, HCQ use was associated with a statistically significant decreased risk of transfer to the ICU (HR = 0.47, 95% CI = 0.27–0.82, p = 0.008) when compared with controls. This effect was not found in the CQ group (HR = 0.80; 95% CI = 0.55–1.15, p = 0.207). In addition, for the composite adverse endpoint, a significantly decreased risk was observed for HCQ (HR = 0.68, 95% CI = 0.49–0.95, p = 0.024) but not for CQ use (HR = 0.85, 95% CI = 0.66–1.10, p = 0.224).
Table 2Clinical outcome hazard ratio (HR) estimates for HCQ and CQ use among COVID19 patients under separate risk models.
N = 1012*
Endpoint: death
Endpoint: ICU admission
Combined endpoint
Unadjusted
Adjusted3
Unadjusted
Adjusted4
Unadjusted
Adjusted5*
Model
Drug use
HR
95% CI
p-value
HR
95% CI
p-value
HR
95% CI
p-value
HR
95% CI
p-value
HR
95% CI
p-value
HR
95% CI
p-value
Overall1
None (ref)
1.0
1.0
1.0
1.0
1.0
1.0
CQ
0.64
0.47–0.88
0.007
1.01
0.71–1.44
0.937
1.50
1.05–2.13
0.024
0.91
0.63–1.31
0.619
0.94
0.74–1.18
0.590
0.97
0.75–1.24
0.795
HCQ
0.62
0.41–0.93
0.020
0.92
0.58–1.46
0.736
0.82
0.49–1.37
0.453
0.56
0.33–0.95
0.031
0.69
0.50–0.95
0.023
0.73
0.51–1.02
0.068
Weighted Competing risk2
None (ref)
1.0
1.0
1.0
CQ
0.86
0.61–1.21
0.392
0.99
0.70–1.43
0.991
0.93
0.64–1.35
0.708
0.80
0.55–1.15
0.207
0.85
0.67–1.10
0.205
0.85
0.66–1.10
0.224
HCQ
0.87
0.58–1.32
0.518
0.96
0.63–1.45
0.681
0.52
0.30–0.89
0.017
0.47
0.27–0.82
0.008
0.66
0.48–0.91
0.011
0.68
0.49–0.95
0.024
1Cox regression model without propensity score (PS) adjustment and competing regression analysis; 2competing risk regression with weighted PS adjustment (see statistical method section for explanation of the different models); HR = hazard ratio; CI = confidence interval; CQ = chloroquine; HCQ = hydroxychloroquine; *total number of patients in the analysis; 3,4,5adjusted for gender, age, comorbidity CVA, comorbidity diabetes, comorbidity asthma/COPD, use of broad-spectrum antibiotics, therapeutic anticoagulation, prophylactic anticoagulation, first day in ED, ICU restriction.
All analyses except the competing risks regression were stratified by ICU restriction to reflect underlying potential differences in adverse incidences and risk factor prevalences.
• Open table in a new tab
Figure 2Cumulative incidence functions (CIF) by type of medication. A. Cumulative risk of death. B. Cumulative risk of transfer to ICU.
• View Large Image
• Figure Viewer
• Download Hi-res image
• Download (PPT)
Secondary outcomes
Since the use of azithromycin (AZM) and angiotensin receptor blockers (ARB) has been postulated to have an effect on COVID-19, we additionally analyzed the effect of this treatment on outcome; 210 patients were started on AZM therapy on admission, while 854 patients did not receive AZM. In the KM analysis there was no significant difference between these two groups in reaching the composite adverse endpoint (Plog rank = 0.071) and no significant interaction effect was found for H(CQ) combined with AZM use (p = 0.2195).
In total, 180 patients were using angiotensin-II receptor antagonists (ARB, n = 70) or angiotensin-converting enzyme inhibitors (ACEi, n = 110), and continued treatment during admission. There was no difference in outcome for the composite adverse endpoint for continued ACEi use (HR = 1.21; 95% CI = 0.78–1.90, p = 0.397) nor for continued ARB use (HR = 1.21; 95% CI = 0.70–2.10, p = 0.498), as compared with no therapy.
(Stata Corporation, College Station, TX).
Table A1Clinical outcome hazard ratio estimates for HCQ and CQ use among COVID19 patients under separate propensity risk models.
N = 1012*
Endpoint: death
Endpoint: ICU admission
Combined endpoint
Unadjusted
Adjusted3
Unadjusted
Adjusted3
Unadjusted
Adjusted4
Model
Drug use
HR
95% CI
p-value
HR
95% CI
p-value
HR
95% CI
p-value
HR
95% CI
p-value
HR
95% CI
p-value
HR
95% CI
p-value
Overall1
None (ref)
1.0
1.0
1.0
1.0
1.0
1.0
CQ
0.64
0.47–0.88
0.007
1.01
0.71–1.44
0.937
1.50
1.05–2.13
0.024
0.91
0.63–1.31
0.619
0.94
0.74–1.18
0.590
0.96
0.75–1.24
0.772
HCQ
0.62
0.41–0.93
0.020
0.92
0.58–1.46
0.736
0.82
0.49–1.37
0.453
0.56
0.33–0.95
0.031
0.69
0.50–0.95
0.023
0.71
0.51–1.01
0.054
Matched paired2
None (ref)
1.0
1.0
1.0
1.0
1.0
1.0
CQ
0.66
0.38–1.13
0.130
0.59
0.23–1.52
0.272
0.85
0.53–1.38
0.523
0.82
0.41–1.64
0.567
0.77
0.53–1.10
0.144
0.85
0.50–1.43
0.535
HCQ
0.66
0.34–1.26
0.207
0.35
0.12–1.03
0.057
0.41
0.22–0.78
0.007
0.48
0.21–1.08
0.075
0.51
0.32–0.81
0.004
0.50
0.28–0.91
0.023
Stratified quintiles2
None (ref)
1.0
1.0
1.0
1.0
1.0
1.0
CQ
0.77
0.54–1.08
0.124
0.98
0.68–1.41
0.919
0.97
0.67–1.40
0.863
0.79
0.54–1.14
0.204
0.86
0.67–1.11
0.242
0.89
0.69–1.15
0.373
HCQ
0.84
0.55–1.30
0.431
0.86
0.54–1.39
0.547
0.48
0.29–0.82
0.007
0.50
0.29–0.84
0.010
0.65
0.46–0.91
0.011
0.66
0.46–0.94
0.020
Weighted2
None (ref)
1.0
1.0
1.0
1.0
1.0
1.0
CQ
0.81
0.58–1.15
0.243
0.94
0.65–1.35
0.732
0.91
0.63–1.32
0.615
0.79
0.55–1.15
0.217
0.85
0.67–1.10
0.205
0.85
0.66–1.10
0.228
HCQ
0.78
0.52–1.18
0.244
0.93
0.61–1.41
0.734
0.50
0.29–0.85
0.011
0.47
0.27–0.82
0.008
0.66
0.48–0.91
0.011
0.68
0.49–0.95
0.022
1Cox regression model without propensity score (PS) adjustment; 2PS adjustment – see statistical method section for explanation of the different models; HR = hazard ratio, CI = confidence interval; CQ = chloroquine, HCQ = hydroxychloroquine; 3adjusted for gender, age, comorbidity CVA, comorbidity diabetes, comorbidity asthma/COPD, use of of broad-spectrum antibiotics, therapeutic anticoagulation, prophylactic anticoagulation, first day in ED; 4adjusted for gender, age, comorbidity asthma/COPD, use of broad-spectrum antibiotics, prophylactic anticoagulation, first day in ED; *all analyses were stratified by ICU restriction to reflect underlying potential differences in adverse incidences and risk factor prevalences.
1.
◦ Google Scholar
Publication History
Published online: September 29, 2020
Accepted: September 23, 2020
Received in revised form: September 23, 2020
Received: August 1, 2020
Identification
DOI: https://doi.org/10.1016/j.ijid.2020.09.1460
Figures
•
Figure 1Number of included COVID-19 patients.
•
Figure 2Cumulative incidence functions (CIF) by type of medication. A. Cumulative risk of death. B. Cumulative risk of transfer to ICU.
An opposing point of view on HCQ: that is stated, not yet analyzed, presented for evenhanded balance: “Hydroxychloroquine does not inhibit SARS-CoV-2 infection in preclinical models
by Lindsay Brownell, Hansjörg Wyss Institute for Biologically Inspired Engineering 8/27/20
Human organ chips lined with lung cells, originally developed at the Wyss Institute, were one of the complex in vitro models used to demonstrate that the drugs did not have a significant impact on the infection rate of human cells. Credit: Wyss Institute at Harvard University
An international collaboration of researchers across more than 80 countries has come to the conclusion that chloroquine (CQ) and hydroxchloroquine (HCQ) are unlikely to provide clinical benefit against COVID-19. In a new commentary paper co-authored by Wyss Founding Director Donald Ingber, MD, PhD, a group of scientists describe multiple recent studies in human Organ Chips and other multi-tissue in vitro models, mice, hamsters, and non-human primates, all of which strongly indicate the drugs do not have the efficacy suggested by earlier results obtained from in vitro studies with cultured cell lines. The paper was published today in Nature Communications.
“Given the urgency of finding a treatment for COVID-19, repurposing existing drugs is a faster approach than developing completely new drugs from scratch. But, as we’ve seen, the hype around hydroxychloroquine and chloroquine as potential therapies was based on studies that didn’t accurately reflect their effects in humans,” said Ingber, who is also the Judah Folkman Professor of Vascular Biology at Harvard Medical School and Boston Children’s Hospital, and Professor of Bioengineering at the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS). “Touting them as ‘wonder drugs’ before they had undergone thorough, systematic evaluation has been extremely detrimental to the fight against COVID-19, and in this article, authors working in independent labs around the world highlight multiple studies that all suggest the drugs should never have been considered to be effective treatments.”
In February 2020 as the COVID-19 outbreak was gaining speed, the World Health Organization (WHO) convened an ad hoc working group of scientists to encourage open data access, help avoid duplication of effort, and reduce the reliance on animal experimentation in the search for treatments for SARS-CoV-2 infection. Preliminary studies conducted using cultured Vero cells, which are derived from kidney cells extracted from an African green monkey, suggested that CQ and HCQ could reduce the likelihood or severity of SARS-CoV-2 infection. However, it is well known that cells grown in a dish, especially from a non-human species, are a poor proxy for the human body.
Mice are regularly used to test potential drugs, but the animals are naturally resistant to SARS-CoV-2; as a result, either the virus needs to be adapted to be more infectious, or the mice’s natural state needs to be changed to allow infection, both of which could compromise the relevance of results obtained from these studies. Nevertheless, work by co-author Matthew Frieman, Ph.D. Associate Professor of Microbiology and Immunology at The University of Maryland School of Medicine demonstrated that when mice were injected with CQ or HCQ, then exposed to a mouse-adapted SARS-CoV strain, lung inflammation in the treated mice was reduced compared to untreated mice. However, there was no difference in the amount of virus present in their lungs, suggesting that CQ and HCQ did not produce an effective an antiviral effect in vivo.In an effort to provide more accurate data about the drugs’ potential activity in humans than could be obtained from in vitro cells or mice, the co-authors of the new paper oversaw research projects in several different countries that evaluated CQ and HCQ’s anti-SARS-CoV-2 activity in human Organ Chips and other more complex in vitro human tissue models, as well as hamsters and two species of non-human primates.
Human lung chips developed at the Wyss Institute and commercialized by Emulate, Inc. were used to test CQ’s effect on lung cells infected with SARS-CoV-2 pseudoviruses (lentivirus particles engineered to express the SARS-CoV-2 spike protein). CQ did not significantly inhibit the replication of the SARS-CoV-2 Spike pseudotyped viruses in the lung cells, and more recent findings confirmed that HCQ is ineffective as well. Meanwhile, in France, a research team at Inserm developed another complex human in vitro model system called MucilAir, which is derived from primary nasal or bronchial cells differentiated and cultivated under an air/liquid interface. In alignment with the findings by the Wyss Institute, Inserm concluded that HCQ does not significantly inhibit SARS-CoV-2 infection in their human respiratory tissue model.
Unlike mice, hamsters are naturally susceptible to the SARS-CoV-2 virus, and therefore provide a more accurate rodent model of human infection. Independent groups at Katholieke Universiteit (KU) Leuven, Belgium and Rocky Mountain Laboratories (RML) in Montana, US investigated HCQ’s effects in hamsters, either alone or in combination with azithromycin, an antibiotic also purported to treat COVID-19 in humans. In the KU Leuven studies, infected hamsters that were given HCQ alone did not display a significant reduction in detectable viral RNA in their lungs, and hamsters that were given HCQ with azithromycin displayed a 3-fold increase in viral RNA. The RML studies tested HCQ’s efficacy as both a prophylactic to prevent SARS-CoV-2 infection and as a treatment post-infection, and revealed no significant difference in infection, disease progression, viral replication, or virus shedding between HCQ-treated and control groups.
Testing drugs in non-human primates is a big step closer to testing them in humans, and two groups evaluated the effect of HCQ on SARS-CoV-2 infection in two different primate species. Researchers at Inserm studied cynomolgus macaques and found no significant anti-viral or clinical benefit of HCQ when given prophylactically or after infection, at several different doses, and with or without azithromycin. The viral loads in the animals’ respiratory tract, lesions observed by chest CT scan, and clinical signs were comparable in the treated vs. untreated groups. RML researchers conducted similar studies in rhesus macaques, and found that animals in HCQ-treated and control groups developed similar mild to moderate disease both when HCQ was given prophylactically and after infection, and displayed indistinguishable SARS-CoV-2 replication and shedding in their lower and upper respiratory tracts.
“The fact that all of these studies in different models produced the same results is really convincing evidence that these drugs are very unlikely to be effective in humans, and we should invest our time and energy into exploring other options,” said Frieman. The Wyss Institute is also collaborating with Frieman’s lab on a DARPA-funded project to identify and test additional drugs that can be repurposed to treat or prevent COVID-19.
More information: S. G. P. Funnell et al. Emerging preclinical evidence does not support broad use of hydroxychloroquine in COVID-19 patients, Nature Communications (2020). DOI: 10.1038/s41467-020-17907-w “
A review of the support for the use of HCQ: A Meta-Analysis on the Effects of Hydroxychloroquine on COVID-19
published https://www.cureus.com/articles/38513 on August 24, 2020. Dean stated that most of the positive studies were done after April of 2020. There are now 53 studies that show positive results of hydroxychloroquine in COVID infections. There are 14 global studies that show neutral or negative results — and 10 of them were of patients in very late stages of COVID-19, where no antiviral drug can be expected to have much effect. The other four have been discredited as fake science. The fact is that if given early enough, HCQ works to stop the disease. It is a scientific fact that HCQ works best when given as prophylaxis or within the first 5 – 7 days of symptoms.
Drug Treatments for Covid-19
BMJ : British Medical Journal
• This meta-analysis of data from 23 randomized trials compared drug treatments with standard care for patients with COVID-19. Despite low certainty of evidence for most comparisons due to imprecision and lack of blinding, there was evidence suggesting that glucocorticoid treatment versus standard care may lower both mortality and mechanical ventilation. Additionally, low-certainty evidence suggested that treatment duration may be shortened for patients receiving hydroxychloroquine, remdesivir, and lopinavir–ritonavir versus standard care, but that there may be a higher risk of adverse events associated with hydroxychloroquine treatment.
• Although this meta-analysis provides some information about the effects of drug treatments versus standard care for COVID-19, the efficacy of most interventions remains unclear due to small sample sizes in trials & important study limitations.
8/6/20 Amer J of Medicine summary of HCQ use with Azithromycin or Doxycycline showing specific dosages: https://www.amjmed.com/article/S0002-9343(20)30673-2/fulltext#%20
Pathophysiological Basis and Rationale for Early Outpatient Treatment of SARS-CoV-2 (COVID-19) Infection
• Peter A. McCullough, MD, MPH
• Ronan J. Kelly, MD
• Gaetano Ruocco, MD
•
• William W. O’Neill, MD
• Marcus Zervos, MD
• Harvey A. Risch, MD, PhD
International Journal of Antimicrobial Agents
Volume 56, Issue 6, Demcember 2020, 106214
COVID-19 outpatients: early risk-stratified treatment with zinc plus low-dose hydroxychloroquine and azithromycin: a retrospective case series study
RolandDerwanda1MartinScholzb1
VladimirZelenkoc
https://doi.org/10.1016/j.ijantimicag.2020.106214Get rights and content
First COVID-19 outpatient study based on risk stratification and early antiviral treatment at the beginning of the disease. Low-dose hydroxychloroquine combined with zinc and azithromycin was an effective therapeutic approach against COVID-19. Significantly reduced hospitalisation rates in the treatment group. Reduced mortality rates in the treatment group.
The aim of this study was to describe the outcomes of patients with coronavirus disease 2019 (COVID-19) in the outpatient setting after early treatment with zinc, low-dose hydroxychloroquine and azithromycin (triple therapy) dependent on risk stratification. This was a retrospective case series study in the general practice setting. A total of 141 COVID-19 patients with laboratory-confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in the year 2020 were included. The main outcome measures were risk-stratified treatment decision and rates of hospitalization and all-cause death. A median of 4 days [interquartile range (IQR) 3–6 days; available for n = 66/141 patients] after the onset of symptoms, 141 patients (median age 58 years, IQR 40–67 years; 73.0% male) received a prescription for triple therapy for 5 days. Independent public reference data from 377 confirmed COVID-19 patients in the same community were used as untreated controls. Of 141 treated patients, 4 (2.8%) were hospitalized, which was significantly fewer (P < 0.001) compared with 58 (15.4%) of 377 untreated patients [odds ratio (OR) = 0.16, 95% confidence interval (CI) 0.06–0.5]. One patient (0.7%) in the treatment group died versus 13 patients (3.4%) in the untreated group (OR = 0.2, 95% CI 0.03–1.5; P = 0.12). No cardiac side effects were observed. Risk stratification-based treatment of COVID-19 outpatients as early as possible after symptom onset using triple therapy, including the combination of zinc with low-dose hydroxychloroquine, was associated with significantly fewer hospitalizations.
In December 2019, the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) started as an outbreak in Wuhan, China. This coronavirus has spread rapidly as a pandemic around the world [1], causing coronavirus disease 19 (COVID-19) pneumonia, acute respiratory distress syndrome (ARDS), cardiac injury, liver and renal injury, thrombosis and death [2].
In contrast to many other studies, the most frequent symptom was cough and not fever [58,59]. Changes in smell or taste in one-third of patients and a negative correlation with age were similar to findings from other groups [60]. While mean time from onset of symptoms to treatment was only 4.8 days (median 4 days), previously reported time spans range from 6.3 days [61] to 8 days [16], up to 16.6 days [14], or it was often even not reported [62]. In most of these studies, COVID-19 disease had most likely already progressed at the time of presentation to stages II or even stage III of the disease [6]. In many studies, often only limited information is provided about co-medications and specifically about clinical symptoms at admission [62]. The latter would be very important to better understand the differences in clinical presentation between inpatients and outpatients and thus the urgency for early anti-COVID-19 treatment in the outpatient setting [63]. The potential of zinc to enhance the antiviral efficacy of HCQ was already described in detail elsewhere [22]. This hypothesis was recently confirmed in a study using a similar triple therapy and treatment duration [23]. Zinc added to HCQ and azithromycin resulted in a significantly increased number of patients being discharged, a reduction in mortality, or transfer to hospice. In another study, when a lower dose of 200 mg of HCQ twice daily was added to basic treatment, mortality of even critically ill patients was significantly reduced [64]. These and our findings indicate that proper dosing of HCQ with its long half-life might be key for a favourable outcome of COVID-19 patients. In critical care, drugs with short half-lives are usually preferred. Especially in critically ill COVID-19 patients, higher doses of HCQ may have unforeseeable effects, e.g. on insulin sensitivity in obese patients [65] and on glucose levels in diabetics [66,67]. Besides glucose levels, it is important to closely monitor renal function, which is increasingly affected during progression of COVID-19 [68]. Because HCQ is substantially excreted by the kidneys, the risk of toxic reactions is greater in patients with impaired renal function [69].
4.1. Potential implications for clinicians and policy-makers
Clinical experience from severely ill inpatients with pneumonia who were treated with high-dose HCQ is not readily transferable to the outpatient setting with upper respiratory tract disease only. For outpatients with a median of only 4 days after onset of symptoms, COVID-19 represents a totally different disease and needs to be managed and treated differently [63]. A simple-to-perform outpatient risk stratification, as shown here, allows for rapid treatment decisions and treatment with the triple therapy of zinc, low-dose HCQ and azithromycin and may prevent a large number of hospitalizations and probably deaths during the SARS-CoV-2 pandemic. This might also help to avoid overwhelming of healthcare systems.
From VuMedi 6/12/21: How to recognize delayed reactions from Corona Virus Vaccines
https://www.vumedi.com/video/delayed-reaction-and-sars-cov-2-mrna-vaccines-how-to-recognize-common-vs-unusual-adverse-reactions/?token=85d9721c-7ff2-4993-97b3-4866d2ae9241&utm_source=COVID%20Interests%20Criteria_84560&utm_medium=Video&utm_campaign=%5Bcovid%20June%2012%5D%20Delayed%20Reaction%20and%20SARS-CoV-2%20mRNA%20Vaccines%3A%20How%20to%20Recognize%20Common%20vs.%20Unusual%20Adverse%20Reactions%3F&utm_content=Delayed%20Reaction%20and%20SARS-CoV-2%20mRNA%20Vaccines%3A%20How%20to%20Recognize%20Common%20vs.%20Unusual%20Adverse%20Reactions%3F&utm_term=COVID%20Vaccines&link_data=eyJidWxrX21haWxfYWN0aW9uIjoiYyIsInJlY2lwaWVudF9pZCI6MTI3MjM3NDIzMywibWFpbF9pZCI6ODQ1NjB9%3A1ls5Eg%3Ag_NLMMHyxpkQQ5P3XTzEc8K2CN8&mail_id=84560
From B. C. Joondeph, MD 12/18/20: Now that the AMA has cleared the use of HCQ for Cov2-19 treatment “…There have been 187 hydroxy studies, 122 of which were peer-reviewed. 100 percent of these studies reported positive effects for early treatment of COVID, meaning, for those not yet in the hospital, and certainly not on a ventilator….”
• Dr. Anthony Fauci NIH director has known since 2005 that chloroquine is an effective inhibitor of coronaviruses. The NIH researched chloroquine and concluded that it was effective at stopping the SARS corona virus. COVID-19 is also a coronavirus, labeled SARS-CoV-2. While not exactly the same virus as SARS-CoV-1, it is genetically related and shares 79% of its genome, as the name SARS-CoV-2 implies. Both CoV1 and 2 use the same host cell receptor, which is what viruses use to gain entry to the human cell and infect the victim. The official publication of the NIH, the Virology Journal, published 8/22/05 “Chloroquine is a potent inhibitor of SARS coronavirus infection and spread.” “We report…that chloroquine has strong antiviral effects on SARS-CoV infection of primate cells. These inhibitory effects are observed when the cells are treated with the drug either before or after exposure to the virus, suggesting both prophylactic and therapeutic advantage.”
•
• American Infectious Disease specialist Joseph Rahimian, MD, explained that, in relation to Covid-19, zinc ‘does the heavy lifting and is the primary substance attacking the pathogen’. HCQ is said to work as a delivery system for zinc in fighting coronavirus
Lower zinc levels in the blood are associated with an increased risk of death in patients with COVID-19
by European Society of Clinical Microbiology and Infectious Diseases9/23/20
Credit: CC0 Public Domain
New research presented at this week’s ESCMID Conference on Coronavirus Disease (ECCVID, held online from 23-25 September, 2020) shows that having a lower level of zinc in the blood is associated with a poorer outcome in patients with COVID-19. The study is by Dr. Roberto Güerri-Fernández, Hospital Del Mar, Barcelona, Spain, and colleagues.
Increased intracellular zinc concentrations efficiently impair replication/reproduction of a number of viruses. However, the effect of plasma zinc levels on SARS-COV-2 is not yet understood. In this study, the authors explored whether plasma zinc levels at admission are associated with disease outcome in COVID-19 patients.
Mean baseline zinc levels among the 249 patients were 61 mcg/dl. Among those who died, the zinc levels at baseline were significantly lower at 43mcg/dl vs 63.1mcg/dl in survivors. Higher zinc levels were associated with lower maximum levels of interleukin-6 (proteins that indicate systemic inflammation) during the period of active infection.
After adjusting by age, sex, severity and receiving hydroxychloroquine, statistical analysis showed each unit increase of plasma zinc at admission to hospital was associated with a 7% reduced risk of in-hospital mortality. Having a plasma zinc level lower than 50mcg/dl at admission was associated with a 2.3 times increased risk of in-hospital death compared with those patients with a plasma zinc level of 50mcg/dl or higher.
HRS, this author, states there is far too much concern and publicity about the very infrequent, “vanishingly low” frequency, although important, irregular heartbeat potential (risk vs benefit) that should be observed for as opposed to the very high frequency disastrous effects of not treating corona virus infections with what has proven to be this very safe combination of medications. THE FDA guidelines that have come with hydroxychloroquine (HCQ) as in the treatment for lupus do not even recommend doing an EKG.
Not yet vetted: “Researchers from the Clinic of Infectious Diseases, Department of Health Science, ASST Santi Paolo e Carlo, University of Milan in Italy reported their new finding in the International Journal of Infectious Diseases. This study shows that hydroxychloroquine and azithromycin are associated with a dramatic 66% reduction in risk of death among the 539 COVID-19 patients hospitalized in Milan between February 24 and May 17, 2020.”
India backs hydroxychloroquine for virus prevention (similar to Brazil): 5/26/20
India’s top biomedical research body on Tuesday backed the use of the anti-malarial hydroxychloroquine as a preventive against coronavirus, after the WHO suspended clinical trials of the drug over safety concerns. The endorsement from the Indian Council of Medical Research came a week after US President Donald Trump said he was taking the drug as a preventative measure.
Observational and case control studies in India showed there were “no major side effects” of taking the drug as a prophylactic, ICMR Director-General Balram Bhargava said. Last week, the ICMR—which is leading the government’s response to the virus—expanded its advisory for the use of hydroxychloroquine as a preventative measure.
“We recommended that for prophylaxis, it should be continued, because there is no harm. Benefit may be there,” Bhargava told reporters.
The Key to Defeating COVID-19 Already Exists. We Need to Start Using It | Opinion
https://www.newsweek.com/key-defeating-covid-19-already-exists-we-need-start-using-it-opinion-1519535
Harvey A Risch, MD, PhD, Professor eof Epidemiology, Yale School of Public Health
7/23/20
“…As professor of epidemiology at Yale School of Public Health, I have authored over 300 peer-reviewed publications and currently hold senior positions on the editorial boards of several leading journals. I am usually accustomed to advocating for positions within the mainstream of medicine, so have been flummoxed to find that, in the midst of a crisis, I am fighting for a treatment that the data fully support but which, for reasons having nothing to do with a correct understanding of the science, has been pushed to the sidelines. As a result, tens of thousands of patients with COVID-19 are dying unnecessarily. Fortunately, the situation can be reversed easily and quickly.
I am referring, of course, to the medication hydroxychloroquine. When this inexpensive oral medication is given very early in the course of illness, before the virus has had time to multiply beyond control, it has shown to be highly effective, especially when given in combination with the antibiotics azithromycin or doxycycline and the nutritional supplement zinc.
On May 27, 2020, I published an article in the American Journal of Epidemiology (AJE) entitled, “Early Outpatient Treatment of Symptomatic, High-Risk COVID-19 Patients that Should be Ramped-Up Immediately as Key to the Pandemic Crisis.” That article, published in the world’s leading epidemiology journal, analyzed five studies, demonstrating clear-cut and significant benefits to treated patients, plus other very large studies that showed the medication safety….”
A longish evenhanded article with references re HCQ use and data:
Hydroxychloroquine for SARS-CoV-2 Infection: How Did We Get Here?
David C. Helfgott, MD, in Rheumatology Advisor 5/8/20
Larger randomized controlled clinical trials are required to better understand if hydroxychloroquine has a role in the treatment of COVID-19.
Hydroxychloroquine is a less toxic metabolite of the antimalarial drug chloroquine and is used as an immunomodulator for the treatment of autoimmune diseases.1-3 Chloroquine and hydroxychloroquine have been demonstrated to inhibit viral infection in cell culture,4-6 leading investigators to hypothesize that they may have an in vivo antiviral effect. Despite the absence of good controlled clinical trial evidence of its effectiveness, hydroxychloroquine has gained widespread use in the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection.
In other times, the absence of good clinical data would have precluded such use of a drug in patients. However, during this difficult time of the coronavirus disease 2019 (COVID-19) pandemic, news reports on the scant data that currently exists on the use of hydroxychloroquine for SARS-CoV-2 and the endorsement of hydroxychloroquine by the President of the United States has influenced the public perception of its effectiveness and the medical response. On March 28, 2020, the US Food and Drug Administration issued an emergency use authorization for hydroxychloroquine for patients with COVID-19.7
Research conducted during and after the 2003 SARS-CoV-1 outbreak in China demonstrated in vitro antiviral effects of chloroquine and hydroxychloroquine against this virus.4,8 Chloroquine2,9 and hydroxychloroquine2,10 have been shown to also inhibit SARS-CoV-2 growth in cell culture.
In February 2020, it was announced in China that chloroquine was found to be more effective than control treatment in clinical trials of patients with COVID-19.11 Officials announced that chloroquine treatment prevented worsening of pneumonia, improved findings on lung imaging, facilitated conversion to virus-negative status, and reduced disease duration, without significant side effects,11 leading to a panel recommendation in that country for its use in COVID-19.12 This soon led to the global use of hydroxychloroquine for COVID-19.
Gautret et al subsequently published a study that set out to examine the effect of hydroxychloroquine (200 mg 3 times a day for 10 days) on nasopharyngeal SARS-CoV-2 viral load in patients with confirmed infection.14 They enrolled 26 hospitalized patients with COVID-19 infection at a single hospital to receive hydroxychloroquine; they also enrolled 16 patients with COVID-19 infection who refused inclusion or did not meet inclusion criteria at that hospital, as well as patients at 3 other hospitals, as controls.
Of the 26 patients who received HYDROXYCHLOROQUINE, 6 were not included in the final analysis; they were considered lost to follow-up because of transfer to the intensive care unit (ICU; 3 patients), death (1 patient), leaving hospital (1 patient), and stopped treatment (1 patient). The average age of the group receiving HCQ was older than the control group (not quite statistically significant); there was not a statistically significant difference in clinical status. Six patients in the HCQ-treated group also received AZITHROMYCIN to prevent bacterial superinfection.14
The investigators found that on days 3, 4, 5, and 6 there was a statistically significant difference in the number of patients with a negative viral load between the 2 groups, such that by day 6 the viral load was negative in 70% of patients in the HCQ-treated group vs 12.5% in the control group.14
The researchers went on to compare the HCQ-treated group (n=14) with the HCQ plus AZITH-treated group (n=6). They found a significant difference in the number of patients with a negative viral load on days 3, 4, 5, and 6 in favor of the combination treatment, with 100% of patients in the combination group virus-negative compared with 57.1% in the HCQ-alone group on day 6.14 Of note, however, of the 6 patients in the hydroxychloroquine-treated group who did not have a negative viral load at day 6, four participants demonstrated a higher viral load on day 0 than any of the patients who received HCQ + AZITH,14 implying that initial viral load may have played an important role in day 6 viral load.
Subsequent to this study, another group from France reported on 11 consecutive patients who received HCQ ) AZITH dosed as per the Gautret study.15 Of these patients, 1 died and 8 of the remaining 10 had persistent positive SARS-CoV-2 viral loads at days 5 and 6.15
In another study conducted in China, 30 patients were randomly assigned to receive HCQ (400 mg/day for 5 days) or control standard treatment; clinical findings were similar between the groups at study onset.16 In this study, there was no difference in viral load between the 2 groups on day 7, with 86.7% of the study group and 93.3% of the control group reported as being virus-negative.16
Most recently, a report by Chen et al presented data from a study including 62 patients with nonsevere, noncritical COVID-19 who were randomly assigned to receive HCQ (200 mg twice a day for 5 days) or standard treatment.17 Results showed that duration of fever (2.2 vs 3.2 days) and cough (2.0 vs 3.1 days) was shorter among members of the group receiving hydroxychloroquine, and that more patients receiving HCQ had improved findings on chest computed tomographic imagingy.17 The study authors also noted that of the 62 patients enrolled, 4 patients, all in the standard treatment group, demonstrated progression to severe infection.17
Given the encouraging in vitro data against a host of viruses, animal models have been used to study the efficacy of chloroquine in treating a variety of non-COVID-19 viral infections, and results have been variable.18 Human trials of chloroquine for the prevention or treatment of influenza,19, dengue,20, and chikungunya21, 22 viruses have not demonstrated efficacy. The evidence thus far for the use of HCQ in the treatment of human infection with SARS-CoV-2 is based on encouraging in vitro data, very small clinical studies, and anecdotal observation.
The randomized study by Chen et al17 was small and did not include patients with severe disease. It is notable, however, that only 4 of 62 patients progressed from non-severe disease to severe disease, implying that the study population had quite mild illness. The other randomized study reported16 examined viral loads and did not find a difference in viral load between hydroxychloroquine-treated and untreated patients at day 7. Conversely, Gautret et al noted improved viral loads among patients in the HCQ-treated group compared with untreated patients. However, this was a small, nonrandomized study in which the control group was culled from several hospitals with likely differing standard therapies, and 4 patients in the HCQ group who required care in an intensive care unit or died were not included in the analysis.14 The study that evaluated AZITH was observational in nature and few conclusions could be surmised from the set of AZITH data.14 It should also be noted that there is concern for is prolongation and torsades de pointes with even short-term use of hydroxychloroquine for COVID-19.23
Thus, larger randomized controlled trials are required to better understand if hydroxychloroquine has a role in the treatment of COVID-19. In the United States and elsewhere, several such trials are ongoing or planned and hopefully data will be available soon.24
References
1. Schrezenmeier E, Dorner T. Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nature Rev. 2020;16:155-166.
2. Lee SJ, Silverman E, Bargman JM. The role of antimalarial agents in the treatment of SLE and lupus nephritis. Nat Rev Nephrol. 2011;7:718-729.
3. Liu J, Cao R, Xu M, et al. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro [published online March 18, 2020]. Cell Discovery. doi:10.1038/s41421-020-0156-0
4. Colson P, Rolain J-M, Lagier J-C, et al. Chloroquine and hydroxychloroquine as available weapons to fight COVID-19 [published online March 4, 2020]. Int J Antimicrob Agents. doi:10.1016/j.ijantimicag.2020.105932
5. Rolain J-M, Coilson P, Raoult D. Recycling of chloroquine and its hydroxyl analogue to face bacterial, fungal and viral infections in the 21st century. Int J Antimicrob Agents. 2007;30:297-308.
6. Savarino A, Boelaert JR, Cassone A, et al. Effect of chloroquine on viral infections: an old drug against today’s diseases. Lancet Infect Dis. 2003;3:722-727.
7. Lenzer J. Covid-19: US gives emergency approval to hydroxychloroquine despite lack of evidence [published online April 1, 2020]. British Med J. doi:10.1136/bmj.m1335
8. Keyaerts E, Vijgen L, Maes P, et al. In vitro inhibition of severe acute respiratory syndrome coronavirus by chloroquine. Biochem Biophys Res Comm. 2004;323:264-268.
9. Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019 nCoV) in vitro [published online February 4, 2020]. Cell Res. doi:10.1038/s41422-020-0282-0
10. Yao X, Ye F, Zhang M, et al. In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [published online March 9, 2020]. Clin Infect Dis. doi:10.1093/cid/ciaa237
11. Gao J, Tian Z, Yang X. Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies [published online February 29, 2020]. BioSci Trends. doi:10.5582/bst.2020.01047
12. Multicenter Collaboration Group of Department of Science and Technology of Guangdong Province and Health Commission of Guangdong Province for Chloroquine in the Treatment of Novel Coronavirus Pneumonia. Expert consensus on chloroquine phosphate for the treatment of novel coronavirus pneumonia [in Chinese]. Zhonghua Jie He He Hu Xi Za Zhi. 2020;43;185-188.
13. Devaux CA, RolaIn J-M, Colson P, et al. New insights on the antiviral effects of chloroquine against coronavirus: what to expect for COVID-19? [published online March 31, 2020]. Int J Antimicrob Agents. doi:10.1016/j.ijantimicag.2020.105938
14. Gautret P, Lagier J-C, Parola, P, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial [published online March 20, 2020. J Antimicrob Agents. doi:10.1016/j.ijantimicag.2020.105949
15. Molina J, Delaugerre C, Le Goff Breno J, et al. No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection [published online March 30, 2020]. Med et Mal Infect. doi:10.1016/j.medmal.2020.03.006
16. Chen J, Ping L, Liu, L, et al. A pilot study of hydroxychloroquine sulfate in patients with common 2019 coronavirus disease-19 (COVID-19) [in Chinese]. J ZheJiang University (Medical Sciences). 2020;49(1):0-0.
17. Chen Z, Hu J, Zhang Z, et al. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial [published online March 31, 2020]. MedRxiv. doi: 10.1101/2020.03.22.20040758
18. Touret F, de Lamballerie X. Of chloroquine and COVID-19 [published online March 5, 2020]. Antiviral Res. doi:10.1016/j.antiviral.2020.104762
19. Paton NI, Lee L, Xu Y, et al. Chloroquine for influenza prevention: a randomized, double-blind, placebo controlled trial. Lancet Infect Dis. 2011;11:677-683.
20. Tricou V, Minh NN, Van TP, et al. A randomized controlled trial of chloroquine for the treatment of dengue in Vietnamese adults. PLOS Neglected Trop Dis. 2010;4:e785.
21. De Lamballerie X, Boisson V, Reynier J-C, et al. On chikungunya acute infection and chloroquine treatment [published online December 15, 2008]. Vector Borne Zoonotic Dis. doi:10.1089/vbz.2008.0049
22. Roques P, Thiberville S-D, Dupuis-Maguiraga L, et al. Paradoxical effect of chloroquine treatment in enhancing chikungunya virus infection [published online May 17, 2018]. Viruses. doi:10.3390/v10050268
23. Giudicessi JR, Noseworthy P, Friedman PA, et al. Urgent guidance for navigating and circumventing the QTc prolonging and torsadogenic potential of possible pharmacotherapies for COVID-19 [published online March 25, 2020]. Mayo Clin Proc. doi:10.1016/j.mayocp.2020.03.024
24. United States National Library of Medicine. Clinical Trials. Accessed April 8, 2020. https://clinicaltrials.gov/ct2/results?term=hydroxychloroquine&cond=Covid-19
This article originally appeared on Medical Bag
From JAMA 4/24/20 regarding the safety of high dose HCQ:
https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2765270?utm_source=silverchair&utm_medium=email&utm_campaign=article_alert-jamanetworkopen&utm_content=wklyforyou&utm_term=042420 “…high doses, such as HCQ 600 mg twice daily for 28 days, were already studied in patients with cancer, showing good safety even in phase I trials25-27…” Here are the current Heart Rhythm Society recommendations:
◦ Electrocardiographic/QT interval monitoring:
◦ HCQ and AZITH should be withheld in patients with baseline QT prolongation or with known congenital long QT syndrome. HRS (c’est moi) states that QT prolongation is a very uncommon condition (“rare”) and unless known, in view of the seriousness of the need to treat a quite ill corona virus infected patient, this is very unlikely to need to be checked and seems an unreasonable request at this point in time.
◦ Cardiac rhythm and QT interval should be monitored; however, this may be difficult in critically ill patients as frequent contact may need to be minimized. HRS says this is absolutely correct and is as just stated above.
◦ If QTc exceeds a present threshold of 500 msec, the drugs should be discontinued. HRS again states this is unlikely to be as important as the need to treat a quite ill corona virus infected patient and unnecessarily exposes the EKG technician who will be required to do the EKG.
• Correcting hypokalemia and hypomagnesemia: HRS says this is routine and ALWAYS a good idea. So doing would likely reduce the complication rate reported in adverse studies.
◦ Potassium levels > 4 mEq/L
◦ Magnesium levels > 2 mg/dL
• Avoiding other QTc prolonging agents whenever feasible:
◦ These may include quinolones, antifungals, atypical antipsychotics, antidepressants and opioids, among others.
Mayo Clinic Proceedings 8/2020 95:1696- O Voisin: “Acute QT Interval Modifications … HCQ-Azithromycin: “…we did not observe any relevant consequences of these transitory (QT) modifications…”
See, also, alkaline and dietary suggestions in the Preventive Medicine Center Considerations below. As of 4/7/21, the population of the USA is 330 million Americans with 32,000,000 known CASES of CoV2-19 but likely (!) 65 million total cases; 560,000 have died from this disease, and 20,000,000 have recovered. 58,000,000 filed for unemployment as of 9/2020. Some of these deaths labeled as due to corona virus are actually due in large part due to other contributing causes but the patient ALSO had a corona virus infection and so is counted as a corona virus death, even though the virus may not be the dominant cause of death.
It is likely that CoV2-19 has CONTRIBUTED to an overall increased mortality of 20%, the 7/1/20 JAMA reported increase in overall mortality was 28%:
Oral drug blocks SARS-CoV-2 and influenza transmission
by Georgia State University 12/3/20
Treatment of SARS-CoV-2 infection with a new antiviral drug, MK-4482/EIDD-2801 or Molnupiravir, completely suppresses virus transmission in ferrets within 24 hours, researchers in the R Plemper @ the Institute for Biomedical Sciences at Georgia State University have discovered. “This is the first demonstration of an orally available drug to rapidly block SARS-CoV-2 transmission,” said Plemper. “MK-4482/EIDD-2801 could be game-changing.”
MK-4482/EIDD-2801 has broad-spectrum activity against respiratory RNA viruses and that treating infected animals by mouth with the drug lowers the amount of shed viral particles by several orders of magnitude, dramatically reducing transmission,” said Plemper. COVID-19 patients treated with the drug could become non-infectious within 24 hours after the beginning of treatment. It causes a 50 % reduction in hospitalizations and deaths.
Advanced photon source helps Pfizer create COVID-19 antiviral treatment: PAXLOVID
by Argonne National Laboratory 11/8/21
The IMCA-CAT beamline at the Advanced Photon Source, where work was done to determine the structure of Pfizer’s new COVID-19 antiviral treatment candidate.
IMCA-CAT/Hauptman-Woodward Medical Research Institute
Pharmaceutical company Pfizer has announced the results of clinical trials of its new oral antiviral treatment against COVID-19. The new drug candidate, Paxlovid, proved to be effective against the SARS-CoV-2 virus, which causes COVID-19, according to results released by Pfizer on 11/5/21.
Scientists at Pfizer created Paxlovid with the help of the ultrabright X-rays of the Advanced Photon Source (APS), a U.S. Department of Energy (DOE) Office of Science user facility at DOE’s Argonne National Laboratory. …
IMCA-CAT uses the X-rays of the APS to probe the atomic structures of proteins, like those that make up the SARS-CoV-2 virus. Scientists use this information to see how potential drug compounds may interact with the virus.
If approved, Paxlovid would be the first oral antiviral specifically designed to inhibit a key protease (3CL) of the SARS-CoV-2 virus. For the latest phase of clinical trials, Pfizer tested the treatment on more than 1200 adults, all of whom had been diagnosed with COVID-19 no more than five days prior, and had at least one medical condition or characteristic that enhanced the risk of severe illness.
Results reported by Pfizer showed an 89 percent reduction in risk of COVID-19-related hospitalization or death, compared with a similar number of patients who took a placebo.
Results of the first phase of trials were recently published in Science.
“This is excellent news, and we’re very pleased to have played a part in the creation of this potentially life-saving antiviral treatment candidate,” said Stephen Streiffer, Argonne’s deputy laboratory director for science and technology and director of the APS.
More information: Dafydd R. Owen et al, An oral SARS-CoV-2 M pro inhibitor clinical candidate for the treatment of COVID-19, Science (2021). DOI: 10.1126/science.abl4784
Robert M. Cox et al. Therapeutically administered ribonucleoside analogue MK-4482/EIDD-2801 blocks SARS-CoV-2 transmission in ferrets, Nature Microbiology (2020). DOI: 10.1038/s41564-020-00835-2
mainternalmedicine/fullarticle/2767980?guestAccessKey=8d5b07ea-8aed-434b-86e7-e2cbdf11b9d4&utm_source=silverchair&utm_medium=email&utm_campaign=article_alert-jamainternalmedicine&utm_term=mostread&utm_content=olf-widget_09182020
Worldwide as of 9/16/21 there are 228,000,000 reported corona virus/CoV2-19 cases with 4,700,000 deaths 9/16/21 and 205,000,000 recovered: 25% of cases are asymptomatic, 5% of cases are labelled as “SERIOUS”, 0.37% die = 4 million deaths. Documented risk factors for developing CoV2-19 are inter- or multi-generational living (together), obesity, diabetes, hypertension, age, higher population density, air pollution, and asthma, … which are more prevalent in the African American and Latino communities. Europe, as the EU = European Union, which is about the same size and population as the USA, is has similar TOTAL CASES but Has more total deaths due to the corona virus. As of 9/16/21 Europe/the European Union has 38,000,000 known corona virus cases with 760,000 deaths. Peru, Brazil, and Belgium have the highest frequency of cases on a population basis. The USA 9/16/21 has 43,000,000 cases: 700,000 deaths, 33,000,000 recovered & the current reported death rate was 3.4 -> is 1.3 % (actually 0.37%) vs 1.5% in South Korea. On a per 100,000 population, the USA mortality is EIGHTH in the world, less than Italy, Spain, France, Switzerland, and the Netherlands. Sweden, with much less lockdown, has the same mortality rate as the USA and is SIXTH in Europe for mortality. A German report based on ANTIBODIES, states that “One in Seven May Be Immune”: that reduces COVID lethality from 2% to 0.37 %, still nearly 4 times that of the current flu. Early on, most corona virus-19/CoV2-19 cases in the USA were elderly nursing home residents in Washington state. Now New York state leads, followed by New Jersey. Repeating, in the USA there are 500,000 FLU hospitalizations and 35,000 flu deaths per YEAR. In the 2019-2020 flu year there are 45,000,000 cases of the flu 43,000 deaths according to the CDC. Both the flu and CoV2-19 cause PNEUMONIA, “the old man’s friend”, and that is the usual cause of death for CoV2-19. It can affect the heart and elsewhere. 7,500 Americans die of all causes every DAY normally.
Here is an article that discusses the POSSIBLE need for 44 days of LOCKDOWN to DEFEAT CoV2-19. This below article does NOT TAKE INTO ACCOUNT the diagnosis and treatment developments that are rapidly happening in the USA: ANTIBIOTICS, CELLULAR ALKALINIZATION, IMMUNE ENHANCEMENT, TRANSFUSED ANTBODIES, newly developed monoclonal antibodies (file:///C:/Users/hrobe/AppData/Local/Temp/s41423-020-0426-7.pdf) , VACCINES, and INTERFERING RNA, etc. Gerard J. Tellis et al. “How Long Should Social Distancing Last? Predicting Time to Moderation, Control, and Containment of COVID-19”, SSRN Electronic Journal (2020). DOI: 10.2139/ssrn.3562996
Here is the plan for how Germany plans to REOPEN the country after Covid-19. See also the plan outlined below with the associated table: https://www.ifo.de/en/publikationen/2020/monograph-authorship/making-fight-against-coronavirus-pandemicsustainable
A COUGH can send infected droplets 15 feet. A strong SNEEZE can send infected droplets 25 feet. The virus can live in the air for three hours, on wet surfaces for three days, 24 hours on cardboard, and 3 days on plastic: after 45 minutes the viral count is reduced by half on copper. The half-life of the virus in infected droplets is 5 hours on stainless steel. The virus count decreases by half every 7 hours on plastic so that by day 2 there is only 1/100th of the original viral count on plastic.
In China, with its strong QUARANTINE and ISOLATION procedures, new cases have REPORTEDLY slowed to a trickle. This is exactly similar to the reaction of certain, but not all, USA cities during the 1918 SPANISH FLU that killed millions. USA cities that most effectively “locked down” with what we now call “social isolation” had the best health and economic recoveries then. There is a major difference: once widespread testing, detection, isolation, and treatment begin, such isolation will be much less necessary in the USA. Presently, South Korea has been the best and most effective country in dealing with this infection by using strong QUARANTINE and GPS TRACKING of contacts: “acceptance of (relevant public) surveillance” is the key. Their success occurred with high frequency testing of the public, tracing of contacts of those who are test-positive, and treating based on risk profile.
Diagnosing COVID-19 in just 30 minutes
by Pohang University of Science & Technology (POSTECH) 10/5/20
The reaction is composed of four main components: a set of probes, SplintR ligase, T7 RNA polymerase and a fluorogenic dye. In the presence of target RNA, hybridization, ligation, transcription and aptamer-dye binding reactions occur sequentially in a single reaction tube at a constant temperature.
The team: Professor Jeong Wook Lee and Ph.D. candidate Chang Ha Woo and Professor Gyoo Yeol Jung and Dr. Sungho Jang of the Department of Chemical Engineering at POSTECH developed a SENSR (SENsitive Splint-based one-pot isothermal RNA detection) technology that allows anyone to easily and quickly diagnose COVID-19 in 30 minutes based on the RNA sequence of the virus.
A diagnostic kit can be developed within weeks even if a new infectious disease appears other than COVID-19.
Using this technology, the research team found the SARS-CoV-2 virus RNA, the cause of COVID-19, from an actual patient sample in about 30 minutes. In addition, five other pathogenic viruses and bacterial RNAs were detected which proved the kit’s usability in detecting pathogens other than COVID-19.
Covid-19 contact tracing system with roots in MERS
Reuters 4/15/20: What distinguishes the Korean model in controlling COVID-19 is its ability to trace individuals diagnosed with the disease who may have come into contact with the infected individuals. It’s known as the COVID-19 Smart Management System (SMS).
South Korea’s Centers for Disease Control and Prevention (KCDC) runs the contact tracing system that uses data from 28 organizations such as National Police Agency, The Credit Finance Association, three smartphone companies, and 22 credit card companies to trace the movement of individuals with COVID-19. This system takes 10 minutes to analyze the movement of the infected individuals. For people who come in contact with an infected person, the KCDC informs the local public health center near the infected citizen’s residence and the health center sends the notification to them. If they test positive, they are hospitalized at the COVID-19 special facilities. Those without symptoms are asked to remain self-quarantined for 14 days.
The legal basis for accessing such personal information was prepared after the 2015 MERS outbreak when the government learned that tracing the movement of infected individuals and people who came in contact with them is crucial. As a safety measure, only epidemic investigators at KCDC can access the location information and once the COVID-19 outbreak is over, the personal information used for the contact tracing will be purged.
Israel has developed such a tracking app called Hamagen. Israel has developed an app to monitor when a person’s home and reduce the need to be visually evaluated by a person: This increases quarantine efficiency 30 times. 11/5/20 Brian Subirana of MIT has developed an AI cell phone cough sensing app that can ID 100 % of ASYMPTOMATIC CoV2-19 carriers and 97 % of those with symptoms. 4/13/20 Apple and Alphabet’s Google will work together to create contact tracing technology in order to slow the spread of the coronavirus by allowing users to opt into a system that catalogs other phones they have been near. The 2 companies make the world’s dominant smartphone operating systems for iPhones and Android devices. This allows mobile devices to trade information via Bluetooth connections to alert people when they have been in close proximity with someone who has tested positive for COVID-19.
The technology will not track location or identity, but instead will only capture data about when users’ phones have been near each other, with data being decrypted on the user’s phone rather than the companies’ servers. GPS location data is not part of the effort.
Prime Minister Jacinda Ardern said NEW ZEALAND will continue to pursue its goal of elimination with a strategy that differs from most other countries. A few recent CoV2-19 recurrences have resulted in the lockdown of Auckland, a city of over 1 million: This seems overly harsh New Zealand would profit immensely by using the 8/2020 Israeli app reducing the need for direct observation/contact tracers being vastly reduced while also allowing greater freedom for those who have been checked and found to be negative.
4/29/20 Newsletter Science X: Yale tracking method: 4/29/20l Nature, differs from existing epidemiological models by exploiting real-time data about population flows, such as phone use data and other “big data” sources that can accurately quantify the movement of people. … “very accurately forecast the timing, intensity, and geographic distribution of the COVID-19 outbreak based on population movement alone,” said Yale’s professor N. A. Christakis. “…, by tracking population flows in real time, our model can provide policymakers and epidemiologists a powerful tool to limit an epidemic’s impact and save lives.” In developing the model, the researchers used nationwide mobile-phone geo-location data to track.
“Success doesn’t mean zero COVID-19 cases. It means zero tolerance, which means that as soon as we know we have a case, we go in straight away, we’re testing around that person, we’re isolating them […] we do our interviews and contact trace to find all the people who have been in contact with them while they may have passed it on, and we ask them to isolate. That’s how we keep stamping out COVID cases.” FLUTRACKER & Singaore’s TRACE TOGETHER apps are being used for this purpose.
6/19/20: “The German government has stuck to its course of gradually reopening the country while seeking to clamp down swiftly on localized outbreaks. A free app launched Tuesday to help trace people who may have been exposed to the virus has already been downloaded 9.6 million times in Germany, which has a population of 83 million. Japan released a similar app Friday, also using technology developed by Apple and Google. Singaporeans were able to wine and dine at restaurants, work out at the gym and socialize with up to five people at a time as of Friday, after the city-state removed most of its pandemic lockdown restrictions. Switzerland announced Friday that gatherings of up to 1,000 people would be permitted next week, in a fourth stage of gradually easing the restrictions imposed to control COVID-19.
Dr. Hillel Kashtan, created the app MDHEALTHTRAK to track various illnesses & now Co V2-19. Not only does it monitor symptoms of the virus, he said, but using the app cuts down on person-to-person contact. For protection, “the physician can assess the patient at home, so others are protected.” The app creates charts for all kinds of symptoms and tracks how those symptoms change over time. That information can then be sent to doctors to allow them to easily see the changes.
Oxford University’s Professor Christopher Fraser developed a similar app that can also assist in reducing transmission and resurgence of CoV2-19 infections.
South Korea has drive-through testing which is ramping up in the USA. Unfortunately, South Korea, Hong Kong and Taiwan are seeing a SECOND WAVE of CoV2-19 infections as infected returnees come back to these areas from elsewhere. False negative testing and reinfection may be possibilities. Sweden is not doing our strong social distancing.
An excellent WEBSITE to follow the virus worldwide is by 17-year-old self-taught prodigy Avi Schiffman: http://ncov2019.live/data . Johns Hopkins University website is also excellent. https://coronavirus.jhu.edu/map.html The website www.bing.com has excellent data. HARVARD has jut (6/2020) produced a MAP that that shows the frequency of CoV2-19 throughout the USA, INLUDING EACH SPECIFIC COUNTY: https://globalepidemics.org/ For optimism, check out the twitter of a garbage man whose handle I lost: it had 3 letters in caps at the end. Another fine source of information & perspective on CoV2-19 is Harvard’s infectious disease specialist Dr. Lindsey R. Baden of Brigham and Women’s Hospital.
7/21/20 JAMA Network Tyler S Brown: “The estimated total number of infections (extrapolated from the measured proportion of individuals with positive SARS-CoV-2 serologies) was between 6- and 24-fold higher than the number of confirmed COVID-19 cases reported in each location prior to the study.”
Here is a link to what life was like in Wuhan, China during its lockdown:
https://www.quora.com/What-is-it-like-inside-the-quarantine-zone-in-Wuhan-City
A rather remarkable interview with world famous VIROLOGIST David Ho, MD
https://medicalxpress.com/news/2020-03-iceberg-virologist-discusses-covid-.html?utm_source=nwletter&utm_medium=email&utm_campaign=daily-nwletter
Here is a very MODERATE, EVENHANDED, optimistic yet sober STATISTICIAN’s perspective on this corona CoV2-19 virus:
https://www.powerlineblog.com/archives/2020/03/a-data-driven-look-at-the-wuhan-coronavirus.php
A GENETICIST discusses what a virus is, what it does inside a cell, and what CoV2-19 is.
https://medicalxpress.com/news/2020-03-covid-virus.html?utm_source=nwletter&utm_medium=email&utm_campaign=daily-nwletter
Stanford University EPIDEMIOLOGIST John IOANNIDIS, MD, has published a profound article that says that the USA and all other countries simply lack reliable evidence to draw accurate conclusions regarding the seriousness of CoV2-19 infections. This is because the vast majority of cases are MISSED due to limited testing availability of the general public so far. He states that “short term lockdowns may be bearable” with the implication that long-term lockdowns likely will not be tolerable because of “profound financial and social consequences”. I believe he is exactly correct. Douglas McKenzie described the current lockdown effects as “wholesale disruption of the American social fabric and its vibrant economy.”
7/2020: 10 times faster = 560/hour testing: “thermocycler (NEXTGENPCR)” developed by the Dutch company Molecular Biology Systems B.V. 7/20/20 Medical Xpresss: World-first research by Monash University in Australia has been able to detect positive COVID-19 cases using blood samples in about 20 minutes, and identify whether someone has contracted the virus.
New negative pressure ventilator requiring fewer staffing resources developed in fight against COVID-19
by AAGBI 1/20/21
A new negative pressure ventilator which could provide additional treatment options for patients with respiratory failure, including those with COVID-19—and whose design can be easily adapted to developing countries—has been created by a team that includes anaesthetists, nurses and engineers. Details on the new exovent system—which is similar in design but much smaller in scale and easier to use than the devices used to help treat polio patients during the 1950s—are published in Anaesthesia.
Use of this system would offer more comfort to patients, who would not need to be asleep or have an artificial airway in place would also mean less nursing care and could be used anywhere in the hospital, and even potentially at home. Co-author Dr. Malcolm Coulthard of the Translational and Clinical Research Institute, Newcastle University, UK.
Negative pressure is far less intrusive and much more like normal breathing than either positive pressure ventilation through a tube inserted into the windpipe, or delivering CPAP via a tightly-fitting face mask. The exovent system is non-invasive, which means that patients do not need to have their windpipes intubated, so they don’t need to be anaesthetised and oxygen can be delivered in the form of a normal oxygen mask or nasal prongs rather than through a high flow oxygen device that puts hospital oxygen supplies under pressure. Patients remain conscious, and can take food and medication by mouth, and talk to loved ones on the phone. Prototyping by Marshall ADG, and partnership with Warwick Manufacturing Group (WMG) High Value Manufacturing Catapult, the system. Journal information: Anaesthesia
In the American population there are 950,000 HOSPITAL BEDS, 45,000 ICU beds and
150,000 available VENTILATORS. Ventilators have 150 parts and those new to that manufacturing will need to become expert in production of all. Continuous positive airway Pressure = CPAP is a halfway step developed by the Mercedes F1 racing team that can reduce the need for ventilators. “…low sat’ “happy hypoxics” comfortably walking around did well with just high flow nasal cannulas and had no need for ventilators. Critical care physicians are questioning the widespread use of the breathing machines for Covid-19 patients, saying that large numbers of patients could instead be treated with less intensive respiratory support. Ventilators could be of little benefit to many and even harmful to some. Many patients have blood oxygen levels so low they should be dead. But they’re not gasping for air, their hearts aren’t racing, and their brains show no signs of blinking off from lack of oxygen.”
More patients could receive simpler, noninvasive respiratory support, such as the breathing masks used in sleep apnea. An oxygen saturation rate below 93% (normal is 95% to 100%) has long been taken as a sign of potential hypoxia and impending organ damage. Because some patients with Covid-19, blood-oxygen levels fall to hardly-ever-seen levels, into the 70s and even lower, physicians were intubating them sooner.
“Most hospitals, including ours, are now using simpler, noninvasive strategies first,” including the apnea devices and even nasal cannulas, “It doesn’t require sedation and the patient [remains conscious and] can participate in his care.”
As patients get worse, protocols developed for other respiratory conditions call for increasing the force with which a ventilator delivers oxygen, the amount of oxygen, or the rate of delivery. But if oxygen can’t cross into the blood from the lungs in the first place, those measures, especially greater force, may prove harmful. High levels of oxygen impair the lung’s air sacs, while high pressure to force in more oxygen damages the lungs.
Physicians in Germany and Italy said their Covid-19 patients were unlike any others with acute respiratory distress. Their lungs are relatively elastic (“compliant”), a sign of health “in sharp contrast to expectations for severe ARDS.” Their low blood oxygen might result from things that ventilators don’t fix. Such patients need “the lowest possible [air pressure] and gentle ventilation,” they said, arguing against increasing the pressure even if blood oxygen levels remain low. “We need to be patient.”
In the Annals of Intensive Care, physicians who treated Covid-19 patients in China found that the majority of patients needed no more than a nasal cannula. With BiPAP oxygen levels “significantly improved” after an hour or two. The researchers concluded that the more comfortable nasal cannula is just as good as BiPAP and that a middle ground is as safe for Covid-19 patients as quicker use of a ventilator. Helmet CPAP is a newer design of respiratory assistance to make the more troublesome inspiratory effort easier.
Questioning the significance of oxygen saturation levels: those levels often “look beyond awful,” said Scott Weingart, MD, in New York/host of the “EMCrit” podcast. But many can speak in full sentences, don’t report SHORTNESS OF BREATH, and have no signs of the heart or other organ abnormalities that hypoxia can cause.
“The patients in front of me are unlike any I’ve ever seen,” Kyle-Sidell told Medscape about those he cared for in a hard-hit Brooklyn hospital. “They looked a lot more like they had altitude sickness than pneumonia.” Anecdotally, Weingart said, “we’ve had a number of people who improved and got off CPAP or high flow [nasal cannulas] who would have been tubed 100 out of 100 times in the past.”
One reason Covid-19 patients can have near-hypoxic levels of blood oxygen without the usual gasping and other signs of impairment is that their blood levels of carbon dioxide, which diffuses into air in the lungs and is then exhaled, remain low. That suggests the lungs are still accomplishing the critical job of removing carbon dioxide even if they’re struggling to absorb oxygen, REMISCENT OF ALTITUDE SICKNESS more than pneumonia.
Lowcost compact ventilators designed by Southern Miss from hardware store parts now produced by Howard Industries can alleviate shortages and be used in various settings because of its size and ease of use. “This new bag-based ventilator could be produced very quickly,” said Dr. Joe Campbell, Forrest General’s chief anesthesiologist.
Adapt sleep apnea machines: 4/8/20 scientists have developed a way to turn a sleep apnea machine into a ventilator to treat people with COVID-19. The modification of a Nippy3+ began at Leeds Teaching Hospitals at the University of Leeds. The modification is straightforward and involves changes to the device’s settings and reconfiguring the supply of oxygen so it flows to the face mask worn by the patient. The machine operates in a mode called CPAP: constant positive airway pressure. That means the pressure inside the mask is slightly raised, keeping the patient’s airway open and making it easier for them to breathe. It provides enriched oxygen of between 40 to 60 percent and because it is a modification to a device, it does not have to go through a full regulatory approval process. Last week, engineers at University College London and Mercedes announced that they had successfully reversed engineered a CPAP device that had widely been used in China. They said they have a device that has regulatory approval and can be rapidly manufactured. More information: A technical note written by the expert team has been submitted to MedRxiv, an online platform that allows researchers to rapidly disseminate important findings ahead of peer review. The technical note can be downloaded from AlphaGalileo: www.alphagalileo.org/DTControl … ntrol/Images/pdf.png
A PROFOUND article on RESPIRATORY MANAGEMENT from J J Marini of the U of Minn in JAMA Insight 4/24/20
https://jamanetwork.com/journals/jama/fullarticle/2765302?guestAccessKey=e0b408ba-3e6b-4d40-83bb-9e7dd9575c24&utm_source=silverchair&utm_campaign=jama_network&utm_content=covid_weekly_highlights&utm_medium=email
Based on a robust body of clinical evidence, including studies published in the New England Journal of Medicine, a TIDAL VOLUME of around 6 cc per kilogram of patient body weight is the general standard of care for patients with Acute Respiratory Distress Syndrome/ARDS as part of “lung-protective” ventilation. SNORKLE/diving MASKS are being adapted as respirators. Prone positioning & inhaled nitric oxide are recommended, but not steroids. However, some are using 60 mg/day PREDNISONE for 3 days, 40 mg for 2 days, and 20 mg for 1 day-steroids do reduce type1 interferon anti-viral response. See below for the DECADRON “RECOVERY” study for ventilator requiring disease. MIT created an inexpensive and simple respirator using Ambu bags. A very experienced respirator designer puts already FDA approved parts together to make a simple respirator that avoids VILY = Ventilator Induced Lung Injury—yet to be cleared by the FDA:
https://www.facebook.com/brent.regan.370/videos/1333250890200601/
COMING OFF THE VENTILATOR can be problematic. In a 6/4/20 it is reported that 60% of those put on ventilators will survive to discharge from the hospital. In 2017 FDA approved SUGAMMADEX which reverses the effect of muscle relaxants differently. In a new U of Michigan study, Kheterpal et al. compared the rates of serious lung complications in patients who received neostigmine vs sugammadex. The newer drug was associated with significantly reduced rates of complications.
“We saw a 37% decrease across all pulmonary (fibrosis, scarring, and detritus-filled lungs) complications and 55 % decrease in respiratory failure,” said Kheterpal. “In many practices, neostigmine is no longer used in high risk patients or procedures,” he said.
More information: Sugammadex versus Neostigmine for Reversal of Neuromuscular Blockade and Postoperative Pulmonary Complications (STRONGER): A Multicenter Matched Cohort Analysis. Anesthesiology. (2020) DOI: 10.1097/ALN.0000000000003256
“Cough Sync” is a newly developed tool for aspirating thick lung secretions more effectively. 85% of medicines/pharmaceuticals are manufactured in China and India; all of USA’s required rare earth metals for manufacturing come from China.
TRAVEL bans have been set up by Saudi Arabia, Russia, Poland, Kenya, Morocco, Argentina, Brazil, Canada, Denmark, the Netherlands, Germany, the European Union, and many other countries as well as quite appropriately, the USA beginning on 1/31/20. That quarantine was supported by the NIH’s infectious disease chief, Dr. A. Fauci.
At the present time almost all of us feel OFF BALANCE because of the inability to find out if we are (+) or (-) for the CoV2-19. There has been a general LOSS OF JOY across the United States. Being “shut-ins” has led to “cabin fever.” The stock market is responding to FUD: fear, uncertainty, doubt—all of which translates as ANXIOUS UNCERTAINTY. And although I believe allowing oneself to panic is largely a personal responsibility, the nationwide information atmosphere seems responsible for predisposing the susceptible to panic. A psychiatrist trenchantly said this national stressor will make “those not well put together, go over the edge”. In balance, it must also be asked what will be the psychological and economic cost of not returning to our more normal lives sooner than the various quarantines permit? Domestic violence, drug usage, worsening diet, weight increase, and suicide will increase. People will eventually adjust out of reason and/or necessity. It is important to be careful, but not to be paranoid. Humility and perseverance are the keys to dealing with these stressors.
Here is a perspective on how the economy could reasonably be opened based on being low risk:
Very few Americans are dying to date from the coronavirus who had no pre-existing condition. 150 Americans to Date with NO Pre-existing conditions have died from the coroana virus. by Jim Hoft 4/10/20
As of 4/10/20, there are now 16,697 Americans who have died having the corona virus, 96,000 worldwide.
The most recent data shows that only 0.9% of deaths related to the corona virus are related to individuals with no comorbidity (i.e. any pre-existing condition):
Based on this data, 150 Americans have died from the corona virus who had no pre-existing conditions out of 16,697.
In addition, of the top 29 countries in the world based on number of corona virus cases confirmed, eight of these countries have opened their countries up economically in some part or in full. (See countries highlighted above in yellow: China, Brazil, Austria, Sweden, Norway, Denmark, Czechia and Japan.) The death rate per case identified for these countries is 4.1% which is less than the overall world average of 6% but the US death rate was reported at slightly lower than both at 3.6% of identified cases.
The US has shut down its economy because of the corona virus based on 17,000 deaths (less than this year’s flu related deaths at that point). Those people could likely be allowed to safely return to work now.
Early on the CDC (Centers for Disease Control) did not allow TESTING development outside of its requirements. The recent CDC tests had a technical flaw and proved unreliable. Independent test development by D S Chugh, MD, of Washington state allowed the recognition of the first USA case of coronavirus 19/CoV2-19 on 1/21/20 which, remarkably, was in a teenager–as it was felt then and afterwards that those of that age group were relatively immune to the serious consequences of corona 19/CoV2-19. Despite initially being held back by CDC regulations, she eventually decided on her own correctly to develop accurate testing by NOT adhering to the guidelines. The CDC & FDA for quite a while were reported to still be slowing acceptance and release of new innovative testing kits and those for home testing. WHO test kits were made available to lower income countries without testing capability, not the USA. Better PREPARATION for this current viral pandemic after the much more dangerous 2003 SARS, 2007 Zika, 2014 Ebola, and 2012 MERS crises could have been accomplished. The federal Pandemic Office was not eliminated as some suggest, but was merged with other governmental groups.
A $50 sensitive smartphone accessory was developed at U of Illinois at Urbana-Champaign by professors Brian Cunningham and Rashid Bashir was licensed to Reliant Immune Diagnostics and reported in the journal Lab on a Chip.
Israeli firm developing 30-second coronavirus breath test
Medical Xpress 7/24/20 by Jonah Mandel
An Israeli company hss developed a coronavirus NanoScent breathalyser gives results in 30 seconds with 85 % accuracy, a “front line” tool that can help restore a sense of normality during the pandemic. Test subjects inhale through the nose, hold their breath, close one nostril and exhale through the other, pushing breath through a handheld tube into a small bag called the “Air Trap”. It is a mass screening toolhe tube that is plugged into the “Scent Reader”, a small rectangular device that whirrs softly as it sucks the air out of the bag. Within seconds the results—”COVID-19 negative” during AFP’s visit—appear on the phone.The device will likely cost less than $10 per test, “a fraction of the cost of the lab test”, Gavriely said.
An early TESTING BREAKTHROUGH: Abbott’s SARS-CoV-2 IgG test identifies the IgG antibody, which is a protein that the body produces in the late stages of infection and may remain for up to months and possibly years after a person has recovered. Abbott’s IgG antibody test is available on its ARCHITECT® i1000SR and i2000SR laboratory instruments*. More than 2,000 of these instruments are in use in U.S. laboratories. These instruments can run up to 100-200 tests per hour.
False-Negative Rate of RT-PCR SARS-CoV-2 Tests !!!!
May 18, 2020 This is important!!
Kucirka LM, Lauer SA, Laeyendecker O, Boon D, Lessler J. Variation in False-Negative Rate of Reverse Transcriptase Polymerase Chain Reaction–Based SARS-CoV-2 Tests by Time Since Exposure. Ann Intern Med 2020;May 13:[Epub ahead of print]. Tests for SARS-CoV-2 based on RT-PCR add little diagnostic value in the first 5 days immediately after exposure. Decisions regarding removing contact precautions or ending quarantine should not be made on the basis of results obtained in the first 5 days post-exposure and absence of symptoms. Serial testing may improve test performance
Only confirmed cases and studies in which samples were collected from the upper respiratory tract (nasopharyngeal and oropharyngeal) were included. A Bayesian hierarchical model was fitted to estimate the false-negative rate by day since exposure and symptom onset. The model assumed a specificity of 100% for the RT-PCR, and a 5-day incubation period for the virus. Over the 4 days of infection before the typical time of symptom onset (day 5), the probability of a false-negative result in an infected person decreased from 100% (95% confidence interval [CI], 100%-100%) on day 1 to 67% (CI, 27%-94%) on day 4. On the day of symptom onset, the median false-negative rate was 38% (CI, 18%-65%). This decreased to 20% (CI, 12%-30%) on day 8 (3 days after symptom onset) then began to increase again, from 21% (CI, 13%-31%) on day 9 to 66% (CI, 54%-77%) on day 21. The false-negative rate was minimized 8 days after exposure—that is, 3 days after the onset of symptoms on average.
Similarly, J Zhao in 2020 Clin Infectious Disease found that “… in days 1 through 7 after onset of illness, 11% of sputum, 27% of nasal, and 40% of throat samples were deemed falsely negative. Zhao studied 173 hospitalized patients with acute respiratory symptoms and a chest CT “typical” of Covid-19, or SARS-CoV-2 detected in at least one respiratory specimen. Antibody seroconversion was observed in 93%….” as reported in the NEJM 2020.
POOLED TESTING: is testing small groups, called pools, using only one test. More people can be tested faster, using fewer tests, and for less money.
Instead of testing one person at a time, samples from multiple individuals would be mixed together and tested as one. If the test comes back negative, everyone in the pool is clear. If positive, each member of the pool is then tested individually. This is cost effective if less than 15% of people will be found to be positive. That is MOST of the USA, even most “hot spots.”
NEJM 10/2020: “…LY-CoV555 (also known as LY3819253), a potent antispike neutralizing monoclonal antibody that binds with high affinity to the receptor-binding domain of SARS-CoV-2, was derived from convalescent plasma obtained from a patient with Covid-19. The antibody was developed by Eli Lilly after its discovery by researchers at AbCellera and at the Vaccine Research Center of the National Institute of Allergy and Infectious Diseases. The discovery of LY-CoV555 and its passive protection against SARS-CoV-2 in nonhuman primates has been reported previously.19….”
5/5/20 Israel Defense Minister Naftali Bennett on Monday discussed a “significant breakthrough” by Israel’s Israel Institute for Biological Research (IIBR) in its developing an antibody to COVID-19/CoV2-19. The “antibody attacks the virus in a monoclonal way and can neutralize it within the bodies of those ill.” The researchers finished the development phase.
Subcutaneous REGEN-COV Antibody Combination to Prevent Covid-19
List of authors.
• Meagan P. O’Brien, MD, et al
• for the Covid-19 Phase 3 Prevention Trial Team*
REGEN-COV (previously known as REGN-COV2), a combination of the subcutaneous monoclonal antibodies casirivimab and imdevimab, has been shown to markedly reduce the risk of hospitalization or death among high-risk persons with coronavirus disease 2019 (Covid-19).
Participants (≥12 years of age) were enrolled within 96 hours after a household contact received a diagnosis of SARS-CoV-2 infection to receive a total dose of 1200 mg of REGEN-COV. The primary efficacy end point was the development of symptomatic SARS-CoV-2 infection through day 28 in participants who did not have SARS-COV-2 infection (as measured by reverse-transcriptase–quantitative polymerase-chain-reaction assay) or previous immunity (seronegativity).
Symptomatic SARS-CoV-2 infection developed in 11 of 753 participants in the REGEN-COV group (1.5%) and in 59 of 752 participants in the placebo group (7.8%) (relative risk reduction 81.4%; P<0.001). 2 of 753 participants in the REGEN-COV group (0.3%) and 27 of 752 participants in the placebo group (3.6%) had symptomatic SARS-CoV-2 infection (relative risk reduction, 92.6%). REGEN-COV also prevented symptomatic and asymptomatic infections overall. Among symptomatic infected participants, the median time to resolution of symptoms was 2 weeks shorter.
Abbott is significantly scaling up its manufacturing for antibody testing and is expecting to immediately ship close to 1 million tests this week to U.S. customers, and will ship had 4 million tests. The company ramped up to 20 million tests in the U.S. in June and beyond as it expanded tests to run on its new Alinity™ i system. Abbott also will be expanding its laboratory antibody testing to the detection of the antibody, IgM, in the near future.
Lab-on-a-chip COVID-19 antibody test could offer rapid, portable, low cost, and accurate results.
From Newsletter X: Optofluidic Bioassay at the U of Michigan: a microfluidic device shrinks multiple lab functions onto a single chip just millimeters or centimeters in size: faster results with smaller sample sizes. It is “enzyme-linked immunosorbent assay,” or ELISA. The U-M researchers have previously published results showing that their device can work as well as the slower, larger, standard ELISA set-up. Anyone working on COVID-19 antibody tests can use their reagents in this device.
ELISA tests are typically quantitative and accurate, showing the concentration of antibodies. That makes them more reliable and less prone to false positives than the rapid diagnostic tests. But standard ELISA results take several hours, and the machines that provide them are the size of refrigerators. In addition, the sample needs to be sent to the test lab for analysis. But microfluidic ELISA can give a quantitative and accurate result in just 15 minutes, with a finger-prick’s worth of blood.
This technique can monitor patients’ immune response to infection, treatment, and vaccination. The estimated cost of testing is a few dollars per test of 2 to 3 different antibodies. The machine can be the size of a microwave, and can test multiple simultaneous samples of little more than a drop of blood from a fingertip in 15 minutes.
One vaccine dose enough for COVID-19 survivors
by American Chemical Society 6/23/21
Two mRNA vaccines against COVID-19 have proven safe and effective in clinical trials, as well as in the millions of people who have been vaccinated so far. But how prior SARS-CoV-2 infection affects vaccine response, and how long that response lasts, are still uncertain. Now, a new study in ACS Nano supports increasing evidence that people who had COVID-19 need only one vaccine dose, and that boosters could be necessary for everyone in the future.
In clinical trials, the Pfizer-BioNTech and Moderna COVID-19 vaccines were about 95% effective in protecting against symptomatic infections. Both mRNA vaccines trigger the immune system to produce antibodies against the SARS-CoV-2 spike protein receptor binding domain (RBD), and two doses are necessary to provide immunity in people who haven’t previously had COVID-19.
However, the clinical trials included very few people who had already recovered from the disease, so the immune response of these individuals is less well known. Also, the time course of antibody development in both groups, and how long virus-neutralizing antibodies persist, haven’t been well characterized. So, Otto Yang and colleagues wanted to compare antibody levels, quality and persistence after one and two doses of mRNA vaccine in people with or without prior SARS-CoV-2 infection.
The researchers used an enzyme-linked immunosorbent assay (ELISA) to measure antibodies against the RBD in people who received the Pfizer-BioNTech or Moderna vaccine, and in unvaccinated people soon after mild or severe COVID-19 cases. In the 28 participants without prior infection, one dose of either vaccine triggered antibody levels similar to those seen after mild COVID-19 infections, whereas two doses were required to obtain anti-RBD antibodies approaching those observed after severe cases.
In contrast, in 36 participants who had COVID-19 prior to vaccination, the first dose produced a vigorous antibody response similar to severe natural infection, but the second dose provided no additional increase in antibody levels. The quality of antibodies, indicated by their ability to neutralize the SARS-CoV-2 spike protein and their potency, followed similar patterns. After the second vaccine dose, antibody levels waned in both groups comparably to natural infection, resulting in an average loss of 90% within 85 days. Although more research on T cell responses to the vaccines is needed, this result suggests that booster vaccinations will likely be required for everyone, the researchers say.
More information: F. Javier Ibarrondo et al, Primary, Recall, and Decay Kinetics of SARS-CoV-2 Vaccine Antibody Responses, ACS Nano (2021). DOI: 10.1021/acsnano.1c03972
New COVID-19 test results in 45 minutes: 4/16/20 : The CRISPR-based test, SARS-CoV-2 DETECTR, uses gene-targeting technology, requires no specialized equipment, and is published in 4/16/20 Nature Biotechnology by developer Dr. Charles Chiu of UCSF. The test targets any genetic sequence, so test developers “programmed” it to find 2 sequences in the genome of SARS-CoV-2, the cause of COVID-19. One sequence is common to all SARS-like coronaviruses, while the other is unique to SARS-CoV-2. Checking both sequences ensures that the new test can distinguish between SARS-CoV-2 and related viruses. The test can detect coronavirus in samples from respiratory swabs, provides results in about 45 minutes, can be performed in virtually any lab using off-the-shelf chemical agents and common equipment, and it is easy to interpret. Much like a store-bought pregnancy test, dark lines appear on test strips. PCR-based tests require specialized equipment, limiting them to well-equipped diagnostic labs. More information: CRISPR–Cas12-based detection of SARS-CoV-2, Nature Biotechnology (2020). DOI: 10.1038/s41587-020-0513-4 , https://www.nature.com/articles/s41587-020-0513-4
CRISPR Cell 2020 Dec 04;[EPub Ahead of Print], P FozouniR and Mobile Phone Microscopy to Detect SARS-CoV-2
Cell 2020 Dec 04;[EPub Ahead of Print], P Fozouni
CRISPR diagnostics to aid in detecting symptomatic, asymptomatic, and pre-symptomatic carriers of the SARS-CoV-2 virus. Results showed that an amplification-free CRISPR-Cas13a assay, for direct detection read using mobile phone microscopy, accurately detected pre-extracted RNA from nasal swabs in less than 5 minutes & can enable a quicker, lower-cost, and portable screening tool for SARS-CoV-2
TINY IRON OXIDE NANOPARTICLES coated with SILICA have a strong affinity for the RNA genetic material inside the virus that causes COVID-19/CoV2-19. NORWEGIAN U of Science & Technology NTNU’s & St. Olavs Hospital’s new test uses the nanoparticles to extract RNA from a solution containing a sample from the patient. The solution contains substances that crack the virus open so that its RNA genetic material can be extracted.
“We can then identify the genetic code from the RNA and compare it to the coronavirus,” Bjørås said. The researchers tested the accuracy of their method by running tests from patients in parallel with commercial tests. Bjørås said the new method is more sensitive than commercial tests. Bjørås said the lab at NTNU’s Department of Chemical Engineering that is making the magnetic particles can make 30-40,000 tests a day, a rate that can be increased after Easter. The plan is to scale up to be able to produce a minimum of 150,000 tests per week.
Unvaccinated people twice as likely to be reinfected with COVID: study
August 6, 2021
Unvaccinated people are more than twice as likely to be reinfected with COVID-19 as the fully vaccinated, a study by the Centers for Disease Control and Prevention said Friday.The agency said the finding supports its recommendation “that all eligible persons be offered COVID-19 vaccination, regardless of previous SARS-CoV-2 infection status.”
They were compared with 492 “controls” who were matched by sex, age, and time of initial positive test.The analysis found that unvaccinated people were 2.34 times as likely to be reinfected compared to people fully vaccinated with the Pfizer, Moderna or Johnson & Johnson vaccines.
Laboratory studies have shown that blood samples from people previously infected with the original Wuhan strain had poor antibody responses to the Beta variant first identified in South Africa. HRS reports, but DOUBTS, this article.
To a point, this article speaks againt the just above artice: Antibodies to SARS-CoV-2 remain stable, or even increase, seven months after infection
by Barcelona Institute for Global Health 8/6/21
The levels of IgG antibodies against SARS-CoV-2 Spike protein remain stable, or even increase, seven months after infection, according to a follow-up study in a cohort of healthcare workers coordinated by the Barcelona Institute for Global Health (ISGlobal), in collaboration with the Hospital Clinic of Barcelona. The results, published in Nature Communications, also support the idea that pre-existing antibodies against common cold coronaviruses could protect against COVID-19.
The team led by ISGlobal researcher Carlota Dobaño followed a cohort of healthcare workers at the Hospital Clinic (SEROCOV study) from the beginning of the pandemic, in order to evaluate the levels of antibodies against different SARS-CoV-2 antigens over time.
The research team analyzed blood samples from 578 participants, taken at four different timepoints between March and October 2020.
The results show that the majority of infections among healthcare workers occurred during the first pandemic wave (the percentage of participants with SARS-CoV-2 antibodies increased only slightly between March and October—from 13.5% to 16.4%). With the exception of IgM and IgG antibodies against the nucleocapsid (N), the rest of IgG antibodies (including those with neutralizing activity) remained stable over time, confirming results from other recent studies.
“Rather surprisingly, we even saw an increase of IgG anti-Spike antibodies in 75% of the participants from month five onwards, without any evidence of re-exposure to the virus,” says Gemma Moncunill, senior co-author of the study. No reinfections were observed in the cohort.
Regarding antibodies against human cold coronaviruses (HCoV), the results suggest that they could confer cross-protection against COVID-19 infection or disease. People who were infected by SARS-CoV-2 had lower levels of HCoV antibodies. Asymptomatic individuals had higher levels of anti-HCoV IgG and IgA than those with symptomatic infections. “Although cross-protection by pre-existing immunity to common cold coronaviruses remains to be confirmed, this could help explain the big differences in susceptibility to the disease within the population,” says Dobaño.
More information: Ortega N, Ribes M, Vidal M, et al. Seven-month kinetics of SARS-CoV-2 antibodies and protective role of pre-existing antibodies to seasonal human coronaviruses on COVID-19. Nature Communications. DOI: 10.1038/s41467-021-24979-9
BELGIAN ZenTech test rapidly detects antibodies against coronavirus infections. It has started making tens of thousands of the government-certified tests & can ramp up output to make up to 3 million per month. Diagnosis takes just 10-15 minutes and sensitivity is 100 percent: meaning it identifies all patients who have COVID-19 antibodies.
POSITIVE RT-PCR Test Results in Patients RECOVERED From COVID-19. Lan Lan, MD JAMA. 2020;323(15):1502-1503. doi:10.1001/jama.2020.2783. This article shows the test can become NEGATIVE and then LATER POSITIVE with no new CoV2-19 exposure!
SOUTH KOREAN S D Biosensor is making 350,000 test a day and ramping up to 1.5 million tests a day to be exported to the USA and other countries.
8/2020: Israel has developed an accurate 30 second nasal virus detection system
From Trends-In-Medicine 4/2/20: TESTING. The list of available/approved/cleared Covid-19 tests continues to grow. Most of the tests are laboratory or point-of-care tests for diagnosing Covid-19. The rapid 5-15 minute Abbott ID NOW test is starting to be used and even FDA Commissioner Stephen Hahn, MD, called it a game changer. It is a one-at-a-time test for individuals, not for larger populations. However, what is needed next is a serologic (ELISA) test that can detect who has been exposed to SARS-CoV-2 and developed antibodies – and therefore can’t get sick and can’t transmit the virus. Those are the people whose blood may be therapeutic and who could go back to work immediately. Dr. Deborah Birx said she has tasked researchers at the major universities in the country to develop a simple ELISA test, saying, “It is easy to do. In a day or two they could SCREEN AN ENTIRE HOSPITAL. I call on every university in every state to develop Elisas. You can buy antigens and controls online. … Our universities can do that by Friday [April 3]. I put that challenge out to them. We are not waiting. We are asking for help now. It could happen this month if the universities help us.” She said this was done with HIV, and it is exactly the same concept and process for SARS-CoV-2.
Newsletter Science X 4/30/20: “…the most common tests rely on the reverse-transcriptase-polymerase chain reaction (RT-PCR), which amplifies a tiny amount of viral RNA collected from nasopharyngeal swabs. Because RT-PCR requires expensive instruments, trained personnel and often several days to generate results, researchers are avidly exploring other methods, such as isothermal nucleic acid amplification and transcription-mediated amplification, as well as CRISPR technologies….”
Antibody responses to SARS-CoV-2 in patients of novel coronavirus disease 2019
Clinical Infectious Diseases — Zhao J, Yuan Q, Wang H, et al. March 31, 2020
The antibody response in infected patient was tested by serial plasma samples (n = 535) from a total of 173 patients with SARS-CoV-2 infection during hospitalization for total antibodies (Ab), IgM and IgG against SARS-CoV-2. The seroconversion rate for Ab, IgM and IgG was 93.1%, 82.7% and 64.7%, respectively. For Ab, IgM and then IgG, the median seroconversion time was day11, day12 and day14, separately. The presence of antibodies was < 40% within 1 week since onset and increased rapidly to 100.0% (Ab), 94.3% (IgM) and 79.8% (IgG) as of day15 after onset. There was a decrease in viral RNA detectability from 66.7% (58/87) in samples collected before day 7 to 45.5% (25/55) during days 15-39. Significant improvement in the sensitivity for diagnosis of COVID-19 is when RNA and antibody detection are combined; this improvement was evident even in early phase of 1 week since onset. There was an independent association of a higher titer of Ab with a worse clinical condition.
Trends -In-Medicine 5/7/20 Coronavirus Page 8 Among the Covid-19 diagnostic tests that recently got an EUA from the FDA are:
AIT Laboratories’ SARS-CoV-2 Test
Altona Diagnostics’ RealStar SARS-CoV-2, real-time PCR kit for research use only Autobio Diagnostics’ Anti-SARS-CoV-2 Rapid Test
Biocerna’s RT-PCR test, a modified version of Thermo Fisher Scientific’s TaqPath Covid-19 test
GenoSensor’s GS Covid-19 RT-PCR kit
Hologic’s Aptima SARS-CoV-2 test, a molecular test that runs on its Panther system, was submitted for an EUA
KorvaLabs’ Curative-Korva SARS-CoV-2 Assay
LabGenomics’ LabGun Covid-19 RT-PCR Kit
MicroGenDX’ Covid-19 Key assay
Nationwide Children’s Hospital’s SARS-CoV-2 assay
Ortho Clinical Diagnostics’ VITROS Immunodiagnosti
Trends-In-Medicine 5/7/20 Coronavirus
Antibody (serology) testing demand for these tests has skyrocketed but so has the misunderstanding about what they can and cannot do. Basically, these tests can identify people who have antibodies to Covid-19, which means they were exposed to the virus, whether they got sick and recovered or were asymptomatic. Antibody positivity makes people potential donors for convalescent plasma, and it provides epidemiologists with a picture of the spread of the virus within the general population. The problem is there still is no evidence that a person with antibodies has immunity to SARS-CoV-2 or, if there is immunity, how long it lasts. The FDA and NIH continue to remind people of this, but there remains a popular misconception about immunity related to a positive test. And the specificity and sensitivity of the tests are still unclear. The White House proposed that, in some circumstances, two antibody tests be administered to the same person. And the WHO warned against plans for proposed “immunity passports,” which would allow people who have recovered from the coronavirus to resume unrestricted travel and work. Yet, there are a growing number of antibody tests getting either an EUA from the FDA or a CE Mark from the EMA, and there are more than 180 in development.
The latest antibody tests include:
A new approach is the Quidel 15 minute antigen test that detects fragments of virus proteins swabbed from nasal samples
Abbott’s SARS-CoV-2 IgG antibody test, which claims 99% sensitivity and specificity – EUA and CE Mark Erba Mannheim’s ErbaLisa Covid-19 antibody Elisa detection kit – CE Mark
Quest Diagnostics’ Covid-19 antibody test, which consumers can buy online for $119.
Quotient’s MosaiQ Covid-19 antibody microarray test, which claims 99.8% accuracy – CE Mark
Roche’s Elecsys Anti-SARS-CoV-2 antibody test, which claims specificity >99.8% and sensitivity of 100% – EUA and CE Mark
Siemens Healthineers’ fast total antibody test for SARS-CoV-2, which claims specificity and sensitivity of >99% – not yet approved.
Among the tests still in development are: “Cue Health got a $13 million grant from the U.S. Biomedical Advanced Research and Development Authority (BARDA) to create, validate, and gain approval for a fast and portable SARS-CoV-2 point-of-care test. GenMark Diagnostics got a grant for up to $749,000 from BARDA to develop and seek EUA for a diagnostic panel that combines a new SARS-CoV-2 viral target with the company’s ePlex Respiratory Pathogen panel.”
The current lack of preparedness was due to governmental and the wider society not having the necessary vision to understand the implications of what was happening then and then not preparing the appropriate response that could have been used now. CDC regulations are now updated. Now cities, states, and the private market are allowed independently to create their own testing. The Roche pharmaceutical company has developed a simplified and automated technology that will increase testing from 30 to 1,000 tests per day and Roche said it will be quickly able to upscale its production and distribution of this simpler and accurate CoV2-19 virus testing. AS ABOVE, ABBOTT has developed the ID NOW test that is a small, convenient, and easier to perform “point of care self-swab” that gives the answer in 5-15 minutes. Its disadvantage is that a one at-a-time test that cannot screen large populations effectively.
The PROPER SAMPLE culture areas are nasopharyngeal, oropharyngeal, and sputum samples, but not urine, blood, or stool. Companies that are making TEST KITS: ABBOT’s ID NOW is the quickest and most convenient presently. However, it only tests 1 sample at a time, so an army of those machines will be necessary at any one location. Cepheid is a quick test providing positive or negative results for the virus in 45 minutes. S D Biosensor of Korea is ramping up making test kits. GeneMatrix, Chembiao Diagnostics, Hologic, GenMark, Integrated DNA, Pharma Mar, and Thermo Fisher are all developing tests. A new and quick results saliva test is being developed. It is in mass production and simplifies collection, not requiring stringent protective (PPE) masks and gowns. Doing the test would still occur in a healthcare setting under the supervision of a qualified professional. MASKS: N95 means 95% of 300 nm diameter ‘test particles’ are stopped. The CoV2-19 virus is smaller at 125 nm per the CDC and 70-90 nm per the NIH website.
“SALIVA testing will help with the global shortage of swabs for sampling and increase testing of patients, and it will not require health care professionals to be put at risk to collect samples,” Andrew Brooks, the chief operating officer of RUCDR Infinite Biologics, said. RUCDR is backed by Rutgers U. The saliva test builds on the existing TaqPath SARS-CoV-2 Assay used in existing COVID-19 testing to identify RNA from the virus. In addition to identifying carriers of the virus, this form of testing could also make it easier to re-test people who have recovered so they can end their isolation.
JAMA 2/10/21 J T Brooks Effectivenss of Community Mask Wearing as protection to reduce wearers’ exposure to infection.
COVID-19 spreads primarily through respiratory droplets exhaled when infected people breathe, talk, cough, sneeze, or sing. Most of these droplets are smaller than 10 μm in diameter, often referred to as aerosols. The amount of small droplets and particles increases with the rate and force of airflow during exhalation (eg, shouting, vigorous exercise). Exposure is greater the closer a person is to the source of exhalations. Larger droplets fall out of the air rapidly, but small droplets and the dried particles formed from them (ie, droplet nuclei) can remain suspended in the air. In circumstances with poor ventilation, typically indoor enclosed spaces where an infected person is present for an extended period, the concentrations of these small droplets and particles can build sufficiently to transmit infection.
Community mask wearing substantially reduces transmission of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in 2 ways. First, masks prevent infected persons from exposing others to SARS-CoV-2 by blocking exhalation of virus-containing droplets into the air (termed source control). This aspect of mask wearing is especially important because it is estimated that at least 50% or more of transmissions are from persons who never develop symptoms or those who are in the presymptomatic phase of COVID-19 illness.1 In recent laboratory experiments, multilayer cloth masks were more effective than single-layer masks, blocking as much as 50% to 70% of exhaled small droplets and particles.2,3 In some cases, cloth masks have performed similar to surgical or procedure masks for source control. Second, masks protect uninfected wearers. Masks form a barrier to large respiratory droplets that could land on exposed mucous membranes of the eye, nose, and mouth. Masks can also partially filter out small droplets and particles from inhaled air. Multiple layers of fabric and fabrics with higher thread counts improve filtration. However, the observed effectiveness of cloth masks to protect the wearer is lower than their effectiveness for source control,3 and the filtration capacity of cloth masks can be highly dependent on design, fit, and materials used. Standards for cloth masks are needed to help consumers select marketed products.
Epidemiological investigations have helped quantify the benefit of mask wearing to prevent the spread of SARS-CoV-2 (Table; Supplement). At a hair salon in which all staff and clients were required to wear a mask under local ordinance and company policy, 2 symptomatic, infected stylists attended to 139 clients and no infections were observed in the 67 clients who were reached for interviewing and testing. During a COVID-19 outbreak on the USS Theodore Roosevelt, persons who wore masks experienced a 70% lower risk of testing positive for SARS-CoV-2 infection.4 Similar reductions have been reported in case contact investigations when contacts were masked5 and in household clusters in which household members were masked.6
Yale’s rapid COVID-19 saliva test receives FDA emergency use authorization
by Michael Greenwood, Yale University
A saliva-based laboratory diagnostic test developed by researchers at the Yale School of Public Health to determine whether someone is infected with the novel coronavirus has been granted an emergency use authorization by the U.S. Food and Drug Administration (FDA).
The method, called SalivaDirect, is a test for asymptomatic individuals SalivaDirect is simpler, less expensive, and less invasive than the traditional method for such testing, known as nasopharyngeal (NP) swabbing. Results so far have found that SalivaDirect is highly sensitive and yields similar outcomes as NP swabbing.
With the FDA’s emergency use authorization, the testing method is immediately available to other diagnostic laboratories that want to start using the new test, which can be scaled up quickly for use across the nation—and, perhaps, beyond—in the coming weeks, the researchers said. A key component of SalivaDirect, they note, is that the method has been validated with reagents and instruments from multiple vendors.
3M is scaling up producing N95 face masks. Construction and other companies are donating their N95 mask and gown stockpiles while the federal government is shipping ventilators and other stockpiled necessities to infection hot spots. A safe and effective way to sterilize used N95 respirators to further conserve PPE (www.battelle.org/newsroom/news-details/battelle-cleared-to-sterilize-n95-masks-at-max-capacity-operate-in-other-states-to-fight-coronavirus-ppe-shortage. opens in new tab)
NEJM 6/6/20 M C Weinstein: “…But we believe that the WHO is dead wrong to suggest that we cannot act until we “guarantee” the accuracy of the immunity-certification process. Demanding incontrovertible evidence may be appropriate in the rarefied world of scholarly scientific inquiry. But in the context of a raging pandemic, we simply do not have the luxury of holding decisions in abeyance until all the relevant evidence can be assembled….”
“Pipeline: investigational therapies of COVID-19/CoV2-19
Diana Ernst, RPh of MPR wrote on 3/11/20:
“Currently, there are no antivirals licensed by the FDA to treat patients with COVID-19. While no specific treatment for corona 2019 (COVID-19/CoV2-19) is currently available, several therapies are being investigated globally.”
The OXFORD University “RECOVERY” study of 6/2020 showed that DECADRON/dexamethasone at 6 mg a day X 7-10 days costing $6.00 a day, reduced ventilator deaths by 30% & 20% in those with less severe disease. PREDNISONE or prednisolone (see just below) in comparable doses would likely do the same. INHALED STEROIDS in high dose like CICLESONIDE/ALVESCO and BECLOMETHASONE/ QVAR have the same logic with documented scientific proof that supports their therapeutic equality with oral or injected (gluco-) corticoid, not androgenic, steroids. For more moderate disease, see the above PREDNISONE discussion in the section on ventilators.
“Aarhus University in Denformark. Senicapoc binds to calcium-activated potassium channels involved in fluid secretion on the surface of the airway in the lungs. The drug also binds to potassium channels in macrophages and T-cells—cells involved in immune responses. What Simonsen and his colleagues discovered was that this combination—blocking secretions and mitigating the immune system reaction—was able to inhibit the development of severe acute respiratory syndrome (SARS) and damage to the lungs.”
Improved Outcomes With Methylprednisolone in Hospitalized Hypoxic Patients With COVID-19
Brandon May 6/4/21
In addition to other benefits, a lower proportion of patients in the methylprednisolone group required a ventilator during hospitalization. Credit: Getty Images
Treatment with methylprednisolone led to significantly greater improvements in clinical status and shortened hospital length of stay than treatment with dexamethasone in hospitalized COVID-19 patients with hypoxia, according to the results of a study published in BMC Infectious Diseases.
This prospective trial included 86 hospitalized patients with COVID-19 in Iran. Participants were randomly assigned to either methylprednisolone 2 mg/kg/d (n=44) or dexamethasone 6 mg/kg/d (n=42). Treatment was administered in conjunction with standard of care for 10 days.
The primary endpoint was 28-day mortality rate and clinical status at 5 and 10 days. The data were examined using a 9-point World Health Organization (WHO) ordinal scale ranging from uninfected (point 0) to death (point 8). Secondary endpoints included intensive care unit admission and the need for invasive mechanical ventilation.
No significant differences were observed between the treatment groups in terms of demographic variables, comorbid diseases, or disease severity at time of admission. At day 5, however, patients treated with methylprednisolone reached a significantly better clinical status compared with patients who received dexamethasone (4.02 vs 5.21, respectively; P =.002). Patients in the methylprednisolone group also had better clinical status at day 10 (2.90 vs 4.71; P =.001).
Patients in the methylprednisolone group had a significantly better overall mean 9-point WHO score (3.909 vs 4.873; P =.004). The use of methylprednisolone was also associated with a significantly shorter mean length of hospital stay (7.43±3.64 vs 10.52±5.47 days; P =.015). A lower proportion of patients in the methylprednisolone group required a ventilator during hospitalization (18.2% vs 38.1%; P =.040).
Limitations of the study included its small sample size and the lack of robust data on computer tomography features and disease-related complications.
The study investigators concluded that larger randomized trials with longer follow-up periods are needed to further examine “the beneficial effect of methylprednisolone in patients with COVID-19 pneumonia.”
Reference
Ranjbar K, Moghadami M, Mirahmadizadeh A, et al. Methylprednisolone or dexamethasone, which one is superior corticosteroid in the treatment of hospitalized COVID-19 patients: a triple-blinded randomized controlled trial. BMC Infect Dis. 2021;21(1):337. doi:10.1186/s12879-021-06045-3
This 7/17/20 Journal of Experimental Medicine “Rationale for CXCR2 antagonists for the treatment of COVID-19”
by L F Koening discusses using “inhibitors of chemokine/chemokine receptor pathways to block excessive infiltration of neutrophils to interrupt the self-reinforcing hyperinflammation in severe cases of COVID-19 infection. … There is strong evidence to investigate the usage of CXCR2 antagonists in the treatment of severe COVID-19…. overreactive immune system with infiltration of inflammatory monocytes and neutrophils to the site of infection alongside an exaggerated release of proinflammatory cytokines is an important driver of severe lung damage in COVID-19 (Vabret et al, 2020).”
https://www.practiceupdate.com/c/103914/1/24/?elsca1=emc_enews_daily-digest&elsca2=email&elsca3=practiceupdate_diab&elsca4=diabetes&elsca5=newsletter&rid=MjA2NjE0OTY0Mzg2S0&lid=10332481
Nasal Povidone-Iodine Solutions Effectively Inactivate SARS-CoV-2 In Vitro
Brandon May October 2, 2020
All 3 nasal povidone-iodine solutions completely inactivated SARS-CoV-2 within 15 seconds of contact.
Nasal povidone-iodine (PVP-I) solutions at concentrations between 0.5% and 2.5% were capable of rapidly inactivating the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in vitro at relatively short contact times, according to study results published in JAMA Otolaryngology–Head & Neck Surgery.
High viral loads of SARS-CoV-2, the virus that causes coronavirus disease 2019 (COVID-19), have been detected in both the nasopharynx and oropharynx of asymptomatic and symptomatic carriers. Physical barriers and personal protective equipment are typically employed to reduce transmission of the virus through aspiration, but some research has supported the use of intranasal PVP-I solutions as an effective agent against SARS-CoV-2.
In this in vitro laboratory study, researchers tested nasal antiseptic solutions composed of aqueous PVP-I as the active ingredient against the fully sequenced USA-WA1/2020 strain of SARS-CoV-2. Diluted concentrations of PVP-I at 0.5%, 1.25%, and 2.5% were studied, and efficacy of these solutions was compared with controls. The positive control consisted of 70% ethanol on test media infected with SARS-CoV-2. The virus-absent test media were added to 2 tubes of the compounds, which served as toxicity and neutralization controls.
Investigators incubated both the test solutions and the virus at a mean room temperature of 22 °C for 15 & 30 seconds. The log reduction value following 15 seconds and 30 seconds of the given treatment comprised the primary outcome.
All 3 PVP-I solutions completely inactivated SARS-CoV-2 within 15 seconds of contact, as represented by a reduction of greater than 3log10 of the 50% cell culture infectious dose (CCID50) of the virus (3.67 log10 CCID50/0.1 mL to ≤0.67 log10 CCID50/0.1 mL). In contrast, the positive ethanol control did not completely inactivate the virus after this same time period. There were no cytotoxic effects observed on cells following contact with the tested nasal antiseptics.
A limitation of the study included the lack of in vivo assessment of the efficacy and safety of the PVP-I solutions against SARS-CoV-2.
Based on their findings, the investigators concluded that “povidone-iodine nasal irrigation may be beneficial for the population at large as an adjunct to mask usage as a means of virus mitigation.”
Reference
Frank S, Brown SM, Capriotti JA, Westover JB, Pelletier JS, Tessema B. In vitro efficacy of a povidone-iodine nasal antiseptic for rapid inactivation of SARS-CoV-2. JAMA Otolaryngol Head Neck Surg. Published online September 17, 2020. doi:10.1001/jamaoto.2020.3053
Allied BioScience: (Newsletter Science X 5/18/20) manufactures antimicrobial surface coating that are a continuously active with the potential use against the transmission of viruses. “We evaluated this technology by testing a modified antimicrobial coating against the human coronavirus 229E, which is one of the viruses that causes the common cold,” Gerba said. “Even two weeks after the coating was applied, it was capable of killing more than 99.9% of the coronaviruses within two hours.”
8/2020 Israel has developed the AURA disinfecting air purifier which struck me as similar to the Sun Pure air purifier with an internal UV light. Israel has also developed a virus killing dry mist 30 feet long glass enclosed virus killing tunnel.
Here is a common sense article about HCQ use in Costa Rica that is not a double blind, placebo controlled cross-over study that still has power and influences my thinking more than the Lancet 5/2020 (negative) article discussed further below of which I suspect data legerdemain.
Hydroxychloroquine: the drug Costa Rica uses successfully to fight covid-19
HYDROXYCHLOROQUINE by R. Moss, MD 5/2020
CHLOROQUINE, the precursor of HCQ, was invented by Bayer in 1934, HYDROXYCHLORO- QUINE was developed during World War II as a safer alternative and approved for medical use in the USA in 1955. The World Health Organization/WHO considers it an essential medicine, among the safest and most effective. In 2017, USA doctors prescribed it 5 million times, the 128th most commonly prescribed drug in the country-no EKG was required. There have been hundreds of millions of prescriptions for malaria worldwide since its inception. Doctors also prescribe it for LUPUS or RHEUMATOID ARTHRITIS patients who may use it their entire lifetimes with few or no ill effects.
The medical and standard media high-lighted “QT interval” prolongation and the risk of sudden cardiac death. The FDA and NIH joined in requiring randomized, controlled, double-blind studies before physicians prescribed HCQ: not so for EFFEXOR, CELEXA, PROZAC, CIPRO, ECONOAZOLE, HALDOL, etc. which ALSO prolong the QT interval and for which there is no requirement to perform an EKG. No one mentioned that the risk of cardiac arrest was far higher from watching the SUPERBOWL. Nor did the media declare that HCQ and CHLOROQUINE have been used throughout the world for half a century, making them among the most widely prescribed drugs in history with not a single reported case of “arrhythmic death” according to the WHO and the American College of Cardiology. Physicians on the frontlines have found benefit in treating patients with a variety of agents including HCQ such as azithromycin, zinc, quercetin, vitamins D and C with few, if any, complications.
Virology, transmission, and pathogenesis of SARS-CoV-2
BMJ 2020; 371 doi: https://doi.org/10.1136/bmj.m3862 (Published 23 October 2020) Cite this as: BMJ 2020;371:m3862
N-acetylcysteine as a possible adjuvant in the treatment of SARS-CoV-2
Dear Editor,
We refer to the article Virology, transmission and pathogenesis of SARS-CoV-2 (1) stating that a distinctive feature of Covid 19 is the presence of mucous plugs in the respiratory tract explaining the severity of this disease. In an earlier contribution in NTVG (2) we recommended treatment of these mucous plugs with mucolytic agent N-acetylcysteine reliving bronchiolar obstruction.
Furthermore the SARS-CoV-2 virus is, like the influenza virus, an RNA virus with a lipid double membrane envelope. Treatment strategies that favorably influence the course of influenza may also be effective in SARS CoV-2. In 1997 an Italian study was published on the effects of n-acetylcysteine on influenza at a dosage of 600mg twice daily. The outcome showed a significant decrease in the frequency of flu-like episodes, the severity and the duration of the illness. (3) NAC has been shown in vitro to inhibit the replication and expression of pro-inflammatory molecules in cells infected with the H5N1 influenza A virus. (4) NAC has been shown to have an antioxidant and immunomodulating effect in the treatment of influenza pneumonia (5), mitigating the chance of the cytokine storm responsible for the lung pathology.
We suggest considering N-acetylcysteine as an adjuvant to the standard treatment of SARS-CoV-2, also taking into account the favorable risk / benefit ratio and low costs of this drug.
Dr. D.J. Der Kinderen ([email protected])
Dr. J.W. Koten ([email protected])
1. Cevic, M, Kuppalli J, Kindrachuck J, Peiris M. Virology, transmission and pathogenesis of SARS-CoV-2. BMJ 2020;371:m3862
2. DerKinderen D, Koten JW. N-acetylcysteïne als adjuvant bij de behandeling van SARS-CoV-2. https://www.ntvg.nl/artikelen/chloroquine-als-mogelijke-behandeling-van-…
3. The Flora S, Grassi C, Carati L. Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment. Eur Respir J 1997; 10: 1535-1541
4. Geiler J, Michaelis M, Naczk P, Leutz A, Langer K, et al. N-acetyl-L-cysteine (NAC) inhibits virus replication and expression of pro-inflammatory molecules in A549 cells infected with highly pathogenic H5N1 influenza A virus. Biochem Pharmacol 2009; 79: 413
5. Casanova T, Garigliany M. N-Acetylcysteine: An Old Drug With Variable Anti-Influenza Properties. Journal of Controversies in Biomedical Research 2016; 2: 1–8.
Newsletter Science X 5/18/20: COVID-19 Research Outcomes Worldwide Network (CROWN) Collaborative, is testing whether the antimalaria drug chloroquine (HRS says it should be hydroxychloroquine with vitamin D3 25,000 units a week + Zinc sulphate or gluconate 50-100 mg a day 5 days a week) can prevent COVID-19 infection or decrease its severity in front-line health-care workers. An estimated 30,000 such workers from across the globe will participate in the clinical trial, which the collaborative is calling the CROWN CORONATION trial.
The collaborative and the trial are funded by the COVID-19 Therapeutics Accelerator. HRS wonders if this trial is literally designed to fail.
Antivirals
Drug Combination with Hydroxychloroquine Promising: NYU Study
by A PaoOLICELLI NEW YORK CITY 5/12/20
NEW YORK – Researchers at New York University’s Grossman School of Medicine found patients given the antimalarial drug hydroxychloroquine along with zinc sulphate 100 mg a day and the antibiotic azithromycin were 44 percent less likely to die from the coronavirus less like to need the ICU
“A 5 day treatment with CHLOROQUINE or HYDROXYCHLOROQUINE (HCQ or Plaquenil) combined with AZITHROMYCIN (AZITH) seems quite EFFECTIVE for COV2-19. An open-label study investigated hydroxychloroquine in hospitalized patients with confirmed COVID-19 at The Méditerranée Infection University Hospital Institute in Marseille, France. (The study was run by epidemiologist Didier Raoult, MD.) Patients received oral HCQ 200 mg 3 times daily for 10 days (n = 20) vs control group (n=16). Patients were age 12 years and older, and had PCR documented SARS-CoV-2 in nasopharyngeal samples at admission. Treatment with the antibiotic AZITH was also provided. The end point was virological clearance at day 6. (There have been no cardiac complications due to the medicine combination in an experience now exceeding1,000 patients.)
Results showed that by day 6 post-inclusion, 70% of HCQ-only treated patients were cured of the virus vs 12.5% in the control group (p =.001). At day 6, 100% of patients treated with HCQ + AZITH was cured of the virus compared with 57.1% of patients treated with hHCQ only, and 12.5% of the control group (p <.001). A significant difference between the HCQ and control groups was reported as early as day 3.” Similar results were found at the University of Minnesota.
While the results look promising, the researchers noted limitations to their study including small sample size, limited long-term outcome follow-up, and dropout of 6 patients from the study.” There is data that this combination is more effective in milder cases and less helpful in severe in extremis cases. A Brazilian study found no benefit and some cardiac down sides
The Effect of Chloroquine, Hydroxychloroquine and Azithromycin on the Corrected QT Interval in Patients with SARS-CoV-2 Infection 4/29/20
Moussa Saleh , et al
Originally published29 Apr 2020https://doi.org/10.1161/CIRCEP.120.008662Circulation: Arrhythmia and Electrophysiology.
Abstract
Background – The novel SARs-CoV-2 coronavirus is responsible for the global COVID-19 pandemic. Small studies have shown a potential benefit of chloroquine/hydroxychloroquine ± azithromycin for the treatment of COVID-19. Use of these medications alone, or in combination, can lead to a prolongation of the QT interval, possibly increasing the risk of Torsade de pointes (TdP) and sudden cardiac death.
Results – 221 patients were treated for COVID-19 with chloroquine/hydroxychloroquine. Ten patients (5.0%) received chloroquine, 191 (95.0%) received hydroxychloroquine and 119 (59.2%) also received azithromycin. The primary outcome of Torsade de Pointe (TdP) was not observed in the entire population. Baseline QTc intervals did not differ between patients treated with chloroquine/hydroxychloroquine (monotherapy group) vs. those treated with combination group (chloroquine/hydroxychloroquine and azithromycin) (440.6 ± 24.9 ms vs. 439.9 ± 24.7 ms, p =0.834). The maximum QTc during treatment was significantly longer in the combination group vs the monotherapy group (470.4 ± 45.0 ms vs. 453.3 ± 37.0 ms, p = 0.004). Seven patients (3.5%) required discontinuation of these medications due to QTc prolongation. No (!) arrhythmogenic deaths were reported.
Conclusions – In the largest reported cohort of COVID-19 patients to date treated with chloroquine/hydroxychloroquine {plus minus} azithromycin, no instances of TdP or arrhythmogenic death were reported. Although use of these medications resulted in QT prolongation, clinicians seldomly needed to discontinue therapy.
Hydroxychloroquine rated ‘most effective’ coronavirus treatment, poll of doctors finds
By Natalie O’Neill 4/2/20: “Hydroxychloroquine rated ‘most effective’ coronavirus treatment, poll of doctors finds an international poll of thousands of doctors rated the Trump-touted anti-malaria drug hydroxychloroquine the best treatment for the novel coronavirus.Of the 2,171 physicians surveyed, 37 percent rated hydroxychloroquine the “most effective therapy” for combating the potentially deadly illness. The survey, conducted by the global health care polling company Sermo, (HRS says I receive the legitimate polls from Sermo all the time) also found that 23 percent of medical professionals had prescribed the drug in the US — far less than other countries. “Outside the US, hydroxychloroquine was equally used for diagnosed patients with mild to severe symptoms whereas in the US it was most commonly used for high risk diagnosed patients,” the survey found. The medicine was most widely used in Spain: 72 %. 6,227 physicians were questioned. Sermo CEO Peter Kirk called the polling results a “treasure trove of global insights for policymakers.” The 30 countries where doctors were surveyed included Europe, South America and Australia — and no incentives were provided to participate, the company said.”
A suspicious and negative study was just published in Lancet 5/2020. From within the Lancet article: “671 hospitals, six continents … this is an observational study that cannot account for unmeasured confounding factors… automatic data extraction … key missing values are kept to a minimum.” HRS believes this is data legerdemain. HRS speaks: It is not a case of “Don’t confuse me with facts”, but the best clinical insights exceed so-called knowledge by at least one step.
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)31180-6/fulltext
A researcher friend, DDH, speaks regarding the just above Lancet article-slightly edited:
“(1) It is one publication relying on what may be a significantly skewed selection of data sources. I nevertheless extend my congratulations to the authors for their ability to likely conceal this flaw in the structuring of the report. They are true artists.
(2) A major ethical factor is being sidestepped. Specifically, that taking hydroxychloroquine is an individual choice that should certainly be done with the advice from a physician. Ultimately this is the sole and rightful decision of each individual and their physician and NOT with the intervention by some State authority. Indeed, in most matters, the individual, not the state, must be the final arbiter of almost all human activity.
The authors, with great artistry and elegance, have likely perpetrated scientific fraud. Even if their assertions are correct, they have nonetheless disregarded scientific integrity if only for the ethical, personal choice reason mentined above. The State has no right to compel me to take or not take a particular agent, if through my own individual resources, I make a decision one way or the other.”
About That Big HCQ Study…
— Questions arise over inconsistencies in data; confounders may impact future COVID-19 treatments
by Molly Walker, Associate Editor, MedPage Today 5/26/20
“As more outside experts have had a chance to review the huge observational study released last week on the safety and efficacy of hydroxychloroquine (HCQ) and chloroquine for COVID-19, whispers that something was amiss have turned into a loud buzz. The analysis, published on Friday in The Lancet, looked at nearly 100,000 COVID-19 patients including about 15,000 treated with the antimalarials, either with or without an antibiotic. HCQ was associated with nearly doubled risk of death in the hospital and about 20-fold higher rates of ventricular arrhythmias, the investigators reported.
But other researchers looking at the fine print had questions. “The claim to have captured data from over 60,000 hospitalizations at over 550 hospitals in North America by April 13th concerns me, given that there were approximately 60,000 COVID-19 hospitalizations total from approximately 6,000 hospitals across all of the United States through April 13th,” Matthew Spinelli, MD, of University of California San Francisco, told MedPage Today.
Data from the COVID Tracking Project through April 13 bore him out. Sapan Desai, MD, PhD, one of the Lancet authors and founder of Surgisphere Corp., a physician-led public service organization in Chicago that provided much of the data for the analysis, told MedPage Today there were multiple reasons for the discrepancy between the data in the study and that in the COVID Tracking Project.
“There is often a delay before public health reporting catches up to data at the hospital level,” he said. Desai also pointed to “issues with data capture at the public health level from various hospitals that could lead to inaccuracies or delays in public reporting.” Walid Gellad, MD, of the University of Pittsburgh, noted on Twitter that 73 deaths were recorded in Australia according to the Lancet authors, which is “more than the number of deaths in Australia on April 20.”
“Not one healthcare facility that contributed data is named or acknowledged. I have never seen that unless someone was using a public[ly] available dataset,” David Glidden, PhD, also of University of California San Francisco, told MedPage Today. Desai added they are reviewing the analysis to ensure there are no issues with the data. Spinelli also raised questions about the mortality data, saying “prior well-done observational studies did not show such a signal for mortality.”
Indeed, a recent New England Journal of Medicine study on hydroxychloroquine did not find the same effect size for mortality. A blog hosted by statisticians at Columbia University in New York City raised several other issues, including the results being confounded by disease severity, lack of hierarchical modeling, and how the data appeared to be aggregated across continents. Speaking to the latter point, Desai said the sophistication of data retrieval requires they link directly with electronic health records (EHRs); consequently Surgisphere works exclusively with institutions utilizing “well-established EHRs.”
“This requirement allows us to only maintain collaborations with top-tier institutions that are supported by the level of data-integrity and sophistication required for such work,” Desai said. “Naturally, this leads to the inclusion of institutions that have a tertiary care level of practice and provide quality healthcare that is relatively homogenous around the world.” HRS, this author, believes this is out right politically-inspired unethical data distortion intending to discredit hydroxychloquine.
Already ripple effects from the study are starting to emerge, with NPR reporting the World Health Organization (WHO) temporarily halting the Solidarity Trial, which aimed to study a variety of COVID-19 treatments, including HCQ. “I believe that whenever a question arises, it is a responsible action to review the ongoing outcomes as a safety measure in a clinical trial. Their stoppage is temporary based on performing such a review,” the study’s lead author, Mandeep Mehra, MD, of Brigham and Women’s Hospital in Boston, told MedPage Today.
Spinelli said the study merits additional review, ideally including the primary data. “I am concerned that more desperately needed clinical trials may be stopped as a result of this study,” he said.
https://c19study.com/ is a link to a CRUCIAL graph showing the effectiveness of HCQ in the following countries that have much lower death rates than France, United Kingdom, USA (that have 650 deaths per million population): the low frequency death rate countries (100 deaths per million population) are Indonesia (270 million), India (population 1.3 billion: billion, not million), Turkey (population 85 million) Malaysia (population 33 million), Greece (population 33 million)._
A similar, but different format, graph that can be reproduced is just below re the benefit of HCQ use vs not using HCQ consistently in the treatment of CoV2-19.
Credit: AAPS Online, fair use
“Large Proportion of COVID-19 Studies Have Low-Level Evidence
7/31/20
HealthDay News — A large proportion of studies on COVID-19 have a low level of evidence, according to a research letter published online July 27 in JAMA Internal Medicine.
Krishna Pundi, M.D., from the Stanford University School of Medicine in California, and colleagues examined the characteristics and expected strength of evidence of COVID-19 studies registered on ClinicalTrials.gov. A total of 1,551 studies registered as of May 19, 2020, met the inclusion criteria, including 911 interventional (664 randomized clinical trials [RCTs]) and 640 observational studies.
The researchers found that mortality, ventilation requirement, and treatment complications were frequently reported primary and secondary outcomes (33.9, 26.6, and 23.1 percent, respectively). Of the studies, 29.1 percent could potentially yield the highest level of individual study evidence, 2011 Oxford Center for Evidence-Based Medicine level 2 evidence. Blinding was reported for 364 RCTs, of which 29.3, 35.8, and 17.0 percent were placebo-controlled, planned enrollment of more than 100 participants, and reported at least two study centers or sites, respectively. Only 11.3 percent of RCTs were placebo-controlled and blinded with at least two study centers. Overall, 80.8 and 19.2 percent of the observational studies were single-center and multicenter, respectively. Few studies (13.6 percent) were prospective cohort studies that could yield level 2 evidence.
“Even before results are known, most studies likely will not yield meaningful scientific evidence at a time when rapid generation of high-quality knowledge is critical,” the authors write.”
Early Data Show Potential Benefit of Acalabrutinib in Severe COVID-19
Diana Ernst, RPh 6/9/20
Acalabrutinib (Calquence) is a Bruton tyrosine kinase (BTK) inhibitor
that reduces respiratory distress as well as the hyperinflammatory immune response associated with coronavirus disease 2019 (COVID-19), according to an NIH study. BTK is significantly elevated in blood samples of patients with severe CoV2-19
19 hospitalized patients with COVID-19 for hypoxemia who had evidence of inflammation took acalabrutinib 100 mg twice daily for 10 days (supplemental oxygen cohort n=11) or 14 days (mechanical ventilation cohort n=8) plus best supportive care. A subset of patients in both cohorts received concomitant treatment with steroids and/or hydroxychloroquine.
Results showed that by the end of treatment, 8 of the 11 patients in the supplemental oxygen group improved rapidly and had been discharged from the hospital. Also noted were normalization or decrease in C-reactive protein and interleukin-6 (IL-6), & improved lymphopenia.
Cold plasma against the coronavirus
by Max Planck Society 6/11/20
A possible option for the treatment of Covid-19 patients.Terraplasma medical is testing a device originally intended to disinfect chronically infected wounds, in the treatment of Covid-19 patients requiring mechanical ventilation.
Approximately half of who were mechanically ventilated that died had additional infections in hospital. Cold plasma therapy could prevent these superinfections and reduce the risk of hospital staff becoming infected with coronavirus. Preliminary tests by medical GmbH, a subsidiary of Max Planck spin-off suggest that cold atmospheric plasma (i.e. weakly ionized air) can render SARS-CoV-2 harmless in cell cultures.
Plasma is the fuel of the stars. In a highly diluted cold variant, ionized gas—or more precisely, ionized air—inactivates bacteria in chronically infected wounds. Atmospheric plasma can also inactivate viruses, like noro- and adenoviruses in solution & can also help treat COVID-19 patients. “The tests suggest that cold atmospheric plasma kills the corona virus in solution,” says Jens Kirsch, CEO of Terraplasma Medical. “We already know that cold plasmas do not damage the mucous membranes if we use the correct plasma design and the dose does not exceed certain limits,” said Gregor Morfill, former Director of the Max Planck Institute for Extraterrestrial Physics.
“We hope to be able to prevent the virus from spreading from the mouth, nose, and throat to the lower respiratory tract of COVID-19 patients whose lungs are still free of the virus,” says Kirsch. “… thus reduce the number of COVID-19 patients requiring treatment in ICUs or mechanical ventilation.”
‘Aeronabs’ promise powerful, inhalable OTC protection against CoV2-19
ScienceDaily Top Science|August 12, 2020
UC San Francisco scientists devised a novel approach to halting the spread of SARS-CoV-2. UCSF graduate student Michael Schoof & team engineered a completely synthetic, production-ready molecule that straitjackets the crucial SARS-CoV-2 machinery that allows the virus to infect cells. This is reported in preprint bioRxiv. Experiments using live virus show that the molecule is among the most potent SARS-CoV-2 antivirals yet discovered.
In an aerosol formulation dubbed “AeroNabs” these molecules could be self-administered with a nasal spray or inhaler. Used once a day, AeroNabs could provide powerful, reliable protection against SARS-CoV-2 until a vaccine becomes available. The research team is in active discussions with commercial partners to ramp up manufacturing and clinical testing of AeroNabs. If successful these will be an inexpensive, over-the-counter medication to prevent and treat COVID-19 and “serve as an important stopgap until vaccines provide a more permanent solution to COVID-19,” said AeroNabs co-inventor Peter Walter, PhD, professor of biochemistry and biophysics at UCSF and a Howard Hughes Medical Institute Investigator. For those who cannot access or don’t respond to SARS-CoV-2 vaccines, Walter added, AeroNabs could be a more permanent line of defense against COVID-19
Llama-Inspired Design
Though engineered entirely in the lab, AeroNabs were inspired by nanobodies, antibody-like immune proteins that naturally occur in llamas, camels and related animals. “Though they function much like the antibodies found in the human immune system,” explained co-inventor Aashish Manglik, MD, PhD, an assistant professor of pharmaceutical chemistry E.g. nanobodies are an order of magnitude smaller than human antibodies, which makes them easier to manipulate and significantly more stable than the antibodies of other mammals. Unlike human antibodies, nanobodies can be easily and inexpensively mass-produced via E. coli or yeast that are transformed into high-output nanobody factories similar to decades of mass-produced insulin.
SARS-CoV-2 relies on its spike proteins to infect cells. Like a retractable tool, spikes switch from a closed, inactive state to an open, active state. When any of a virus particle’s approximately 25 spikes become active, that spike’s three “receptor-binding domains,” or RBDs, become exposed and are primed to attach to ACE2 (pronounced “ace two”), a receptor found on human cells. Through a lock-and-key-like interaction between an ACE2 receptor and a spike RBD, the virus gains entry into the cell, where it then transforms its new host into a coronavirus manufacturer.
Nanobodies Disable Spikes and Prevent Infection
To find effective candidates, the scientists parsed a recently developed library in Manglik’s lab of over 2 billion synthetic nanobodies. Using cryo(cold)-electron microscopy to visualize the nanobody-spike interface. Veronica Rezelj, PhD, a virologist in the lab of Marco Vignuzzi, PhD, at Institut Pasteur in Paris, tested the three most promising nanobodies against live SARS-CoV-2, and found the nanobodies to be extraordinarily potent, preventing infection even at extremely low doses.
The most potent of these nanobodies, however, not only acts as a sheath over RBDs, but also like a molecular mousetrap, clamping down on spike in its closed, inactive state, which adds an additional layer of protection against the spike-ACE2 interactions that lead to infection.
From Nanobodies to AeroNabs
The scientists then engineered this double-action nanobody in a number of ways to make it into an even more potent antiviral. They mutated every one of the amino-acid building blocks of the nanobody that contacts spike to discover two specific changes that yielded a 500-fold increase in potency.
Three nanobodies were linked together. As noted, each spike protein has three RBDs, any of which can attach to ACE2 to grant the virus entry into the cell. The linked triple nanobody devised by the researchers ensured that if one nanobody attaches itself to an RBD, the other two would attach to the remaining RBDs. They found that this triple nanobody is 200,000 times more potent than a single nanobody alone. It “was so effective that it exceeded our ability to measure its potency.”
Would Be Easy to Administer as an Aerosol: This ultrapotent three-part nanobody construct formed the foundation for AeroNabs. In a final set of experiments, it was proven that AeroNabs are a potent SARS-CoV-2 antiviral that could be practical to administer via a shelf-stable inhaler or nasal spray. This may help reshape the course of the pandemic worldwide.”
Medical Xpress 8/21/20: Online in Cell. “Scientists at Washington University School of Medicine developed a vaccine that targets the SARS-CoV-2 virus that can be given in one dose via the nose and is effective in preventing infection in mice susceptible to the novel coronavirus. Nasal delivery route created a strong immune response throughout the body, but it was particularly effective in the nose and respiratory tract.”
“Famotidine Use Is Associated With Improved Clinical Outcomes in Hospitalized COVID-19 Patients
• In a retrospective cohort study of 1620 patients with COVID-19, 84 (5.1%) who received H2 bocker famotidine were had a significantly reduced risk for death or intubation. There was a no protective effect associated with use of PPIs (aHR, 1.34). In patients hospitalized with COVID-19 and not initially intubated, famotidine use was associated with a twofold reduction in clinical deterioration leading to intubation or death. Randomized controlled trials are underway. In vitro, famotidine inhibits HIV replication (2). Recently, Wu et al. (3) used computational methods to predict structures of proteins encoded by the SARS-CoV-2 genome and identified famotidine as one of the drugs most likely to inhibit the 3-chymotrypsin-like protease (3CLpro) which processes proteins essential for viral replication (4).
T
Famotidine Use Is Associated With Improved Clinical Outcomes in Hospitalized COVID-19 Patients: A Propensity Score Matched Retrospective Cohort Study
Iloprost May Be Useful in the Treatment of COVID-19 Vasculopathy
l
7/31/20 Rheumtology Advisor: Iloprost may help reduce lung damage and thrombotic complications observed in some patients with COVID-19.
Prostacyclin receptor agonist iloprost may be a useful adjunctive treatment for coronavirus disease 2019 (COVID-19) vasculopathy, according to a report published in Lancet Rheumatology.1
3 patients with severe COVID-19 and hypoxemia who tested positive for CoV2-19, 2 had digital ischemia; all 3 received supportive oxygen treatment. Based on clinical diagnoses and persistent oxygen requirement, patients received a 5-day intravenous infusion of iloprost (0.5 mg/kg/min).
Following continuous treatment with iloprost, patients showed sustained clinical improvement in digital ischemia and cardiovascular and respiratory parameters, decreasing oxygen requirements, and increasing Pa02:FiO2 ratio. It was noted that none of the patients required mechanical ventilation or had any serious adverse events. Any complications, including mild rebound tachycardia, observed upon iloprost cessation were resolved before patients were discharged from the hospital.
References
1. Moezinia CJ, Ji-Xu A, Azari A, Horlick S, Denton C, Stratton R. Iloprost for COVID-19-related vasculopathy [published online July 10, 2020]. Lancet Rheumatol. doi:10.1016/S2665-9913(20)30232-0
2. Hughes M, Ong VH, Anderson ME, et al. Consensus best practice pathway of the UK Scleroderma Study Group: digital vasculopathy in systemic sclerosis. Rheumatology. 2015;54(11):2015-2024.
Gastroenterology 2020 May 21;[EPub Ahead of Print], DE Freedberg, J Conigliaro, TC Wang, KJ Tracey, MV Callahan, JA Abrams, , ME Sobieszczyk, DD Markowitz, A Gupta, MR O’Donnell, J Li, DA Tuveson, Z Jin, WC Turner, DW Landry”
https://globalepidemics.org/TOCILIZUMAB (Actemra; Roche) and SARILUMAB (Kevzara, Regeneron and Sanofi) are INTERLEKIN (IL)-6 inhibitors. Both are being studied in patients for their ability to calm the cytokine storm in COVID-19 which is the overactive inflammatory response that occurs in the lungs causing acute respiratory distress syndrome. Tocilizumab is now being studied at Harvard’s Massachusetts General Hospital. International J of Inf Dis 8/6/20: N Potere “Disease progression was experienced by none of the (low dose) tocilizumb-treated patients vs by 5 (50%) patients in the standard of care group. Overall, experts concluded that in hospitalized patients with moderate COVID-19 and hyperinflammation.”
5/2020 Pluristem has pioneering regenerative cell therapy platform, with a focus on the clinical development of a placenta-based treatment for complications associated with COVID-19. The PLX cell-therapy treated six critically ill coronavirus patients who were considered high-risk for mortality – all of whom survived.
Electroceutical fabric eradicates coronaviruses on contact
MedicalXpress MD Linx 5/19/20 “Coronavirus particles attach to Personal Protective Equipment/PPE surfaces spreading the virus. Indiana University researcher Chandan Sen et al published in ChemRxiv pre-print that coronaviruses are killed on exposure to the electroceutical fabric. That fabric is a matrix of embedded microcell batteries creating moisture-activated microcell batteries when moistened. The ability of the virus to infect is fully eliminated within one minute of contact with this fabric that disrupts the electrostatic forces the virus needs. The fabric is called V.Dox Technology and is a proprietary dot-matrix pattern of embedded microcell batteries: it is used as a broad-spectrum antimicrobial wound care dressing in the management of infected wounds as a non-antibiotic solution.”
FDA to Evaluate Opaganib in Patients In Moderate to Severe COVID-19
Brian Park in MPR: 5/11/20 Opaganib is a first-in-class, orally-administered, sphingosine kinase-2 selective inhibitor with anticancer, anti-inflammatory and antiviral properties. It reduces interleukin-6 and tumor necrosis factor-alpha both elevated in CoV2-19. 6 hospitalized patients treated with opaganib decreased oxygenation requirements, higher lymphocyte counts, and decreased C-reactive protein (CRP). Clinical improvement occurred in patients with and without hydroxychloroquine. Opaganib was well tolerated. All 6 patients were weaned from oxygen and discharged from the hospital.
TREATMENT of the CYTOKINE STORM:
by Cincinnati Children’s Hospital Medical Center 5/28/20
Patients taking ruxolitinib were randomly selected to receive two daily 5mg oral doses plus the standard of care treatment for COVID-19. “Ruxolitinib recipients had a numerically faster clinical improvement”… Significant chest CT improvement, a faster recovery from lymphopenia (low lymphocyte count), a favorable side-effect profile, & a shorter median time to clinical improvement compared to the control group.” 90 % of ruxolitinib patients showed CT scan improvement within 14 days, compared with 9 percent of control patients. Three patients in the control group died of respiratory failure. All the severely ill patients who received ruxolitinib survived. “This is the first therapy we know that appears to work effectively to quiet the cytokine storm in severe COVID-19 disease, and there are no significant toxicities to patients who take the drug at two pills a day,” said Yang Cao et al, J of All and Clin Immunol (2020). DOI: 10.1016/j.jaci.2020.05.019
High Dose IVIG to Be Investigated for Severe COVID-19
Brian Park, PharmD MPR 5/21/20 The Food and Drug Administration (FDA) has approved Octapharma’s Investigational New Drug Application (IND) allowing the Company to initiate a phase 3 trial of Octagam® (intravenous human immune globulin) in patients with coronavirus disease 2019 (COVID-19) with severe disease progression.
TREATMENTS per Trend In Medicine 5/7/20
Treatments Gilead Sciences’ remdesivir, a direct-acting antiviral (an RNA polymerase inhibitor), was granted an EUA – not approval – for treating hospitalized Covid-19 patients. This is the first drug with an EAU for treating Covid-19 that has randomized trial data to back it up. Dr. Fauci called the results of that trial “quite good news,” adding, “This is really quite important. What it has proven is that a drug can block this virus.” He said remdesivir is now the “new standard of care” for all other trials. In preliminary results from the 1,063-patient ACTT trial (at 47 sites in the U.S and 21 in Europe and Asia) sponsored by NIAID, remdesivir was shown to shorten the time to recovery from 15 days down to 11 days, on average, a significant improvement vs. placebo (10 days after onset of symptoms). When data were pooled across treatment arms, by day 14, 62% of patients treated early were able to be discharged from the hospital vs. 49% of patients who were treated late. A different analysis noted: intubated placebo group started to have better survival than remdesivir. There was no benefit to high flow. Some benefit to supplemental oxygen group. No benefit to no oxygen group. Viral data is in the supplement: there is NO virologic, immunologic, or biochemical data to support remdesivir. Treatment group (remdesivir) and control group were not very similar. 23.1% mechanically ventilated patients in remdesivir group and 28.2% in control group.
Almost 20% difference.
Remdesivir is also being studied with baricitinib that is currently marketed under the brand name Olumiant for the treatment of rheumatoid arthritis.
No new safety signals were identified.
The most common adverse events with both regimens were nausea (10.0% vs. 8.6%), acute respiratory failure (6.0% vs. 10.7%). Grade ≥3 ALT elevations occurred in 7.3% of patients, with 3.0% discontinuing treatment as a result.
Here is how SARS CoV2-19 actually takes over human cells and reduplicates itself: “Kinases”
SARS-CoV-2 turns on a cellular switch to build the tubes called filopodia – that might help viral particles – the little spheres – spread more easily. Dr Elizabeth Fischer, NIAID NIH / Bouhaddou et al. © Elsevier 2020, CC BY-ND
Coronavirus and cancer hijack the same parts in human cells to spread – and our team identified existing cancer drugs that could fight COVID-19
June 28, 2020 10.05am EDT
Author
Nevan Krogan: Professor and Director of Quantitative Biosciences Institute & Senior Investigator at the Gladstone Institutes, University of California, San Francisco
Most antivirals in use today target parts of an invading virus itself. Unfortunately, SARS-CoV-2 – the virus that causes COVID-19 – has proven hard to kill. But viruses rely on cellular mechanisms in human cells to help them spread, so it should be possible to change an aspect of a person’s body to prevent that and slow down the virus enough to allow the immune system to fight the invader off.
I am a quantitative biologist, and my lab built a map of how the coronavirus uses human cells. We used that map to find already existing drugs that could be repurposed to fight COVID-19 and have been working with an international group of researchers called the QBI Coronavirus Research Group to see if the drugs we identified showed any promise. Many have.
For years, researchers have suspected that kinases – biological control switches that viruses use to take over cells – could be targeted to fight infections. Over the last few months, we built a second, more detailed map looking specifically for the kinases that the coronavirus is hijacking.
Using this map, we identified a few already existing cancer drugs which alter the function of the kinases that SARS-CoV-2 hijacks, and began testing them in coronavirus-infected cells. The results of these early tests are promising enough that we are working with some groups and have already begun human clinical trials.
This map shows how the coronavirus changes the function of kinases – cellular switches involved in most biological processes – and the proteins they control. It guided researchers from UCSF to cancer drugs that could fight COVID-19. Bouhaddou et al. © Elsevier 2020, CC BY-ND
Kinases in disease and as drug targets
Kinases are proteins found in every cell of our body. There are 518 human kinases, and they act as major control hubs for virtually all processes in the body. They are able to add a small marker – a process called phosphorylation – to other proteins and thus change how, if and when a phosphorylated protein can do its work.
For example, if a cell is preparing to grow – say to heal a cut on your finger – specific kinases will turn on and start telling proteins involved in cell growth what to do. Many cancers are caused by overactive kinases leading to uncontrolled cell growth, and drugs that slow kinases down can be highly effective at treating cancer.
Kinases are central players in cellular function as well as in most diseases, so researchers and pharmaceutical companies have studied them in great detail.
Kinases are also fairly easy to target with drugs because of how they add phosphorylation markers to proteins. Researchers have developed a huge number of drugs, particularly cancer drugs, that work by essentially throwing a wrench into the mechanics of specific kinases in order to stop cell growth.
So what does this have to do with the coronavirus? Well, viruses and cancer actually have more in common than you might think. Cancer is essentially a malfunctioning of cellular machinery that causes runaway cell growth.
Viruses also change the function of cellular machinery – albeit on purpose – but instead of causing cell growth, the machinery is repurposed to produce more viruses. Not surprisingly, viruses take control over many kinases to do this.
Microscope image shows a cell infected with SARS-CoV-2 and filopodia growths (in white) extending out from the cell surface containing viral particles (in red). Dr. Robert Grosse, CIBSS, University of Freiburg / Bouhaddou et al. © Elsevier 2020, CC BY-ND
Coronavirus at the controls
This idea – that SARS-CoV-2 is using kinases to hijack cellular machinery – is why we wanted to build a map of every kinase that is taken over by the coronavirus. Any virus–kinase interaction could be a potential target for drugs.
To do this, we first infected green monkey cells – which are fairly good surrogates for human cells when it comes to virus infection – with SARS-CoV-2. We then ground up these infected cells and used a device called a mass spectrometer to see which proteins were phosphorylated in these infected cells. We then did the same thing with healthy cells.
It is impossible to actually see which kinases are activated at any time, but since each kinase can attach phosphorylation markers to only a few specific proteins, researchers can look at the phosphorylated proteins to determine what kinases are active at any time.
We made two lists: one list of phosphorylated proteins in healthy cells and one list of phosphorylated proteins in infected cells. We then compared the two, and by looking at the differences between the infected and uninfected lists, we were able to determine which kinases the coronavirus uses to reproduce.
Because researchers still don’t fully understand what all 518 human kinases do, we were able to look for effects in only 97 of the ones we know most about. But that turned out to be more than enough. Of those 97 kinases, we found 49 that the virus affects.
Some of the more interesting ones include Casein Kinase 2, which is involved in controlling how a cell is shaped. We also identified several kinases that work together in what is called the p38/MAPK signaling pathway. This pathway responds to and controls our body’s inflammation reaction. It is possible these kinases could be involved in the cytokine storm – a dangerous immune system overreaction – that some patients with severe COVID-19 experience.
While identifying the kinases involved in SARS-CoV-2 replication, we were also able to learn a lot about how the virus changes our bodies. For example, CK2 becomes much more active during the course of coronavirus infection and causes the growth of little tubes that extend from the surface of the cell. Under a microscope, it looks as if the cell has a full head of hair. We think SARS-CoV-2 might be using these long cell outgrowths – called filopodia – as viral highways to get new viruses closer to neighboring cells, thereby making infection easier.
Testing the promising cancer drugs in the lab was the first step, and after dozens showed promise, we began the process of starting clinical trials. QBI UCSF, CC BY-ND
Kinases inhibitors in the lab and clinical trials
Learning more about the virus’s function is interesting for a biologist like me and could be useful down the road, but the ultimate goal of our project was to find drugs to treat COVID-19.
Once we knew which kinases SARS-CoV-2 uses to replicate and the proteins they change, we looked through a database of around 250 kinase-inhibiting drugs to see if any of them targeted the kinases used by the virus. To increase our chances, we also looked for drugs that hit some of the proteins the kinases act on. And sure enough, we found some.
There are 87 existing drugs that change the kinase-controlled pathways used by the coronavirus. Most of these drugs are already approved for human use or are currently in clinical trials to treat cancer, and could be quickly repurposed to treat COVID-19 patients.
With these leads, our collaborators in New York and Paris tested the effect of 68 of those drugs on cells infected with SARS-CoV-2. Several of these were effective in killing the virus in cells. A few that we are especially excited about – silmitasertib, gilteritinib, ralimetinib, apilimod and dinaciclib – are either approved for treatment, in clinical testing or under preclinical development for various diseases.
Silmitasertib stops Casein Kinase 2, the kinase that causes cells to grow the virus spreading filopodia tubes. As soon as the company that makes silmitasertib heard this news, they announced that they wanted to test the drug against COVID-19 in the clinic.
Drugs hitting kinase pathways have been on the radar of researchers as potential powerful antivirals for years, but none have come to fruition. By looking to this new area of drug applications and using our new mapping approach, our team has added dozens of drugs to the growing list of potential tools to help fight this pandemic.
CONVALESCENT PLASMA
There have been anecdotal reports of the efficacy of this therapy. It has been used in 7,200 people in the past several months. Data on those patients are being analyzed, will be released in a couple of weeks, and should offer some useful insights, but it is not a randomized study.
The FDA issued guidance for healthcare providers and investigators on the administration and study of investigational convalescent plasma collected from people who have recovered from Covid-19, with recommendations on patient eligibility, donor eligibility/qualifications, labeling, recordkeeping, and more.
Johns Hopkins plans to start enrolling patients into two randomized clinical trials of convalescent plasma in the outpatient setting, with results expected in a couple of months. This is a preventive study in nursing home patients to see if convalescent plasma will prevent them from catching Covid-19. Also there is a treatment study in people with confirmed Covid-19 who are remaining at home to see if giving them convalescent plasma at home will prevent them from worsening to the point they need to be hospitalized. Asked how many recovered Covid-19 patients would qualify to donate plasma, Arturo Casadevall, MD, chair of the Department of Molecular Microbiology and Immunology at the Johns Hopkins Bloomberg School of Public Health, said, “The majority have high titers of antibodies…However, a small percentage have antibodies but not very high levels…And it depends on the antibody test used. They (antibody tests) are not standardized very well. We are using an Elisa test developed at Mount Sinai.”
* Hydroxychloroquine (HCQ). President Trump is one of the few people still speaking positively about HCQ, though he isn’t pushing it as hard as he used to do. Some states are still stockpiling it. So, is it safe? Does it work? TPro and con information exists.
A 568-patient retrospective Chinese study, available as a preprint on medRxiv.org, looked at critically ill Covid-19 patients who had severe acute respiratory distress syndrome (ARDS) despite antiviral + antibiotic therapy. Of the 568 patients, 48 also received HCQ (200 mg BID for 7-10 days). Mortality (the primary endpoint) was 18.8% with HCQ vs. 45.8% without it. Length of stay before death was 15 days with HCQ and 8 days without it. Trends-In-Medicine May 7, 2020 Coronavirus IL-6 levels were significantly lowered by the end of treatment with HCQ but not without it. The researchers concluded that HCQ significantly decreased mortality in critically ill patients through attenuation of inflammatory cytokine storm and should be prescribed for treatment of critically ill Covid-19 patients.
A 1,061-patient retrospective analysis of HCQ in Marseille, France, in preprint, in which HCQ was combined with azithromycin, found that: 91.7% had a good clinical outcome and virological cure within 10 days. 4.3% of patients had a poor clinical outcome, and 8 died (0.75%). All the deaths were from respiratory failure, not cardiac toxicity. Poor clinical outcome was associated with older age, severity at admission, and low HCQ serum concentration. The researchers concluded that the combination of HCQ
+ azithromycin is safe and associated with a very low fatality rate.
The negative news. A report on 90 Covid-19 patients treated at a Boston hospital, published in JAMA Cardiology, found a potential for serious cardiac arrhythmias – significant QTc prolongation (>500 ms). One patient developed torsade de pointe when given HCQ + azithromycin.
Roche’s Actemra (tocilizumab). This anti-IL-6R met the primary endpoint in the 129-patient French CORIMUNO-19 trial in hospitalized patients with moderate-to-severe Covid-19, with significantly fewer patients needing ventilation (mechanical or non-invasive) or dying by Day 14. . This drug makes sense because it is already used to treat cytokine storms in immunotherapy patients, and a key issue with Covid-19 is cytokine storm. The WHO announced the launch of Access to COVID-19 Tools Accelerator, a global project focused on developing and producing new treatments, vaccines, and tests for Covid-19, while ensuring global access to the products. Among other therapies to add to the long list of medications in development to treat Covid-19 are:
Study shows the experimental drug AR-12 could be a promising COVID-19 treatment
by Virginia Commonwealth University Massey Cancer Center 9/21/20
A team of scientists led by Paul Dent, Ph.D., at Virginia Commonwealth University Massey Cancer Center has discovered that an experimental cancer drug called AR-12 inhibits the SARS-CoV-2 virus from infecting cells and replicating. Published online 9/21/20 in Biochemical Pharmacology,
AR-12 has been studied as both an anti-cancer and anti-viral drug, with prior peer-reviewed publications from Dent and others showing it to be effective against viruses including Zika, mumps, measles, rubella, chikungunya, RSV, CMV, drug resistant HIV and influenza. Recently, collaboration with Jonathan O. Rayner, Ph.D., at the U of S Alabama and Laurence Booth, PhD, from Dent’s lab, has demonstrated that AR-12 is highly effective against SARS-CoV-2.
“AR-12 works in a unique way. Unlike any other anti-viral drug, it inhibits cellular chaperones, which are proteins that are required to maintain the right 3-D shape of viral proteins. The shape of the virus is critical to its ability to infect and replicate.” One of the cellular chaperones inhibited by AR-12 is GRP78, which is essential for the reproduction of all viruses. GRP78 acts as a sort of cellular stress sensor and is required for the life cycle of all mammalian viruses.
AR-12 is an oral therapy will be from C19 Therapeutics, which recently licensed AR-12 from VCU. Another observation made in Dent’s research may also shed light into why African Americans have been more affected by severe illness during the COVID-19 pandemic. People of non-European descent, particularly those with African ancestry, make a protein called ATG16L1 T300, while those with primarily European ancestry make a different variant, ATG16L1 A300.
“We found that cells making the T300 form made more GRP78 and more of the virus receptor ACE2,” said Dent. “This, of course, does not prove that those with the T300 form are more susceptible to COVID-19, but it provides a biomarker that could be evaluated to help explain why some people get more severe illness than others.”
More information: Jonathan O. Rayner et al. AR12 (OSU-03012) suppresses GRP78 expression and inhibits SARS-CoV-2 replication, Biochemical Pharmacology (2020). DOI: 10.1016/j.bcp.2020.114227
Bradykinin inhibitors to prevent the hydrogel pneumonia of CoV2-19, the longer acting LANADELUMAB (Takhzyro) and the shorter acting icatibant (Firazyr)
AbCellera and Lilly are collaborating on research for development of an antibody to treat Covid-19, and AbCellera got some help (up to $175.6 million) from the Canadian government’s Innovation, Science, and Economic Development Canada Strategic Innovation Fund.
BerGenBio’s bemcentinib, an oral selective AXL inhibitor – A 120-patient Phase II trial has started in the U.K. in hospitalized Covid-19 patients.
CAR T – Researchers at Duke-NUS Medical School in Singapore are studying whether there might be utility for CART and/or TCR-T therapies in Covid-19.
Karyopharm Therapeutics’ Xpovio (selinexor) – The company announced the first patient was dosed with this cancer drug in a Phase II trial in severely ill Covid-19 patients.
Johnson & Johnson and Merck’s H2 blocker Pepcid (famotidine) – given IV at a dose 9-times the over-the-counter dose of this heartburn drug – is being tested in a clinical trial in New York City by Northwell Health.
Sarepta Therapeutics is initiating a discovery program to see if some of its antisense oligonucleotides can inhibit viral infection.
Trends-In-Medicine 5/7/20: Coronavirus Re-purposed Drugs Being Investigated for Covid-19 Company Alexion Pharmaceuticals Ultomiris ravulizumab C5 complement inhibitor.
Amgen Otezla apremilast PDE4 inhibitor.
AstraZeneca Farxiga dapagliflozin SGLT2 inhibitor Johnson & Johnson
Novartis and Incyte Jakafi ruxolitinib oral JAK inhibitor (for ventilator patients).
Novartis Cosentyx secukinumab anti-IL-17A Diovan valsartan ARB Ilaris canakinumab interleukin-1 inhibitor Xolair omalizumab.
IgE inhibitor Pulmotect –- inhaled superoxide –- Synairgen –- SNG-001 inhaled interferon beta-1a
VACCINES:. 10/2020: an entirely NEW approach that generates antibodies and T-CELL activation is that of Dr. Partick Soon-Shiong’s Immunity Bio.
A research team led by Dr. Larenas-Linnemann working at Medica Sur, Mexico City, reported clinical observations in 255 subjects vaccinated with the mumps-measles-rubella (MMR) vaccine since the start of the COVID-19 pandemic. Many vaccinated patients were family members or caregivers of patients who already had contracted COVID-19, and were thus at extremely high risk. Thirty-six of the patients have now contracted COVID-19, but all with a remarkably mild course, experiencing less severe symptoms than would be expected given their health status and age. The paper is published in the September, 2020, issue of Allergy, the European journal of allergy and immunology.
This is an excellent 7/6/20 J of the Amer Med Assoc article that is both lucid and technically complete about the development of SARS-CoV2-19 Vaccines at WARP-SPEED: my predictions is that one will be ready by 10/15/20.
https://jamanetwork.com/journals/jama/fullarticle/2768155?guestAccessKey=45fbb871-b6cc-4599-b191-c57d248525dd&utm_source=silverchair&utm_medium=email&utm_campaign=article_alert-jama&utm_content=olf&utm_term=070620
AstraZeneca is collaborating with Oxford University on the vaccine Oxford developed. Oxford took an existing chimp vaccine and engineered it to work for SARS-CoV-2, did efficacy studies in monkeys, and has now started a Phase I safety trial in healthy volunteers. The researchers predicted the vaccine could be ready by fall 2020. Leukocare, ReiThera, and Univercells are collaborating on development of a novel adenoviral vector-based vaccine for Covid-19. They launched a clinical trial this past summer and begin manufacturing alongside clinical development. A single case of transverse myelitis occurred, the trial was stopped for 2 days in September, 2020, and then the trial was resumed. ‘Half-measure’ virus vaccine intrigues experts
by Kelly MacNamara 11/23/20 in Medical Xpress Evidence suggesting an initial half dose of the vaccine being developed by AstraZeneca and the University of Oxford is more effective than a full dose is counterintuitive, and even took the researchers by surprise.
Andrew Pollard, the director of the Oxford Vaccine Group, described the findings from the Phase 3 clinical trial as “intriguing”. They showed that the vaccine had an efficacy of 62 percent among the people given two full doses a month apart. But this rose to 90 percent for another group who received a half-dose first and then a full dose after a month.
Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccinations.
• Andreas Greinacher, Thomas Thiele, Theodore E. Warkentin, Karin Weisser, Paul A. Kyrle, and Sabine Eichinger NEJM 4/15/21
Several cases of unusual thrombotic events and thrombocytopenia have developed after vaccination with the recombinant adenoviral vector encoding the spike protein antigen of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (ChAdOx1 nCov-19, AstraZeneca).
11 patients in Germany and Austria in whom thrombosis or thrombocytopenia developed after vaccination with ChAdOx1 nCov-19. We used a standard enzyme-linked immunosorbent assay to detect platelet factor 4 (PF4)–heparin (IgG) antibodies detected by an ELISA assay and a modified (PF4-enhanced) platelet-activation test to detect platelet-activating antibodies. Included in this testing were samples from patients who had blood samples referred for investigation of vaccine-associated thrombotic events, with 28 testing positive on a screening PF4–heparin immunoassay.
Of the 11 original patients, 9 were women, with a median age of 36 years (range, 22 to 49). Beginning 5 to 16 days after vaccination, the patients presented with one or more thrombotic events, with the exception of 1 patient, who presented with fatal intracranial hemorrhage. Of the patients with one or more thrombotic events, 9 had cerebral venous thrombosis, 3 had splanchnic-vein thrombosis, 3 had pulmonary embolism, and 4 had other thromboses; of these patients, 6 died. Five patients had disseminated intravascular coagulation. None of the patients had received heparin before the rare development of immune thrombotic thrombocytopenia mediated by platelet-activating antibodies against PF4, which clinically mimics autoimmune heparin-induced thrombocytopenia. (Funded by the German Research Foundation.) This has been diagnosed with high D-dimer, etc. tests and successfully treated with a a blocking immunoglobulin infusion + steroids + non-heparin anticoagulants reported by the Department of Medicine I of MedUni Vienna and Vienna General Hospital, led by coagulation specialist Paul Knöbl and published in the 2021 Journal of Thrombosis and Haemostasis.
From December 2020 through March 2021, the European Medicines Agency ap proved four vaccines on the basis of randomized, blinded, controlled trials: two messenger RNA–based vaccines — BNT162b2 (Pfizer–BioNTech) and mRNA-1273 (Moderna) — that encode the spike protein antigen of SARS-CoV-2, encapsulated in lipid nanoparticles; ChAdOx1 nCov-19 (AstraZeneca), a recombinant chimpanzee adenoviral vector encoding the spike glycoprotein of SARS-CoV-2; and Ad26.COV2.S (Johnson & Johnson/Janssen), a recombinant adenovirus type 26 vector encoding SARS-CoV-2 spike glycoprotein.
As of April 7, 2021, more than 82 million vaccine doses had been administered in the European Union; in Germany, approximately one quarter of vaccine recipients had received the ChAdOx1 nCov-19 vaccine.1
Moderna is now fast-tracked by the FDA because if its actual testing success. From L A Jackson et al in the 7/14/20 NEJM: “The mRNA-1273 vaccine candidate, manufactured by Moderna, encodes the S-2P antigen, consisting of the SARS-CoV-2 glycoprotein with a transmembrane anchor and an intact S1–S2 cleavage site. S-2P is stabilized in its prefusion conformation by two consecutive proline substitutions at amino acid positions 986 and 987, at the top of the central helix in the S2 subunit.8 The lipid nanoparticle capsule composed of four lipids was formulated in a fixed ratio of mRNA and lipid.” The Moderna vaccine has proven 95% effective in Stage III trials
Results of Phase 1 Trial of Recombinant Adenovirus type-5 Vectored COVID-19 Vaccine
Bradley van Paridon July 7, 2020
ELISA antibodies and neutralizing antibodies were found to have increased significantly by day 14 and peaked 28 days following vaccination.
Data published in The Lancet, this Ad5-vectored COVID-19 vaccine warrants further investigation.
Dose-escalation, single-center, open-label, nonrandomized phase 1 trial in Wuhan, China (ClinicalTrials.gov Identifier: NCT04313127). In total, 108 adults between age 18 and 60 years (51% men, 49% women, and mean age 36.3 years): 3 groups of 36 and received either a low, middle, or high dose of the vaccine.
At least 1 adverse reaction was reported in 83% of individuals in the middle- and low-dose groups and in 75% of individuals in the high-dose groups within the first 7 days following vaccination. Pain was the most common injection: 54%. Systemic adverse reactions: fever (46%), fatigue (44%), headache (39%), and muscle pain (17%). Most reactions were mild or moderate, there was no serious adverse event noted within 28 days.
Day 14, enzyme-linked immunosorbent assay (ELISA) antibodies and neutralizing antibodies increased significantly and peaked 28 days following vaccination. The specific T-cell response peaked at day 14 following vaccination. 7 of 8 ferrets in preclinical studies were protected from having detectable virus when challenged through nasal dripping 21 days after immunization compared with 1 of 8 in the control group. No participant in the trial was older than age 60 and only 16% were older than 50.
The investigators conclude that the Ad5-vectored COVID-19 vaccine is tolerable and immunogenic in health adults, adding, “there is potential for further investigation ofthe Ad5 vectored COVID-19 vaccine for the control of the COVID-19 outbreak.” An ongoing phase 2 trial (ClinicalTrials.gov Identifier: NCT04341389) is set.
Reference
Zhu F-C, Li Y-H, Guan X-H, et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial. Lancet. 2020; 395:1845-1854.
Gene-based (as opposed to protein-based mRNA vaccines “carry the genetic instruction to make the antigen, which closely mimics a natural infection… In this case “it is not the spike itself, but the genetic material that that instructs the cells how to make that spike protein” to which the cells then make an antibody to that spike protein that is protective according to U of Pennsylvania vaccinologist Paul Offit. The entire process is simpler, the mRNA is quickly synthesized in a tank within a week or so, much faSter than the alternative. “In addition to eliciting antibodies and CD4+ helper T cells, they recruit CD8+ cytotoxic T cells, also known as killer T cells, through the major histocompatibility class I pathway.” “Protein-based vaccines (are grown in eggs or cells, a time consuming and costly process) deliver the immune system-stimulating antigen to the body. This includes whole inactivated (killed) vaccines as in polio and flu shots and subunit vaccines and virus-like particles like in the hepatitis B and human papilloma virus vaccines.”
First human trial of COVID-19 vaccine finds it is safe and induces rapid immune response
by The Lancet 5/2020
The first COVID-19 vaccine to reach phase 1 clinical trial has been found to be safe, well-tolerated, and able to generate an immune response against SARS-CoV-2 in humans, according to new research published in The Lancet. The open-label trial in 108 healthy adults demonstrates promising results after 28 days—the final results will be evaluated in six months. A single dose of the new adenovirus type 5 vectored CoV2-19 (Ad5-nCoV) vaccine produces virus-specific antibodies and T-cells in 14 days,” said the responsible Professor Wei Chen from the Beijing Institute of Biotechnology.
July 26, 2021
Clarifying the problem of T-cell ‘exhaustion’
by Perelman School of Medicine at the University of Pennsylvania
Researchers in the Perelman School of Medicine at the University of Pennsylvania have illuminated an important limitation of the immune system in prolonged battles against cancers or viruses: T cells, which are among the most powerful weapons in the immune systems of humans and other vertebrates, remain substantially programmed to stay exhausted even many weeks after exposure to a virus ended.
Scientists have known that T cells can lose their ability to fight viruses and tumors when they have prolonged exposure to these enemies. They have hoped that this “T cell exhaustion” phenomenon could be reversed relatively easily, for example when the T cells are no longer exposed to the virus or tumor in question.
“Our findings suggest that once T cells become exhausted, they remain fundamentally ‘wired’ to be exhausted—thus it may be hard to get them to become effective virus- and cancer-fighters again,” said study senior author E. John Wherry, PhD, chair of the department of Systems Pharmacology and Translational Therapeutics and director of the Penn Institute of Immunology.
The recognition of the T cell exhaustion problem emerged about two decades ago from studies of long-term viral infections, including studies by Wherry and his laboratory. Scientists eventually concluded that long-term exposure not only to viruses but also to cancerous tumors could exhaust T cells. Exhausted T cells start producing much lower amounts of immune response-stimulating proteins, and generally become less able to kill virus-infected cells or tumor cells.
There is evidence now that such reinvigoration, for example with cancer drugs called PD-1 inhibitors, tends to be incomplete and temporary.
Wherry and colleagues, including first author Mohamed Abdel-Hakeem, Ph.D., a postdoctoral research associate in the Wherry Laboratory, addressed this re-invigoration question in the new study. They examined mouse T cells that had been exhausted by chronic exposure to a mouse virus called LCMV—long used as a standard model of T cell-exhausting infection.
The researchers found that most of the exhausted T cells died when they were no longer exposed to LCMV. The small proportion that survived recovered some of the gene expression patterns that would be expected in normal, memory-type T cells that help sustain a long-term immune defense following infection. But for the most part, the T cells remained programmed to stay in the exhausted, ineffective state, especially when called into action again upon reinfection.
A cell is programmed to be in a certain state or identity by an ‘epigenetic’ system of molecules that control which genes are active or inactive in the cell. These molecules often work by altering the structure of coiled DNA in the nucleus of the cell, making some genes more accessible to gene-copying enzymes, and others less accessible. In the study, the researchers observed that the epigenetic, DNA-structure changes that are characteristic of exhausted T cells mostly remained stable in these cells after LCMV exposure ended.
“Exhaustion apparently leaves durable ‘epigenetic scars’ in T cells that constrain their ability to support an immune response. These findings point to a need to discover how to reverse that epigenetic scarring,” Wherry said.
In principle, future treatments that reverse those epigenetic changes could turn exhausted T cells in patients into normal memory T cells again. If the right epigenetic drugs can be found to modulate immune cells that could help patients fight ongoing cancers or viral infections, and could also provide patients with stronger long-term immunity following the eradication of tumors or chronic viral infections. Studies at Penn Medicine are ongoing
Transition to exhaustion: Clues for cancer immunotherapy
More information: Mohamed S. Abdel-Hakeem et al, Epigenetic scarring of exhausted T cells hinders memory differentiation upon eliminating chronic antigenic stimulation, Nature Immunology (2021). DOI: 10.1038/s41590-021-00975-5
The Trump administration was working on a Manhattan Project-style initiative, Operation Warp Speed, to spur rapid development of a SARS-CoV-2 vaccine, with the aim of having a vaccine ready for use by the end of this year. The hope is that 3-4 of the 14 promising vaccines already in development will survive and be successful.
Here are economic estimates of the COST for vaccines: the COVID vaccine will likely cost $35 per injection. 2-4 injections will be required for likely 300 million people in the US. 2-4 x 35 x 300 million = $21-42,000,000,000 – vs zinc & hydroxychloroquine & azithromycin, which costs around $20-40 for the whole protocol to be used only as necessary and not $300,000,000.
11/18/20 Trends-In-Medicine: There are more than 135 vaccines in development, and in the last 10 days two of those vaccines have reported positive results that scientific experts believe are credible –
Pfizer and BioNTech’s BNT162b2 and Moderna’s mRNA-1273. Russia approved a vaccine from the Gamaleya Research Institute, but the data have been slim to non-existent
and so are not convincing, and China has allowed use of a number of unapproved vaccines.
Based on early data, Pfizer said its vaccine was > 90% effective, but the final analysis announced this week, shows it is actually 95% effective overall and 94.5% effective in older adults. Moderna, which is developing its vaccine in partnership with the National Institute of Allergy and Infectious Diseases (NIAID), also says its early analysis shows its vaccine is 94.5% effective and expects to have the final analysis very soon. Both companies now have sufficient data on patients with ≥2 months of follow-up, which is an FDA requirement for an emergency use authorization (EUA).
Arcturus Therapeutics’ ARCT-021 – The company reported positive interim Phase I/II data for this mRNA vaccine, showing promising efficacy and safety with just one dose. CreVac – In interim Phase I data from a preprint on medRxiv , this mRNA-lipid nanoparticle vaccine showed positive results in a Phase I study, with two doses, given 28 days apart, boosted pre-existing immune responses, even at low doses.
And the vaccine was safe, with side effects generally mild and dose-dependent. The 12 g dose was chosen for the Phase IIb/III trial. Gamaleya Research Institute’s Sputnik V – The National Center of Epidemiology and Microbiology in Moscow claimed this Russian vaccine is 92% effective, but the analysis is based on just 20 cases. Johnson & Johnson’s Ad26.COV2.S (JNJ-78436735) – J&J added a new, 30,000-patient Phase III trial (ENSEMBLE 2) of this vaccine, this time using a two-dose regimen, given 56 days apart. It isn’t giving up on its one-dose approach. The trial is more of an insurance policy and to see if double-dosing extends the duration of protection. Novavax’s NVX-CoV2373, a nanoparticle vaccine with a proprietary MatrixM adjuvant, was granted fast track status by the FDA. Sinopharm claimed “better than expected” Phase III data for one of its two vaccines. In a statement posted on WeChat,
the company said that >50,000 volunteers are enrolled in its studies. Sinovac Biotech’s CoronaVac
The company said the vaccine has produced a quick immune response in an ~700-patient study, but the level of antibodies produced was lower than in people who had recovered from the disease.
A trial in Brazil was put on hold by ANVISA, the Brazilian regulatory authority, while the death of a participant is investigated. It turned out to be a suicide, and the trial was allowed to resume.
An Israeli coronavirus drug that claims to have a 100% success rate among severely ill patients is being tested in the United States for the first time.
CBNNews.com Emily Jones 04-16-2020
Pluristem Therapeutics Inc., a biotech in Haifa, reported that 7 who were at a high risk of death due to respiratory failure survived after receiving the medication.
The patients were treated with allogeneic placental expanded (PLX) cells under the compassionate use program and exhibited respiratory failure requiring intubation in the ICU. 4 of the patients had multi-system organ failure, including heart and kidney failure. These cells suppress or reverse the dangerous over-activation of the immune system that causes death in many coronavirus patients. Pluristem uses “donated placentas at the time of delivery of healthy, full-term babies, from healthy women under 35 years old, undergoing an elective caesarean section.”
All seven of the patients who received the drug survived and four patients saw an improvement in respiration. One patient who is still alive saw a continued deterioration of the respiratory system. Now, a critical COVID-19 patient in the US has been treated with PLX cell therapy at Holy Name Medical Center in New Jersey.
COLCHICINE “is a microtubule polymerization inhibitor and an inhibitor of interleukins 1 and 6, granulocyte macrophage colony stimulating factor, and the nucleotide-binding oligomerization leucine-rich repeat and pyrin domain (NLRP3) inflammasome, making it a potent anti-inflammatory agent” that results in a a smaller increase in dimerized plasmin fragment D (D-dimer) in the GRECCO study whch demonstrated much less clinical deterioration in those treated with colchicine. “(Colcrys, Mitigare; Takeda Pharmaceuticals) is an inexpensive, FDA-approved, powerful anti-inflammatory drug used to treat gout and pericarditis. It’s currently being studied for its usefulness in mitigating the cytokine storm caused by the novel coronavirus. Researchers at the Montreal Heart Institute and the U of Montreal hope that colchicine can stop the body’s overproduction of immune cells and cytokines (chemical messengers), which leads to the cytokine storm (an hyperinflammatory state) that damages lung tissue, acute respiratory distress, and multi-organ failure. From Newsletter Science X: Colchicine is different, said researcher Dr. Priscilla Hsue, a professor of medicine at the University of California, San Francisco (UCSF). “One of the unique aspects is that we’re trying to hit this before people need to be hospitalized,” Hsue said. Colchicine is the medication of choice for a few reasons, Hsue explained: unlike drugs tested in hospitalized patients given by infusion or injection, colchicine is easy to take by mouth, inexpensive, and has a long history of safe use, she added.”
Colchicine in JAMA 6/2020
https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2767593?utm_source=silverchair&utm_medium=email&utm_campaign=article_alert-jamanetworkopen&utm_content=wklyforyou&utm_term=062420
7/14/20: Israeli researcher Y Nahmias of Hebrew University and B tenOever at NYC’s Mt. Sinai Hospital reported online in Cell Press and the Times of Israel newspaper, that the very old triglyceride lowering medicine fenofibrate inhibits lung damage related to fat deposition caused by the corona virus. Fenofibrate stops the virus from interfering with the body’s ability to breakdown carbohydrate leading to those cells using lipids/triglycerides =fat which then piles up damaging the lungs and other organs, but also allowing the virus to replicate. Fenofibrate acts on the human DNA site that the virus shuts down, restarting the body’s ability to break down fat, thereby stopping the toxic buildup of fat in the lung and viral replication.
Licensed drug could reduce SARS-CoV-2 infection by up to 70 per cent, reveals study
by University of Birmingham 8/6/21
A licensed drug, fenofibrate, normally used to treat abnormal levels of the blood fat triglyceriedes reduces infection caused by the SARS-CoV-2 virus by up to 70 percent.
The University of Birmingham and Keele University in the UK and the San Raffaele Scientific Institute in Italy, has demonstrated that fenofibrate and its active form (fenofibric acid) reduces SARS-COV-2 infection in human cells in the laboratory. Reduction of infection was obtained in standard safe clinical doses that treat high levels of the blood fat triglycerides.
The team is now calling for clinical trials to test the drug in hospitalized COVID-19 patients, to be carried out in addition to two clinical trials also currently underway in such patients in research being led by the Hospital of the U of Pennsylvania in the US and Hebrew University in Israel.
The team identified fenofibrate as a candidate, they then tested the efficacy of the drug in reducing infection in cells in the laboratory using the original strains of the SARS-CoV-2 virus isolated in 2020. They found fenofibrate reduced infection by up to 70%. Additional unpublished data also indicates that fenofibrate is equally effective against the newer variants of SARS-CoV-2 including the alpha and beta variants and research is ongoing into its efficacy in the delta variant.
Co-author Dr. Elisa Vicenzi, of the San Raffaele Scientific Institute in Milan, Italy, said: “Our data indicates that fenofibrate has the potential to reduce the severity of COVID-19 symptoms and also virus spread. Given that fenofibrate is an oral drug which is inexpepensive and available worldwide, together with its extensive history of clinical use and its good safety profile, our data has global implications—especially in low-middle income countries and in those individuals for whom vaccines are not recommended or suitable such as children, those with hyper-immune disorders and those using immune-suppressants.”
First author Dr. Scott Davies, also of the University of Birmingham, concluded: “We need clinical studies to establish fenofibrate is a therapeutic agent to treat SARS-CoV-2 infection.”
The research, published today in Frontiers in Pharmacology, was also carried out in collaboration with the University of Copenhagen in Denmark and the University of Liverpool in the UK.Frontiers in Pharmacology, DOI: 10.3389/fphar.2021.660490
7/15/20 Medical Xpress “Enzalutamide blocks signals of the male sex hormone, testosterone, which in turn affects the enzyme TMPRSS2, among others. This is the same enzyme that the virus SARS-CoV-2 uses to get into and harm lung cells.
Awaited Ivermectin Review Is Out
— Findings are positive, but critics still want a large randomized controlled trial
by Kristina Fiore, Director of Enterprise & Investigative Reporting, MedPage Today July 8, 2021
Proponents of ivermectin for COVID-19 have long been talking about an expected review and meta-analysis led by Andrew Hill, PhD, of the University of Liverpool.
These results were finally published this week in Open Forum Infectious Diseases, and they’re positive — but they haven’t escaped criticism, and most researchers still want results from a randomized controlled trial.
The review and meta-analysis was conducted as part of the International Ivermectin Project Team from December 2020 to May 2021. Ivermectin proponents said Hill was conducting the analysis for the WHO, but Hill recently told MedPage Today the analysis was supported by Fort Worth, Texas-based Rainwater Charitable Foundation, and not WHO.
Hill and colleagues assessed 24 randomized trials totaling 3,328 patients that involved some type of control, whether it was standard of care or another therapy. Sample sizes ranged from 24 to 400 participants. Eight of the studies had been published, nine were preprints, six were unpublished results shared for the analysis, and one was reported on a trial registry website.
In the 11 trials (totaling 2,127 patients) that focused on moderate or severe infection, there was a 56% reduction in mortality (relative risk [RR] 0.44, 95% CI 0.25-0.77, P=0.004), with 3% of patients on ivermectin dying compared with 9% of controls.
But the researchers noted that the total number of deaths was small (128) and there was no difference between ivermectin and controls in the subgroup with severe disease. As for moderate disease, they reported a 70% improvement in survival with ivermectin (RR 0.30, 95% CI 0.15-0.58, P=0.0004).
Use of ivermectin was also associated with a reduction in time to recovery of 1.58 days compared with controls (95% CI -2.8 to -0.35, P=0.01) and with a shorter duration of hospitalization (-4.27 days, 95% CI -8.6 to -0.06, P=0.05).
However, the drug was not associated with a lower risk of hospitalization, though a sensitivity analysis that included any hospitalizations within 12 hours of taking the drug did show a reduction with ivermectin (RR 0.32, 95% CI 0.13-0.80, P=0.01).
Many studies included in the analysis were not peer reviewed, which was a limitation; additionally, studies varied widely in terms of dosage, treatment duration, and inclusion criteria. Studies also included a range of comparators, including hydroxychloroquine, lopinavir/ritonavir, standard of care, and placebo.
The authors concluded that their results “need to be validated in larger confirmatory trials” — a fact that David Boulware, MD, MPH, of the University of Minnesota, who has been interested in evaluating ivermectin for COVID-19 outpatients, agreed with. His study was retracted for lack of verifiable data.
As for the outpatient findings, he noted that only two of seven trials showed a reduction in symptom duration, and that a small sample size didn’t offer adequate analysis as to whether hospitalization risk was reduced by early treatment.
“Thus, there remains a need for phase 3 randomized clinical trials testing early ivermectin treatment to be completed in order to delineate what is the clinical benefit of early treatment? Quicker resolution of symptoms? Less hospitalization?” he tweeted.
One Argentinian randomized controlled trial of 500 patients recently published in BMC Infectious Diseases found that ivermectin didn’t prevent hospitalization among outpatients with COVID-19.
Satoshi Omura, Nobel Prize winner for his work on Ivermectin, was censored from YouTube for daring to discuss his work.
IVERMECTIN is a safe single dose treatment effective in reducing the virus. The ScienceDirect journal, Antiviral Research, research from Monash U’s K Wagstaff, MD, in Melbourne, Australia. The approved and safe common anti-parasite Ivermectin has broad spectrum antiviral activity and is effective inhibiting the coronavirus that causes COVID-19. Ivermectin is an inhibitor of the COVID-19 causative virus ARS-CoV-2) in the TEST TUBE. A single treatment was able to effect ∼5000-fold reduction in virus at 48h in cell culture.
https://www.breitbart.com/border/2020/04/04/common-anti-parasite-drug-may-kill-coronavirus-in-under-48-hours-say-researchers/?utm_source=newsletter&utm_medium=email&utm_term=todays_hottest_stories&utm_campaign=20200404
Anti-COVID-19 efficacy of ivermectin
Guilherme Dias de Melo, Françoise Lazarini, Florence Larrous, Lena Feige, Lauriane Kergoat, Agnès Marchio, Pascal Pineau, View ORCID ProfileMarc Lecuit, Pierre-Marie Lledo, Jean-Pierre Changeux, Hervé Bourhy
doi: https://doi.org/10.1101/2020.11.21.392639
The use of the anti-parasitic drug ivermectin (IVM), has been proposed, given its possible anti-SARS-CoV-2 activity9. Ivermectin is a positive allosteric modulator of the α-7 nicotinic acetylcholine receptor10, a target for the control of Covid-19 infection11, with a potential immunomodulatory activity12. We assessed the effects of IVM in SARS-CoV-2-intranasally-inoculated golden Syrian hamsters. Even though ivermectin had no effect on viral load, SARS-Cov-2-associated pathology was greatly attenuated. Ivermectin dramatically reduced the Il-6/Il-10 ratio in lung tissue, which likely accounts for the more favorable clinical presentation in treated animals. Our data support IVM as a promising anti-COVID-19 drug candidate.
Effectiveness of a multidrug therapy consisting of ivermectin, azithromycin, montelukast and acetylsalicylic acid to prevent hospitalization and death among ambulatory COVID-19 cases in Tlaxcala, Mexico International Society for Infectious Diseases.
René Lima-Morales, et al
Open Access Published: 2/9/21DOI: https://doi.org/10.1016/j.ijid.2021.02.014
TNR4 is a multidrug therapy (Ivermectin, Azithromycin, Montelukast and ASA) for COVID-19 cases.
TNR4 increased the likelihood of recovery 3.4 times in ambulatory COVID-19 cases.
The multidrug therapy TNR4 reduced the risk of hospitalization by 75%.
The multidrug therapy TNR4 reduced the risk of death by 81%.
There is an urgent need for effective treatments to prevent or attenuate lung and systemic inflammation, endotheliitis, and thrombosis related to COVID-19. The aim of this study was to assess the effectiveness of a multidrug-therapy consisting of Ivermectin, Azithromycin, Montelukast and Acetylsalicylic Acid (“TNR4” therapy) to prevent hospitalization and death among ambulatory COVID-19 cases in Tlaxcala, Mexico.
A comparative effectiveness study was performed among 768 confirmed SARS-CoV-2 cases aged 18 to 80 years, who received ambulatory care at the Ministry of Health of Tlaxcala. A total of 481 cases received the TNR4 therapy, while 287 received another treatment (comparison group). All participants received home visits and/or phone calls for clinical evaluation during the 14 days after enrollment.
Nearly 85% of cases who received the TNR4 recovered within 14 days compared to 59% in the comparison group. Likelihood of recovery within 14 days was 3.4 times greater among the TNR4 group than in the comparison group. Patients treated with TNR4 had a 75% and 81% lower risk of being hospitalized or death, respectively, than the comparison group.
TNR4 therapy improved recovery and prevented risk of hospitalization and death among ambulatory COVID-19 cases.
Accepted: February 4, 2021 In Press Journal Pre-Proof
DOI: https://doi.org/10.1016/j.ijid.2021.02.014
On Feb 21,2021 the British Ivermectin Recommendation Development panel with 75 expert doctors, researchers and data analysts from around the world went through the studies and overwhelmingly recommended IVM for the prevention and treatment of Cov 19.
AVIGAN (FAVIPIRAVIR): 14 days of the Japanese flu drug shortens the illness. It is being studied now at Harvard’s Massachusetts General Hospital.
EIDD-2801 is investigational affecting human lung and airway cells from patients with CoV2-19: Science Translational Medicine. It introduces genetic mutations into coronavirus’ RNA. As the RNA copies itself, these damaged mutations, accumulate and render the virus unable to infect, it is an ORAL medication rather than an IV like remdesivir, so it can be administered at HOME. According to T Sheahan, PhD, Dept of Epidemiology, U of North Carolina. EIDD-2801 is also effective against OTHER RNA viruses, several strains of influenza, respiratory syncytial virus, chikungunya, Venezuelan equine encephalitis, and Eastern equine encephalitis.
Convalescent plasma. Hyperimmune globulin – GigaGen is working on this.
More information: Shilei Hao et al. QTY code-designed water-soluble Fc-fusion cytokine receptors bind to their respective ligands, QRB Discovery (2020). DOI: 10.1017/qrd.2020.4
“…The concentration of immune cells is higher in the skin than in muscle. So-called Langerhans cells are also present in the skin, and these activate and coordinate the body’s antiviral response. Christoph Rademacher’s research group at the Max Planck Institute of Colloids and Interfaces has developed a new platform technology that specifically addresses Langerhans cells: the Langerhans Cell Targeted Delivery System (LC-TDS). This system enables vaccines to be applied directly onto the skin or injected with microneedles, thereby using the immune system’s natural mechanisms. “We expect our system to be able to release all vaccines that use proteins, peptides or mRNA,” said Rademacher, main inventor of the new echnology….”
HRS, MD/this author: posited 7/1/20 s that a corona virus vaccine will be ready to distribute 10/15/20. Let’s see.
7/15/20: Newsletter Science X: Nanomaterials and vaccines: a brief and lucid and technically detailed discussion of vaccine types: https://medicalxpress.com/news/2020-07-nanomaterial-path-covid-vaccine.html?utm_source=nwletter&utm_medium=email&utm_campaign=daily-nwletter
” … 8/9/21 New government data showing that more than 99.99% of fully vaccinated Americans have not had a breakthrough infection resulting in hospitalization or death. There were 6587 COVID-19 breakthrough cases as of July 26 — including 6239 hospitalizations and 1263 deaths — among more than 163 million fully vaccinated people, according to US Centers for Disease Control and Prevention (CDC) statistics, CNN reported. That means that fewer than 0.004% of fully vaccinated people had a breakthrough infection that required hospitalization and less than 0.001% died from a breakthrough infection …”
Monoclonal antibodies – e.g., Brii Biosciences, Tsinghua University, and 3d People’s Hospital of Shenzhen are collaborating on developing fully HUMAN NEURTRALIZING MONOCLONAL ANTIBODIES to Covid-19/CoV2-19. To speed development of potentially safe and effective treatments of CoV2-19, the FDA set up a new program – the Coronavirus Treatment Acceleration Program (CTAP) – which uses all the tools the Agency has to help get therapies to patients quickly. Health and Human Services Secretary Alex Azar said, “As part of this new program, the FDA is cutting red tape, redeploying staff, and working day and night to review requests from companies, scientists, and doctors who are working toward therapies.” FDA staff in the Center for Drug Evaluation and Research (CDER) and the Center for Biologics Evaluation and Research (CBER) are providing regulatory advice, guidance, and technical assistance as quickly as possible. And the FDA is triaging requests from developers and scientists working on new drugs and biologics. Other drugs worth watching include:
Fujifilm/Toyama Chemical’s AVIGAN (FAVIPIRAVIR) – The company started a Phase III trial in Japan of this antiviral flu drug, an RNA polymerase inhibitor, to see if it shortens Covid-19 recovery time. Trends In Medicine 4/1/20/Coronavirus
NeuroRx and Relief Therapeutics’ AVIPTADIL – The FDA gave the green light for the start of a Phase II trial of this erectile dysfunction drug to treat acute respiratory distress in Covid-19 patients. It “is a synthetic form of human vasoactive intestinal peptide (VIP) & is expected to reduce inflammation in the lungs/protect alveolar type II cells in those with severe acute respiratory syndrome (SARS) due to coronavirus 2 (CoV2-19).”
Roche’s Activase (ALTEPLASE, tPA) – An article, published in the Journal of Trauma and Acute Care Surgery, suggests that this stroke drug might be useful in Covid-19-associated acute respiratory distress syndrome (ARDS), particularly in patients who need a ventilator but can’t get one. Their reasoning: “The risk of adverse events…is far outweighed by the certainty of death in patients meeting the eligibility criteria for this treatment.” It has been found beneficial but heparin was not after the alteplase infusion A 12-patient compassionate-use study is planned.
First RCT in COVID Anticoagulation Says Go Full Dose
— Respiratory outcomes better, but 20-person trial far from conclusive
by Crystal Phend, MedPage Today 9/25/20 Therapeutic-level dosing of enoxaparin (Lovenox) improved respiratory outcomes in severe COVID-19, a pilot randomized trial showed.
Gas exchange measured by the PaO2/FiO2 ratio improved significantly over time in the 10-patient therapeutic group (from 163 at baseline to 209 at 7 days and 261 at 14 days, P=0.0004) but not in the 10-patient control group receiving lower prophylactic-level doses in the open-label study (184, 168, and 195, respectively, P=0.487).
Therapeutic dosing also led to four-fold more patients being weaned off of mechanical ventilation (P=0.031) and more ventilator-free days (15 vs 0 days, P=0.028), Carlos Henrique Miranda, MD, PhD, of São Paulo University in Brazil, and colleagues reported in Thrombosis Research.
There were no major bleeding events, but numerically more minor bleeding with the higher dose anticoagulation.
“It’s a remarkable step forward in the sense that now for the first time we are having randomized trial data related to antithrombotic therapy for COVID-19,” commented Behnood Bikdeli, MD, of Brigham and Women’s Hospital and Harvard in Boston.
“It’s such a heated debate,” he said. Proponents cite mechanistic reasons for why low molecular weight heparin like enoxaparin should help in COVID-19 (one recent study showed that heparin blocks SARS-CoV-2 from binding with cells): HRS adds that heparin is negatively charged and makes platelets repel each other, thereby also reducing clotting. Opponents cite retrospective data like that from a small study suggesting higher mortality with preemptive therapeutic dose anticoagulation.
Miranda and colleagues’ assigned 20 patients with severe COVID-19 and elevated D-dimer (>1,000 μg/L) who required mechanical ventilation.
VITAMIN C – A meta-analysis of 8 studies in the Journal of Intensive Care, found that giving vitamin C (4-12 grams a day) to ICU patients on a ventilator reduced the length of time on the ventilator by 14% vs. control. The patients with the most benefit from vitamin C were those on the ventilator the longest. A person in good health maintains a normal plasma vitamin C level with an intake of ~0.1g/day. Critically ill ventilator patients may need much higher doses – grams/day.”
“A two-week course of antiviral therapy with INTERFERON BETA 1b plus LOPINAVIR- RITONAVIR and RIBAVIRIN, started within 7 days of showing COVID-19 symptoms, is safe and more effective at reducing the duration of viral shedding than lopinavir-ritonavir alone in patients with mild to moderate illness, according to the first randomized trial of this triple combination therapy involving 127 adults (aged 18 and older) from six public hospitals in Hong Kong.”
U of Toronto’s Eleanor Fish + Q Zhou at Union Hospital in Wuhan, China, as reported 7/21/20 in MD Linx: The Scientist used “IFN-alpha-2b, first approved by the US FDA for the treatment of cancer in 1986, thanks to its immunomodulatory, antiproliferative, and antiangiogenic effects. The researchers tested the IFN along with arbidol, a widely used broad-spectrum antiviral drug, in 77 patients who were admitted to Union Hospital in January and February, 2020, with a confirmed SARS-CoV-2 infection. Each of them had moderate symptoms and none required intensive care.
The results, published 5/15/20 in Frontiers in Immunology, found that patients treated with IFN-alpha-2b alone or in combination with arbidol cleared the virus from their upper airways an average of seven days faster than the group given arbidol alone. Hand-in-hand with that, blood levels of inflammatory markers such as interleukin-6 and C-reactive protein were dramatically reduced in patients receiving IFN-alpha-2b. Fish adds that as-yet unpublished data show that IFN-alpha-2b also limited lung abnormalities as evidenced by CT scans.” Prolonged interferon use does not allow for normal lung repair, however.
Preventive Medicine Center general suggestions and thoughts based on fact, judgment, reasoning, and experience:
Avoid MILK-DAIRY products 100 (100!!!) %. My belief is that ANY MILK-DAIRY thickens the mucus reducing clearance of the invading virus, allowing it to “settle in and invade.” It is my belief-knowledge that a single drop of any milk dairy begins this allergic type adverse pathway. It is 100% milk-dairy avoidance or incorrectly have as much as you want. SWEETS, including dried fruits, and juices except for Pom Wonderful pomegranate juice, function as sweets = sugar = reduce/immobilize immune functioning at multiple levels. Basically, consume an organic unprocessed whole foods diet, ideally “macrobiotic” grains-vegetables-beans-fruit-nuts-seeds = GVBfns. See the www.thepmc.org website for general wellness information + this paper + how to prevent and/or reverse where possible high blood pressure, diabetes, high triglycerides, overweight at the 95+% level and the need for open heart surgery, angioplasty.
Read http://williamspear.com/2020/03/12/covid-19/ Bill Spear’s summary letter on CoV2-19 & his Macrobiotics Primer: Bill states in a personal letter to me (minimally edited) 4/5/20: “As we know, the virus isn’t actually “living”, it’s just anxious to find a host in your lungs, and when it gets there all hell breaks loose. So, the real job of prevention is to strengthen the host’s blood supply to cells, i.e., alkalinity (HRS states that is similar to the hydroxychloroquine discussion above). My layman’s point of view is that just as the fatty outer coating is broken by sudsy, soapy wash (and stronger) externally, acidic blood breaks that cellular fatty wall internally releasing the cascade of inflammation and lung damage that ensues. Whether that’s accurate or not, relevantly I know of long-time macrobiotic people who are caring for CoV2-19 positive non-macrobiotic family members in the same house, and they all have experienced only very minor symptoms. That may not be causal insofar as their seeming “immunity” but such an interpretation is reasonable”-HRS agrees.
For cooking, rely on The Changing Seasons Cookbook. Make 1 recipe EXACTLY according to directions-avoid as many processed foods, and wheat products as possible therein. Organic MISO, tamari, rice noodles are processed and acceptable/even desired. Take the recipe with you to the natural food store. Be sure to get the exact ingredients in that one recipe. Miso soup with kombu, millet + cauliflower, scallions and daikon; brown rice with pickled shiso are specifically recommended for now as is live refrigerated organic sauerkraut. CLEANING solutions: 4 teaspoons of bleach in a quart of water, 0.125% peroxide, 80% ethanol, and 75% isopropyl alcohol are effective cleaners that kill the virus.
MEDICINE, SUPPLEMENT, AND GENERAL CONSIDERATIONS HERE ARE TO BE SPECIFICALLY DECIDED ON BETWEEN YOU AND YOUR PHYSICIAN: These Preventive Medicine Center thoughts are “invitations to consider” and require your personal judgment. If there are questions or concerns, please contact the Preventive Medicine Center. Usual suggestions are that supplements be taken daily for 2 weeks and then 5 days a week thereafter. Chew gum to keep your throat lubricated in order to “wash out” the virus. For colds or CoV2-19: the PMC position is to take vitamin C 500 mg 3 times a day, and in treatment 4-12 grams IV vitamin C per day reduced respirator use 25%, vitamin D3 5,000 units a day 5 days a week. Maturitas “Immune Role of Vitamins…” by H Shakoor 8/10/20: “….Vitamin D is a fat-soluble steroid hormone precursor that arises from ultraviolet B (UVB) radiation exposure of 7-dehydrocholesterol (7-DHC) in the epidermis of the skin, where it is transformed into the circulating precursor cholecalciferol. In the liver, cholecalciferol is hydroxylated to form 25-hydroxyvitamin D, which is transformed into the active hormone 1,25-hydroxyvitamin D (1,25(OH)2D) in the kidneys. Vitamin D has roles in a wide range of body systems, including in both innate and adaptive immune responses as shown in Fig. 2. Vitamin D enhances innate cellular immunity through stimulation of expression of anti-microbial peptides, such as cathelicidin and defensins. Defensins maintain tight and gap junctions, adherens and enhance the expression of anti-oxidative genes. Viruses such as influenza are known to significantly damage the integrity of epithelial tight junctions increasing the risk of infection and pulmonary oedema. Vitamin D is known to maintain the integrity of these junctions [14]; with low levels of vitamin D receptor expression leading to increased expression of claudin-2 and inflammation. Vitamin D also promotes the differentiation of monocytes to macrophages whilst increasing superoxide production, phagocytosis and bacterial destruction. In addition, vitamin D is able to modulate the adaptive immune response, by suppressing T helper type-1 (Th1) cell function and decreasing the production of pro-inflammatory cytokines IL-2 and interferon-gamma (INF-γ). Vitamin D also promotes anti-inflammatory cytokines by Th2 cells and indirectly suppressing Th1 cells diverting pro-inflammatory cells to an anti-inflammatory phenotype as well as stimulating suppressive regulatory T cells [15].”
This a review of Vitamin C, D, Zinc, … mechanisms of action:
https://www.sciencedirect.com/science/article/pii/S0378512220303467?fbclid=IwAR0F9-TtrF3yNSLSP7rs9ewPQSv0hbOb3yaflQBv271AUcCJSdm2ui0pA2g
Immune Renew (a yeast based immune stimulating beta glucan) 2 twice a day (Host Defense & OM manufacturers also have beta glucan immune stimulating products), as is Brewer’s yeast. AHCC 2 twice a day (as just said, 5 days a week) is the top selling supplement in Japan. Manuka honey has anti-bacterial and possibly anti-viral properties. Pau d’arco is an herbal anti-inflammatory as is nano-curcumin. Berberine functions similarly to metformin, spirulina is the origin of phycocyanobilin -> anti-inflammatory heme oxygenase production, & glucosamine. Singulair (montelukast) is a lung leukotriene inhibitor that reduces lung inflammation and is worth considering in the armamentarium. If you are taking high blood pressure medication, try to have it be an ARB (angiotensin receptor blocker such as losartan). If on cholesterol lowering medicine, Livalo/pitavastatin seems more beneficial than Crestor/rosuvastatin or Lipitor/atorvastatin. Personally, my guess is that the gout treatment medication allopurinol would be helpful for serious CoV2-1 9 infection.
Re statins: AJC “Relation of Statin Use Prior to Admission to Severity and Recovery Among COVID-19 Inpatients” L B Daniels et al, AJC 2021:149-155with 30 references. & “Statin Treatment of COVID-19” D S Fedson AJC 2021;171-173 with 24 references.
Statin use associated with increased survival in severe COVID-19
by Columbia University Irving Medical Center 2/26/21
People who took statins to lower cholesterol were approximately 50% less likely to die if hospitalized for COVID-19, a study by physicians at Columbia University Vagelos College of Physicians and Surgeons and NewYork-Presbyterian has found.
“Our study is one of the larger studies confirming this hypothesis and the data lay the groundwork for future randomized clinical trials that are needed to confirm the benefit of statins in COVID-19,” says Aakriti Gupta, MD, a cardiologist at NewYork-Presbyterian/Columbia University Irving Medical Center and one of the co-lead authors of the study.
“If their beneficial effect bears out in randomized clinical trials, statins could potentially prove to be a low-cost and effective therapeutic strategy for COVID-19,” adds co-lead author Mahesh V. Madhavan, MD, also a cardiologist at NewYork-Presbyterian/Columbia University Irving Medical Center.
Why Look at Statins?
Gupta, Madhavan, and the study’s leadership group are cardiologists who cared for hospitalized COVID-19 patients in the spring and summer of 2020 when the first wave of the pandemic swept through New York City.
“We observed that patients who got very sick and required hospitalization had high rates of hyperinflammation and clotting,” says Elaine Wan, MD, the Esther Aboodi Assistant Professor of Medicine in Cardiology and Cardiac Electrophysiology and a cardiac electrophysiologist at NewYork-Presbyterian/Columbia University Irving Medical Center, one of the study’s senior authors.
“As cardiologists, statins naturally came to mind,” Gupta says. “In addition to their well-known cholesterol-lowering effect, statins are known for their anti-inflammatory, anticoagulant and immunomodulatory properties.”
Study Analyzed Data from Electronic Health Records
Based on their observations, the authors looked at outcomes for 2,626 patients with COVID-19 who were admitted to a quaternary academic medical center in Manhattan during the first 18 weeks of the pandemic.
The researchers compared 648 patients who regularly used statins before developing COVID-19 to 648 patients who did not use statins. Patients in each group were matched so that there were no significant differences in demographics, comorbidities, or use of other medications at home.
50% Fewer Deaths among Statin Users
Among the statin users, 96 (14.8%) died in the hospital within 30 days of admission compared with 172 (26.5%) of patients who did not use statins.
When other differences among the patients were factored in, the researchers found that statin use was significantly associated with a 50% reduction in in-hospital mortality (within 30 days). Patients on statins also tended to have lower levels of C-reactive protein, a marker of inflammation.
Statin use was not associated with a statistically significant decrease in the use of invasive mechanical ventilation (18.6% in statin users vs. 21.9%), days on a ventilator (13.5 vs 12.8), or length of hospital stay (7 vs 7).
Comparison with Other Studies
Other studies and meta-analyses from China have also suggested a survival benefit from statins among COVID-19 patients. However, these results may not apply to patients in Western countries who generally have more cardiovascular disease.
The current study is one of the larger studies confirming the association. Smaller retrospective studies out of North America and Europe have found similar results.
Randomized Clinical Trials Needed
More information: Aakriti Gupta et al, Association between antecedent statin use and decreased mortality in hospitalized patients with COVID-19, Nature Communications (2021). DOI: 10.1038/s41467-021-21553-1
Association between antecedent statin use and decreased mortality in hospitalized patients with COVID-19
• Aakriti Gupta, et al 2/26/21
Nature Communications volume 12, Article number: 1325 (2021) Cite this articleNT
1296 patients (648 statin users, 648 non-statin users) identified with 1:1 propensity-score matching, statin use is significantly associated with lower odds of the primary endpoint in the propensity-matched cohort (OR 0.47, 95% CI 0.36–0.62, p < 0.001). We conclude that antecedent statin use in patients hospitalized with COVID-19 is associated with lower inpatient mortality.
Through effects on lipid rafts in cellular membranes9,10,11,12, statins may influence viral transmission and infectivity. A number of studies have evaluated the use of statins in the treatment of pneumonia and ARDS5,14,15,16,17,18,19,20. While primary results of randomized clinical trials evaluating statins in ARDS have not indicated a benefit19,20, secondary analysis of 540 individuals from the HARP-2 (Hydroxymethylglutaryl-CoA Reductase Inhibition with Simvastatin in Acute Lung Injury to Reduce Pulmonary Dysfunction–2) trial demonstrated improved survival with statin treatment in patients with a hyperinflammatory phenotype15. The current study, in New York City shows that patients with antecedent statin use were generally older with more comorbidities, presented with lower levels of C-reactive protein at time of admission, and experienced lower inpatient mortality at 30 days in a propensity-matched cohort.
Of 2626 patients included in the analysis, 951 (36.2%) were considered antecedent statin users (Table 1). On average, patients who were prescribed statins were older [median 70 (IQR 63–79) vs. 62 (49–76) years, p < 0.001] with no significant differences in sex (p = 0.06) or race/ethnicity (p = 0.12). Patients in the statin group were significantly more likely to have Medicare or Medicaid (63.0% vs. 53.6%) insurance, and less likely to be have commercial insurance (35.4% vs. 42.5%) (p < 0.001 for both). There was no significant difference in the New York City borough of residence in the two groups.
Patients using statins were significantly more likely to have hypertension (74.0% vs. 43.3%), diabetes (55.8% vs. 26.1%), coronary artery disease (22.5% vs. 6.9%), heart failure (17.0% vs. 6.7%), and chronic kidney disease (22.0% vs. 9.6%) compared with patients not receiving statins (p < 0.001 for all). Similarly, patients receiving statins had higher rates of history of stroke/transient ischemic attack (13.9% vs. 5.6%) and atrial arrhythmias (11.0% vs. 5.6%), p < 0.001 for both. There were no significant differences in liver disease.
Patients on statins were significantly more likely to be prescribed ACEi (19.7% vs. 4.2%), angiotensin-receptor blockers (13.1% vs. 3.7%), P2Y12 inhibitors (11.9% vs. 1.1%), oral anticoagulants (20.3% vs. 12.3%), and beta-blockers (44.0% vs. 12.7%) as outpatients compared to those not taking statins (p < 0.001 for all). Of note, 77.0% of patients who were on antecedent statins and 8.6% of patients who were not on antecedent statins, received statins during hospitalization.
Among the 850 patients for whom lipid levels were available, patients receiving statins had significantly lower mean low-density lipoprotein [77.9 (60.0–107.6) vs. 88.0 (67.0–117.0)] and total cholesterol levels [157.3 (127.7–191.0) vs. 164.9 (136.0–201.9)] compared with those who were not receiving them (p < 0.01 for all).
Using 1:1 matching, a propensity-matched cohort, there were no significant differences in demographics, comorbidities, or home medications remained in the propensity-matched cohort. At the time of initial presentation, patients receiving statins were less likely to present with tachypnea (22.1% vs. 28.7%, p < 0.01). There were no significant differences in the presence of fever, tachycardia, peripheral desaturation, or hypotension on initial assessment (Table 2).
An excellent air purifier company: https://www.airpurifiersandcleaners.com/sun-pure-sp-20-portable-air-purifier. Dulera inhaler for bronchial cough issues. Zantac (or Pepcid as famotidine once daily) is off market + Zyrtec (for complete histamine blockade) twice a day for nasal congestion. Immediate (!) use of these combined antihistamines can actually stop the development of “colds.” Fish oil is generally anti-inflammatory: Carlson’s Cod Liver Oil (2 teaspoons = “a swig”) once or twice a day. Elderberry capsules for further immune enhancement. For a bothersome cough for my patients I recommend elderberry syrup 2 tsp 3 times a day. Generic or trade plain Robitussin 2 teaspoons 3 times a day as necessary also only for a bothersome cough. The DM = dextromethorphan may be deleterious in CoV2-19. For chest issues, the glutathione supporting antioxidant NAC 600 mg 2 or 3 a day. If there is a deep cough, in order to prevent scarring due to fibrosis/scarring consider taking anti-fibrosis serrapeptase 2 capsules three times a day. If there is bacterial invasion in the lungs = pneumonia development, antibiotics should be chosen based on sensitivity. HCQ (HCQ $0.40 per pill) + AZITH with zinc and D3 would be the first choice. Otherwise, if treatment is begun without a culture, doxycycline + azithromycin would be my antibiotics of choice as they also have an anti-inflammatory effect.
Treatment with Hydroxychloroquine, Azithromycin, and Combination in Patients Hospitalized with COVID-19: 7/1/20
Samia Arshad, et al
PII:S1201-9712(20)30534-8DOI:https://doi.org/10.1016/j.ijid.2020.06.099Reference:IJID 4404To ap:International Journal of Infectious Diseases 7/1/20
Of 2,541 patients in Detroit’s Henry Ford Hospital system: median total hospitalization time of 6 days (IQR: 4-10 days), 51% male, 56% African American. Overall in-hospital mortality was 18.1% (95% CI:16.6%-19.7%); by treatment: hydroxychloroquine (400 mg BID day one and 200 mg BID days two through five) + azithromycin (250 mg BID day one and then 200 mg daily days two through five) plus zinc (200 mg a day days one through five), 157/783 (20.1% [95% CI: 17.3%-23.0%]), hydroxychloroquine alone, 162/1202 (13.5% [95% CI: 11.6%-15.5%]), azithromycin alone, 33/147 (22.4% [95% CI: 16.0%-30.1%]), and neither drug, 108/409 (26.4% [95% CI: 22.2%-31.0%]). Primary cause of mortality was respiratory failure (88%); no patient had documented torsade de pointes. From Cox regression modeling, predictors of mortality were age>65 years (HR:2.6 [95% CI:1.9-3.3]), white race (HR:1.7 [95% CI:1.4-2.1]), CKD (HR:1.7 [95%CI:1.4-2.1]), reduced O2 saturation level on admission (HR:1.5 [95%CI:1.1-2.1]), and ventilator use during admission (HR: 2.2 [95%CI:1.4-3.3]). Hydroxychloroquine provided a 66% hazard ratio reduction, and hydroxychloroquine + azithromycin 71% compared to neither treatment (p < 0.001).In this multi-hospital assessment, when controlling for COVID-19 risk factors, treatment with hydroxychloroquine alone and in combination with azithromycin was associated with reduction in COVID-19 associated mortality.
Hydroxychloroquine could save up to 100,000 lives if used for COVID-19: Yale epidemiology professor
By Joshua Nelson July 21, 2020
In-hospital mortality was 18.1 percent overall; 13.5 percent with just hydroxychloroquine, 22.4 percent with azithromycin alone, and 26.4 percent with neither drug. “Our results do differ from some other studies,” Dr. Marcus Zervos, who heads the hospital’s infectious diseases unit said the patients were treated early. Risch said that most in the mainstream are not allowing people to speak about the evidence on the effectiveness of HCQ. Risch also said discussions about the drug became “political” as opposed to “medical.” Risch said, arguing that the mainstream media is not covering the benefits of hydroxychloroquine. Dr. Harvey Risch, an epidemiology professor at Yale School of Public Health, said on Tuesday that he thinks hydroxychloroquine (HCQ) could save 75,000 to 100,000 lives if the drug is widely used to treat coronavirus. “There are many doctors that I’ve gotten hostile remarks about saying that all the evidence is bad for it and, in fact, that is not true at all,” Risch said adding that he believes the drug can be used as a “prophylactic” for front-line workers, as other countries like India have done.
Risch lamented that a “propaganda war” is being waged against the use of the drug for political purposes, not based on “medical facts.” Researchers at the Henry Ford Health System in Michigan have found that early administration of HCQ makes hospitalized patients substantially less likely to die. The study, published in the International Journal of Infectious Diseases, determined that HCQ provided a “66 % hazard ratio reduction” & HCQ & azithromycin a 71 % reduction, compared with neither treatment. “Five studies, including two controlled clinical trials, have demonstrated significant major outpatient treatment efficacy,” Risch observed in an 2020 article for the American Journal of Epidemiology. “Hydroxychloroquine + azithromycin has been used as standard-of-care in more than 300,000 older adults with multicomorbidities, with estimated proportion diagnosed with cardiac arrhythmias attributable to the medications 47/100,000 users, of which estimated mortality is <20%, 9/100,000 users, compared to the 10,000 Americans now dying each week. These medications need to be widely available and promoted immediately for physicians to prescribe.”
Entered 7/5/20: Hydroxychloroquine: has significant effect in mild CoV2-19 disease: Double Blind Study at Mt. Sinai Hospital NYC
• Rachel Levantovsky &
• Nicolas Vabret
Nature Reviews Immunology volume 20, page350(2020) Chen, Z. et al. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. Preprint at medRxiv https://doi.org/10.1101/2020.03.22.20040758 (2020)
This preprint reports a double-blind, randomized clinical trial of 62 patients to assess the efficacy of hydroxychloroquine (HCQ) in mild COVID-19. Patients in the treatment arm received 400 mg HCQ per day for 5 days. Fever and cough resolved on average 1 day earlier with HCQ, although the distribution of symptomatic patients at day 0 was not even between groups. No patients receiving HCQ progressed to severe disease, whereas 4 of 31 patients in the control arm progressed. Few clinical data and no viral load measurements were reported, limiting the conclusions that can be drawn from this trial. This study suggests relative efficacy for patients with mild disease and warrants larger clinical trials, but the effects of HCQ on patients with more severe COVID-19 remain unknown.
Tetracyclines may be effective in the treatment of novel coronavirus (COVID-19) in a 4/8/20 letter to the editor online now in Pharmacotherapy. Reuters Health News 4/17/20. Drs. Mahyar Etminan and Mohit Sodhi of the U of British Columbia, Vancouver, Canada. “Tetracyclines have shown to have antiviral activity in other viruses (independent of their antibacterial activity).” They also have “powerful” anti-inflammatory effects “and, of course, inflammation is an important pathological attribute of COVID-19,” he explained. The anti-inflammatory capabilities of tetracyclines include down regulation of the NFKB pathway as well as a decrease in levels of inflammatory cytokines such as tumor necrosis factor alpha, interleukin-1-beta, and interleukin-6. These cytokines have been shown to be significantly elevated when SARS-CoV-2 is exposed to lung tissue in addition to exacerbating the pathogenesis of the infection itself, they point out. Tetracyclines also have “good absorption in the lungs, where COVID-19 attacks, and are relatively safe, safer than hydroxychloroquine.”
“For all of these reasons, we think there should also be a focus on examining this drug in clinical trials as both a prophylactic agent or treatment in early and late disease,” he said. Tetracyclines might be potential therapeutic agents for COVID-19 that are “hiding in plain sight,” write Dr. Etminan and Dr. Sodhi. “We strongly urge international research groups to consider investigating the potential therapeutic efficacy of tetracycline antibiotics in treating COVID-19.”
Read the 2020 Progress in Cardiovascular Diseases article by Mark McCarty et al. regarding nutraceuticals inhibiting NOX2, thereby stimulating type 1 interferon response via Toll Receptor 7 (TLR7). HO-1 (heme oxygenase-1) enhancement to treat RNA viruses. Discussed/“recommended” in that article are alpha lipoic acid, sulforaphane, ferulic acid, resveratrol, spirulina (phycocyanobilin). EGCG as capsules or as green tea, with white tea for its high antioxidant content.
Antivirals:
New vaccine platform for CoV2-19 4/8/20 by University of Bristol Edited for concision.
COVID-19/CoV2-19 SPIKE PROTEIN mediates cell entry. Imophoron’s ADDomer-based vaccine presents exactly (just) these parts to the immune system, giving rise to SPECIFIC antibodies in order to neutralize the virus/protect against infection.
Most COVID-19 vaccines present the ENTIRE SPIKE to the immune system, which reacts by making antibodies. This usual approach RISKS inducing antibodies that bind to the WRONG parts of the spike and could make the disease even worse. In vaccines for SARS-CoV-1 (note “1”), this sometimes resulted in severe lung tissue damage. Imophoron’s vaccine presents only very SPECIFIC parts of the spike essential for cell entry and are much less prone to this risk.
This Imophoron ADDomer platform is a new, highly adaptable, easy-to-manufacture, rapid-response platform for vaccines to combat present and future infectious diseases. It is a synthetic, self-assembling, nature-inspired virus-like particle (VLP). This type of vaccine is extremely stable and requires no refrigeration, enabling unrestricted distribution worldwide.
MIT’s SHERLOCK CoV2-19 TEST: PCR tests require complex instrumentation and are usually performed by skilled personnel in an advanced laboratory setting. An alternative method is SHERLOCK, a nucleic acid-based test developed at MIT stemming from the CRISPR gene editing tool that does not need complex instrumentation and can be read out using a paper strip akin to a pregnancy test, without any loss of sensitivity or specificity. The test is also low-cost and can be performed in less than an hour.
Frederic Garzoni, Founder/CEO at Imophoron: “We … can design and roll-out potential vaccines in about two weeks …& contribute to resolving the major health and economic threats caused by emerging viruses such as COVID-19.”
More information: Charles Vragniau et al. Synthetic self-assembling ADDomer platform for highly efficient vaccination by genetically encoded multiepitope display, Science Advances (2019). DOI: 10.1126/sciadv.aaw2853
The University of Michigan publication authored by Martha Berg implies that if there were universal anti-tuberculosis BCG immunization in the USA, the USA would have only suffered an estimated 94 deaths total, which would have been only 4% of the actual death toll of 2,467 in this country on March 29, 2020.
The report “Mandated Bacillus Calmette-Guérin (BCG) vaccination predicts flattened curves for the spread of COVID-19″ is an analysis of reports of COVID-19 cases and related deaths in more than 50 countries. Researchers say countries that have a current policy mandating the anti-TB BCG vaccination have significantly slower growth of both cases and deaths, as compared to all other countries. This vaccination may or may not be related to these statistics, but it does affect general immunity.
AbbVie: the company is collaborating with select health authorities and institutions to determine the antiviral activity of lopinavir/ritonavir (Kaletra) against COVID-19.
AIM ImmunoTech: developing Ampligen, a broad-spectrum antiviral that will be tested as a potential treatment for COVID-19 in Japan. A significant survival effect was observed in a trial evaluating mice infected with the earlier Severe Acute Respiratory Syndrome (SARS) coronavirus.
Gilead: remdesivir (costs $4000.00 as opposed to HCQ at $0.40/pill and azithromycin = $0.63/pill), a broad-spectrum intravenous antiviral agent that is being investigated in a double-blind, placebo-controlled study sponsored by the National Institute of Allergy and Infectious Diseases, part of the National Institutes of Health (NIH). In addition, Gilead is initiating two phase 3 trials to evaluate the safety and efficacy of remdesivir in adults diagnosed with COVID-19, following a rapid review and acceptance by the Food and Drug Administration (FDA) of the investigational new drug filing for the novel antiviral.
Immunotherapies and other investigational therapies:
The Israeli company MIGAL (see further below) said it HAS A VACCINE that could be finalized in May and ready for distribution in 80 days. J Craig VENTER, the team leader who first sequenced the human genome and an originator of chromosome insertion, has his own California institute that I thought would quickly develop an efficient CoV2-19 testing and an effective CoV2-19 vaccine: this has not yet happened. Distributed Bio/Dr Jacob GLANVILLE is using computational-guided immune-engineering to create an antibody that neutralizes the virus in 20 minutes. It binds the spot that the virus uses to gain entry into your cells. “We have generated extremely potent picomolar antibodies that block known neutralizing ACE2 epitopes, blocking the novel coronavirus-19 from infecting human cells.”
Algernon Pharmaceuticals: developing ifenprodil, an N-methyl-d-aspartate (NDMA) receptor glutamate receptor antagonist, which is being prepared for US clinical trials for COVID-19 based on results of an animal study that showed the investigational therapy significantly reduced acute lung injury and improved survivability in H5N1 infected mice.
CEL-SCI: developing an immunotherapy using LEAPS, a patented T cell modulation peptide epitope delivery technology, to stimulate protective cell-mediated T cell responses and reduce viral load.
Innovation Pharmaceuticals: developing brilacidin, a defensin-mimetic, that mimics the human innate immune system and causes disruption of the membrane of pathogens, leading to cell death. It has already been tested in humans in phase 2 trials for other indications.
Mesoblast Limited: investigating remestemcel-L, an allogeneic mesenchymal stem cell (MSC) product candidate, as a treatment for patients with acute respiratory distress syndrome caused by COVID-19. Remestemcel-L, which is comprised of culture-expanded MSCs derived from the bone marrow of an unrelated donor, is administered in a series of intravenous infusions and is believed to have immunomodulatory properties to counteract inflammatory processes.
Q BioMed: partnering with Mannin Research to develop a potential treatment that addresses vascular leakage and endothelial dysfunction, which may potentially help patients with severe cases of COVID-19.
Takeda: developing an anti-SARS-CoV-2 polyclonal hyperimmune globulin (H-IG) to treat high-risk individuals with COVID-19 (TAK-888). Pathogen-specific antibodies from plasma will be collected from recovered patients (or vaccinated donors in the future) and will be transferred to sick patients to improve the immune response to the infection and increase the chance of recovery.
Tiziana: developing TZLS-501, which has been shown to rapidly deplete circulating levels of interleukin-6 (IL-6) in the blood, a key driver of chronic inflammation. Excessive production of IL-6 is believed to be associated with severe lung damage observed with COVID-19 infections.
Vaccines:
University of Pittsburgh in EBioMedicine “Band Aid” Vaccine.
American Ingenuity: COVID Vaccine Simply Applied to Skin Believed To Have Stopped Virus
Researchers at the U of Pittsburgh published in EBioMedicine have created a mouse-tested and easily scalable vaccine for corona virus-19 that is “delivered through a fingertip-sized patch” with “a micro-needle array” that would inject the vaccine through 400 small needles applied like a Band-Aid. The vaccine created “a surge of antibodies” sufficient to eliminate the coronavirus but hasn’t been followed long term. This U of P vaccine has potential advantages over the vaccine being tested and developed by Moderna which uses a more experimental method. This U of P vaccine was developed along the line of the flu shots, “using lab-made pieces of viral protein to build immunity.” Strange about who wins the horse race and how.
From Trends -In-Medicine: Altimmune Inc: developing a single-dose, intranasal vaccine against COVID-19 using its proprietary NasoVAX technology. The vaccine is moving toward animal testing.
Applied DNA Sciences: collaborating with Takis Biotech to develop a DNA vaccine candidate using PCR-based DNA (“LinearDNA”) manufacturing systems; preclinical testing in animals is expected to begin by July, 2020.
Codagenix Inc: co-developing a live-attenuated vaccine with the Serum Institute of India using viral deoptimization.
GlaxoSmithKline: collaborating with Clover Biopharmaceuticals to develop a protein-based corona virus vaccine candidate (COVID-19 S-Trimer) using Clover’s proprietary technology (Timer-Tag©) and combining it with GSK’s pandemic adjuvant system.
Inovio Pharmaceuticals: developing a DNA vaccine (INO-4800) to address COVID-19; human trials to begin in the US in April.
Johnson & Johnson: partnering with the Biomedical Advanced Research and Development Authority (BARDA) to develop a vaccine using Janssen’s AdVac® and PER.C6® technology, which provide the ability to rapidly upscale production of an optimal vaccine candidate. Just cleared for elease this weekend.
Moderna Inc: The Moderna vaccine has proven 95% effective in phase 3 trials
Novavax: currently evaluating multiple recombinant nanoparticle vaccine candidates in animal models; initiation of phase 1 testing is expected in late spring of 2020. The COVID-19 vaccine candidates will likely include the saponin-based Matrix-M™ adjuvant to enhance immune responses.
Sanofi: collaborating with BARDA to develop a vaccine using Sanofi’s recombinant DNA platform. The DNA sequence encoding the antigen will be combined into the DNA of the baculovirus expression platform and used to produce large quantities of the coronavirus antigen which will be formulated to stimulate the immune system to protect against the virus.
*This list is not all inclusive.
Israel: a Covid 19/CoV2-19 VACCINE
by Howard Richman 3/15/20
“Israeli scientists at the MIGAL Galilee Research Institute had worked for four years and had successfully developed a Coronavirus vaccine for chickens which passed clinical trials. When they saw the genetic sequencing of the COVID-19 virus, they realized that they could quickly adapt their chicken vaccine to the human virus. Ella Dagan, a spokesman for MIGAL told Europorter:
Dr. Shahar, one of the scientists told nocamels.com:
It’s a little bit like fate that we were working on this coronavirus vaccine at the same time that the world was suddenly hit by this epidemic of coronavirus for humans.
MIGAL created its vaccine by synthesizing two proteins. Unlike vaccines that are created by injecting a dead or weakened disease-causing virus, there is little danger that synthetic virus protein segments will give patients a disease.
Its vaccine creates antibodies in the mucosal immune system of the body which consists of thin permeable barriers to infection in the lungs, gut, eyes, nose, throat, uterus, and vagina. Dr. Chen Katz, MIGAL’s biotechnology group leader, gave Europorter a detailed cellular-level description of how MIGAL’s vaccine works:
Israel’s Minister of Science and Technology, Ofir Akunis, is expediting the human vaccine through Israel’s approval process. According to Europorter.
The minister has instructed the Director General of the Ministry of Science and Technology to fast-track all approval processes with the goal of bringing the human vaccine to market as quickly as possible.
Dr. Katz of MIGAL told Times of Israel that Israel’s approval process only involves about two months of actual testing:
The clinical testing experiments themselves are not so long, and we can complete them in 30 days, plus another 30 days for human trials. Most of the time is bureaucracy — regulation and paperwork.
Given the urgent global need for a human Coronavirus vaccine, we are doing everything we can to accelerate development. Our goal is to produce the vaccine during the by July, 2020, and to achieve safety approval in by September, 2020. This did not come to pass; Israel effectively used the Pfizer vaccine instead.
There are at least 3 available American COVID-19 vaccines in the works:
1. Moderna Therapeutics and Pfizer-BioNtech have developed synthetic mRNA double injection virus vaccines made and are now approved by NIAID (National Institute of Allergy and Infectious Diseases) and are now being widely administered. This week, Johnson and Johnson received clearance of its single injection DNA vaccine.
2. Regeneron Pharmaceuticals will soon have a treatment that will serve as a vaccine for those who don’t have coronavirus and a treatment for those who do. President Trump received these corona virus antibodies directly into the bloodstream instead of relying upon a vaccine to create those antibodies 10/1/20. A similar treatment was used to prevent and cure Ebola.
These are thoughts as of 11/22/21
—
H. Robert Silverstein, MD, FACC
Medical Director, Preventive Medicine Center
1000 Asylum Avenue #2109
Hartford, CT 06105
(860) 549-3444 or (800) 789-PREV fax (860) 549-3569
http://www.thepmc.org